Abstract
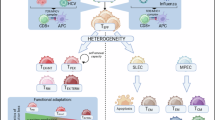
Virus-specific CD8+ T cells that differentiate in the context of resolved versus persisting infections exhibit divergent phenotypic and functional characteristics, which suggests that their differentiation trajectories are governed by distinct cellular dynamics, developmental pathways and molecular mechanisms. For acute infection, it is long known that antigen-specific T cell populations contain terminally differentiated effector T cells, known as short-lived effector T cells, and proliferation-competent and differentiation-competent memory precursor T cells. More recently, it was identified that a similar functional segregation occurs in chronic infections. A failure to generate proliferation-competent precursor cells in chronic infections and tumors results in the collapse of the T cell response. Thus, these precursor cells are major therapeutic and prophylactic targets of immune interventions. These observations suggest substantial commonality between T cell responses in acute and chronic infections but there are also critical differences. We are therefore reviewing the common features and peculiarities of precursor cells in acute infections, different types of persistent infection and cancer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
McLane, L. M., Abdel-Hakeem, M. S. & Wherry, E. J. CD8+ T cell exhaustion during chronic viral infection and cancer. Annu Rev. Immunol. 37, 457–495 (2019).
Blank, C. U. et al. Defining ‘T cell exhaustion’. Nat. Rev. Immunol. 19, 665–674 (2019).
Franco, F., Jaccard, A., Romero, P., Yu, Y. R. & Ho, P. C. Metabolic and epigenetic regulation of T cell exhaustion. Nat. Metab. 2, 1001–1012 (2020).
Hashimoto, M. et al. CD8+ T cell exhaustion in chronic infection and cancer: opportunities for interventions. Annu. Rev. Med. 69, 301–318 (2018).
Thommen, D. S. & Schumacher, T. N. T cell dysfunction in cancer. Cancer Cell 33, 547–562 (2018).
Philip, M. & Schietinger, A. CD8+ T cell differentiation and dysfunction in cancer. Nat. Rev. Immunol. https://doi.org/10.1038/s41577-021-00574-3 (2021).
Kallies, A., Zehn, D. & Utzschneider, D. T. Precursor exhausted T cells: key to successful immunotherapy? Nat. Rev. Immunol. 20, 128–136 (2020).
Klenerman, P. & Oxenius, A. T cell responses to cytomegalovirus. Nat. Rev. Immunol. 16, 367–377 (2016).
Welten, S. P. M. et al. TCF-1+ cells are required to maintain the inflationary T cell pool upon MCMV infection. Nat. Commun. 11, 2295 (2020).
Williams, M. A., Holmes, B. J., Sun, J. C. & Bevan, M. J. Developing and maintaining protective CD8+ memory T cells. Immunol. Rev. 211, 146–153 (2006).
Paley, M. A. et al. Progenitor and terminal subsets of CD8+ T cells cooperate to contain chronic viral infection. Science 338, 1220–1225 (2012).
He, R. et al. Follicular CXCR5-expressing CD8+ T cells curtail chronic viral infection. Nature 537, 412–428 (2016).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Utzschneider, D. T. et al. T cell factor 1-expressing memory-like CD8+ T cells sustain the immune response to chronic viral infections. Immunity 45, 415–427 (2016). Together, the studies by He et al., Im et al. and Utzschneider et al. identified a subset of virus-specific CD8+ T cells, characterized by the expression of TCF-1 or CXCR5, that exhibits proliferative capacity and stem-cell traits such as self-renewal and the ability to differentiate into terminally differentiated cells. This population preferentially expanded after blockade of the PD-1 inhibitory pathway.
Leong, Y. A. et al. CXCR5+ follicular cytotoxic T cells control viral infection in B cell follicles. Nat. Immunol. 17, 1187–1196 (2016).
Wu, T. et al. The TCF-1–Bcl6 axis counteracts type I interferon to repress exhaustion and maintain T cell stemness. Sci. Immunol. https://doi.org/10.1126/sciimmunol.aai8593 (2016).
Lugli, E., Galletti, G., Boi, S. K. & Youngblood, B. A. Stem, effector and hybrid states of memory CD8+ T Cells. Trends Immunol. 41, 17–28 (2020).
Omilusik, K. D. & Goldrath, A. W. Remembering to remember: T cell memory maintenance and plasticity. Curr. Opin. Immunol. 58, 89–97 (2019).
Martin, M. D. & Badovinac, V. P. Defining memory CD8+ T cell. Front. Immunol. 9, 2692 (2018).
Ahmed, R., Bevan, M. J., Reiner, S. L. & Fearon, D. T. The precursors of memory: models and controversies. Nat. Rev. Immunol. 9, 662–668 (2009).
Zebley, C. C., Gottschalk, S. & Youngblood, B. Rewriting history: epigenetic reprogramming of CD8+ T cell differentiation to enhance immunotherapy. Trends Immunol. 41, 665–675 (2020).
Chang, J. T., Wherry, E. J. & Goldrath, A. W. Molecular regulation of effector and memory T cell differentiation. Nat. Immunol. 15, 1104–1115 (2014).
Wherry, E. J., Barber, D. L., Kaech, S. M., Blattman, J. N. & Ahmed, R. Antigen-independent memory CD8+ T cells do not develop during chronic viral infection. Proc. Natl Acad. Sci. USA 101, 16004–16009 (2004).
Wherry, E. J., Blattman, J. N., Murali-Krishna, K., van der Most, R. & Ahmed, R. Viral persistence alters CD8+ T cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 77, 4911–4927 (2003).
Utzschneider, D. T. et al. High antigen levels induce an exhausted phenotype in a chronic infection without impairing T cell expansion and survival. J. Exp. Med. 213, 1819–1834 (2016).
Sandu, I., Cerletti, D., Claassen, M. & Oxenius, A. Exhausted CD8+ T cells exhibit low and strongly inhibited TCR signaling during chronic LCMV infection. Nat. Commun. 11, 4454 (2020).
Welten, S. P. M., Sandu, I., Baumann, N. S. & Oxenius, A. Memory CD8+ T cell inflation vs tissue-resident memory T cells: same patrollers, same controllers? Immunol. Rev. 283, 161–175 (2018).
Grassmann, S. et al. Early emergence of T central memory precursors programs clonal dominance during chronic viral infection. Nat. Immunol. 21, 1563–1573 (2020).
Schober, K. et al. Reverse TCR repertoire evolution toward dominant low-affinity clones during chronic CMV infection. Nat. Immunol. 21, 434–441 (2020).
Toth, I. et al. Decreased frequency of CD73+CD8+ T cells of HIV-infected patients correlates with immune activation and T cell exhaustion. J. Leukoc. Biol. 94, 551–561 (2013).
Jeannet, G. et al. Essential role of the Wnt pathway effector Tcf-1 for the establishment of functional CD8+ T cell memory. Proc. Natl Acad. Sci. USA 107, 9777–9782 (2010).
Zhou, X. et al. Differentiation and persistence of memory CD8+ T cells depend on T cell factor 1. Immunity 33, 229–240 (2010).
Beltra, J. C. et al. Developmental relationships of four exhausted CD8+ T cell subsets reveals underlying transcriptional and epigenetic landscape control mechanisms. Immunity 52, 825–841 (2020). In this report, the authors defined, based on transcriptional and epigenetic analyses, a CD8+ T cell differentiation landscape of exhausted CD8+ T cells during chronic viral infection, which involves four differentiation stages; these comprise two TCF-1+ progenitor subsets that gradually lose TCF-1 upon division and convert into a third intermediate population, which eventually differentiates into a fourth and terminally differentiated subset.
Kanev, K. et al. Proliferation-competent TCF-1+ CD8+ T cells in dysfunctional populations are CD4+ T cell help independent. Proc. Natl Acad. Sci. USA 116, 20070–20076 (2019).
Zander, R. et al. CD4+ T cell help is required for the formation of a cytolytic CD8+ T cell subset that protects against chronic infection and cancer. Immunity 51, 1028–1042 (2019). In this report, the authors demonstrate that the effector-like CX3CR1-expressing CD8+ T cell subset is required for viral control and that the differentiation of this population depends on CD4+ T cell provision of IL-21.
Hudson, W. H. et al. Proliferating transitory T cells with an effector-like transcriptional signature emerge from PD-1+ stem-like CD8+ T cells during chronic infection. Immunity 51, e1044 (2019).
Utzschneider, D. T. et al. T cells maintain an exhausted phenotype after antigen withdrawal and population reexpansion. Nat. Immunol. 14, 603–610 (2013). This study established that T cells undergo a stable and inheritable form of differentiation in chronic infection and that cells obtained from chronic infection retained their exhausted phenotype following transfer and re-expansion in acute infections.
Pauken, K. E. et al. Epigenetic stability of exhausted T cells limits durability of reinvigoration by PD-1 blockade. Science 354, 1160–1165 (2016). This study demonstrated that Tex cells acquire a stable epigenetic signature that is maintained even after PD-L1 blockade, suggesting that limited plasticity of the epigenetic state may limit the long-term reprogramming of exhausted CD8+ T cells.
Ghoneim, H. E. et al. De novo epigenetic programs inhibit PD-1 blockade-mediated T cell rejuvenation. Cell 170, 142–157 (2017). This study showed that blocking de novo DNA methylation sustained effector functions in activated CD8+ T cells during persistent viral infection and in the context of cancer, demonstrating that de novo DNA methylation imprints T cell exhaustion and therefore limits T cell rejuvenation by inhibitory receptor blockade.
Philip, M. et al. Chromatin states define tumour-specific T cell dysfunction and reprogramming. Nature 545, 452–456 (2017).
Angelosanto, J. M., Blackburn, S. D., Crawford, A. & Wherry, E. J. Progressive loss of memory T cell potential and commitment to exhaustion during chronic viral infection. J. Virol. 86, 8161–8170 (2012).
Utzschneider, D. T. et al. Early precursor T cells establish and propagate T cell exhaustion in chronic infection. Nat. Immunol. 21, 1256–1266 (2020). This study established that TCF-1+ precursors with an exhausted phenotype can be detected 5 days after infection in chronic infections.
Guo, Y. et al. Metabolic reprogramming of terminally exhausted CD8+ T cells by IL-10 enhances antitumor immunity. Nat. Immunol. 22, 746–756 (2021).
Gabriel, S. S. et al. Transforming growth factor-beta-regulated mTOR activity preserves cellular metabolism to maintain long-term T cell responses in chronic infection. Immunity 54, 1698–1714 (2021). In this report, the authors demonstrate that Tpex cells sustain mitochondrial fitness, while Tex cells deteriorate metabolically over time, with Tpex cells exhibiting TGFβ-mediated suppression of mTOR kinase signaling but retaining the ability to activate the mTOR pathway upon TCR signaling.
Martinez, G. J. et al. The transcription factor NFAT promotes exhaustion of activated CD8+ T cells. Immunity 42, 265–278 (2015).
Alfei, F. et al. TOX reinforces the phenotype and longevity of exhausted T cells in chronic viral infection. Nature 571, 265–269 (2019).
Khan, O. et al. TOX transcriptionally and epigenetically programs CD8+ T cell exhaustion. Nature 571, 211–218 (2019).
Scott, A. C. et al. TOX is a critical regulator of tumour-specific T cell differentiation. Nature 571, 270–274 (2019).
Yao, C. et al. Single-cell RNA-seq reveals TOX as a key regulator of CD8+ T cell persistence in chronic infection. Nat. Immunol. 20, 890–901 (2019). All four studies have in parallel identified TOX as a key transcription factor for the generation or maintenance of exhausted T cell populations.
Seo, H. et al. TOX and TOX2 transcription factors cooperate with NR4A transcription factors to impose CD8+ T cell exhaustion. Proc. Natl Acad. Sci. USA 116, 12410–12415 (2019).
Scott-Browne, J. P. et al. Dynamic changes in chromatin accessibility occur in CD8+ T cells responding to viral infection. Immunity 45, 1327–1340 (2016). In this report, the authors perform genome-wide comparisons of chromatin accessibility and gene expression in virus-specific CD8+ T cells during chronic infection and report that exhausted CD8+ T cells exhibit distinct and stable changes in chromatin accessibility comprising consensus binding sites for NFAT and NR4A family members.
Beltra, J. C. et al. IL2Rβ-dependent signals drive terminal exhaustion and suppress memory development during chronic viral infection. Proc. Natl Acad. Sci. USA 113, E5444–E5453 (2016).
Snell, L. M. et al. CD8+ T cell priming in established chronic viral infection preferentially directs differentiation of memory-like cells for sustained. Immunity 49, 678–694 (2018).
Sun, J. C. & Bevan, M. J. Defective CD8+ T cell memory following acute infection without CD4+ T cell help. Science 300, 339–342 (2003).
Wieland, D. et al. TCF-1+ hepatitis C virus-specific CD8+ T cells are maintained after cessation of chronic antigen stimulation. Nat. Commun. 8, 15050 (2017). The study found in humans that a TCF-1+CD127+PD-1+ HCV-specific CD8+ T cell subset has memory-like characteristics, including antigen-independent survival and recall proliferation.
Galletti, G. et al. Two subsets of stem-like CD8+ memory T cell progenitors with distinct fate commitments in humans. Nat. Immunol. 21, 1552–1562 (2020). This study identified that the human memory T cell pool includes a subpopulation that expresses signature genes of T cell exhaustion.
Kusnadi, A. et al. Severely ill COVID-19 patients display impaired exhaustion features in SARS-CoV-2-reactive CD8+ T cells. Sci. Immunol. 6, eabe4782 (2021).
Reina-Campos, M., Scharping, N. E. & Goldrath, A. W. CD8+ T cell metabolism in infection and cancer. Nat. Rev. Immunol. 21, 718–738 (2021).
Barber, D. L. et al. Restoring function in exhausted CD8+ T cells during chronic viral infection. Nature 439, 682–687 (2006).
Oestreich, K. J., Yoon, H., Ahmed, R. & Boss, J. M. NFATc1 regulates PD-1 expression upon T cell activation. J. Immunol. 181, 4832–4839 (2008).
Kao, C. et al. Transcription factor T-bet represses expression of the inhibitory receptor PD-1 and sustains virus-specific CD8+ T cell responses during chronic infection. Nat. Immunol. 12, 663–671 (2011).
Frebel, H. et al. Programmed death 1 protects from fatal circulatory failure during systemic virus infection of mice. J. Exp. Med. 209, 2485–2499 (2012).
Odorizzi, P. M., Pauken, K. E., Paley, M. A., Sharpe, A. & Wherry, E. J. Genetic absence of PD-1 promotes accumulation of terminally differentiated exhausted CD8+ T cells. J. Exp. Med. 212, 1125–1137 (2015).
Chen, Z. et al. TCF-1-centered transcriptional network drives an effector versus exhausted CD8+ T cell-fate decision. Immunity 51, 840–855 (2019). This report showed that PD-1 expression on virus-specific CD8+ T cells early during chronic viral infection supported TCF-1 expression, which stabilized the Tpex cell pool via promoting EOMES and c-MYB expression, being relevant for Bcl-2 expression and survival. These data identified PD-1 as a protector of the early established TCF-1+ memory-like subset during chronic viral infection.
Menner, A. J. et al. Id3 controls cell death of 2B4+ virus-specific CD8+ T cells in chronic viral infection. J. Immunol. 195, 2103–2114 (2015).
Jadhav, R. R. et al. Epigenetic signature of PD-1+ TCF-1+ CD8+ T cells that act as resource cells during chronic viral infection and respond to PD-1 blockade. Proc. Natl Acad. Sci. USA 116, 14113–14118 (2019).
Man, K. et al. Transcription factor IRF4 promotes CD8+ T cell exhaustion and limits the development of memory-like T cells during chronic infection. Immunity 47, 1129–1141 (2017).
Seo, H. et al. BATF and IRF4 cooperate to counter exhaustion in tumor-infiltrating CAR T cells. Nat. Immunol. https://doi.org/10.1038/s41590-021-00964-8 (2021).
Lynn, R. C. et al. c-Jun overexpression in CAR T cells induces exhaustion resistance. Nature 576, 293–300 (2019).
Yao, C. et al. BACH2 enforces the transcriptional and epigenetic programs of stem-like CD8+ T cells. Nat. Immunol. 22, 370–380 (2021).
Wagle, M. V. et al. Antigen-driven EGR2 expression is required for exhausted CD8+ T cell stability and maintenance. Nat. Commun. 12, 2782 (2021).
Pearce, E. L. et al. Control of effector CD8+ T cell function by the transcription factor Eomesodermin. Science 302, 1041–1043 (2003).
Intlekofer, A. M. et al. Effector and memory CD8+ T cell fate coupled by T-bet and Eomesodermin. Nat. Immunol. 6, 1236–1244 (2005).
McLane, L. M. et al. Role of nuclear localization in the regulation and function of T-bet and Eomes in exhausted CD8+ T cells. Cell Rep. 35, 109120 (2021).
Buggert, M. et al. T-bet and Eomes are differentially linked to the exhausted phenotype of CD8+ T cells in HIV infection. PLoS Pathog. 10, e1004251 (2014).
Chen, Y. et al. BATF regulates progenitor to cytolytic effector CD8+ T cell transition during chronic viral infection. Nat. Immunol. https://doi.org/10.1038/s41590-021-00965-7 (2021).
Shin, H. et al. A role for the transcriptional repressor Blimp-1 in CD8+ T cell exhaustion during chronic viral infection. Immunity 31, 309–320 (2009).
Chen, J. et al. NR4A transcription factors limit CAR T cell function in solid tumours. Nature 567, 530–534 (2019).
Yates, K. B. et al. Epigenetic scars of CD8+ T cell exhaustion persist after cure of chronic infection in humans. Nat. Immunol. 22, 1020–1029 (2021).
Tonnerre, P. et al. Differentiation of exhausted CD8+ T cells after termination of chronic antigen stimulation stops short of achieving functional T cell memory. Nat. Immunol. 22, 1030–1041 (2021).
Abdel-Hakeem, M. S. et al. Epigenetic scarring of exhausted T cells hinders memory differentiation upon eliminating chronic antigenic stimulation. Nat. Immunol. 22, 1008–1019 (2021).
Hensel, N. et al. Memory-like HCV-specific CD8+ T cells retain a molecular scar after cure of chronic HCV infection. Nat. Immunol. 22, 229–239 (2021). The studies by Tonnerre et al., Abdel-Hakeem et al. and Hensel et al. identified that the molecular signature of T cell exhaustion is maintained in T cells in mice and human HCV-specific CD8+ T cells even after the cessation of chronic antigen stimulation.
Schuch, A. et al. Phenotypic and functional differences of HBV core-specific versus HBV polymerase-specific CD8+ T cells in chronically HBV-infected patients with low viral load. Gut 68, 905–915 (2019).
Kefalakes, H. et al. Hepatitis D virus-specific CD8+ T cells have a memory-like phenotype associated with viral immune escape in patients with chronic hepatitis D virus infection. Gastroenterology 156, 1805–1819 (2019).
Karimzadeh, H. et al. Mutations in hepatitis D virus allow it to escape detection by CD8+ T cells and evolve at the population level. Gastroenterology 156, 1820–1833 (2019).
Sen, D. R. et al. The epigenetic landscape of T cell exhaustion. Science 354, 1165–1169 (2016).
Charmoy, M., Wyss, T., Delorenzi, M. & Held, W. PD-1+ TCF-1+ CD8+ T cells from established chronic infection can form memory while retaining a stableimprint of persistent antigen exposure. Cell Rep. 36, 109672 (2021).
Thimme, R. T cell immunity to hepatitis C virus: lessons for a prophylactic vaccine. J. Hepatol. 74, 220–229 (2021).
Rutishauser, R. L. et al. TCF-1 regulates HIV-specific CD8+ T cell expansion capacity. JCI Insight https://doi.org/10.1172/jci.insight.136648 (2021).
Numazaki, Y., Yano, N., Morizuka, T., Takai, S. & Ishida, N. Primary infection with human cytomegalovirus: virus isolation from healthy infants and pregnant women. Am. J. Epidemiol. 91, 410–417 (1970).
Ho, M. Epidemiology of cytomegalovirus infections. Rev. Infect. Dis. 12, S701–S710 (1990).
Reeves, M. & Sinclair, J. Regulation of human cytomegalovirus transcription in latency: beyond the major immediate-early promoter. Viruses 5, 1395–1413 (2013).
Poole, E. & Sinclair, J. Sleepless latency of human cytomegalovirus. Med. Microbiol. Immunol. 204, 421–429 (2015).
Schwartz, M. & Stern-Ginossar, N. The transcriptome of latent human cytomegalovirus. J. Virol. https://doi.org/10.1128/JVI.00047-19 (2019).
Holtappels, R., Thomas, D., Podlech, J. & Reddehase, M. J. Two antigenic peptides from genes m123 and m164 of murine cytomegalovirus quantitatively dominate CD8+ T cell memory in the H-2d haplotype. J. Virol. 76, 151–164 (2002).
Karrer, U. et al. Memory inflation: continuous accumulation of antiviral CD8+ T cells over time. J. Immunol. 170, 2022–2029 (2003).
O’Hara, G. A., Welten, S. P., Klenerman, P. & Arens, R. Memory T cell inflation: understanding cause and effect. Trends Immunol. 33, 84–90 (2012).
Beyranvand Nejad, E. et al. Demarcated thresholds of tumor-specific CD8+ T cells elicited by MCMV-based vaccine vectors provide robust correlates of protection. J. Immunother. Cancer 7, 25 (2019).
Borkner, L. et al. Immune protection by a cytomegalovirus vaccine vector expressing a single low-avidity epitope. J. Immunol. 199, 1737–1747 (2017).
Hansen, S. G. et al. Profound early control of highly pathogenic SIV by an effector memory T cell vaccine. Nature 473, 523–527 (2011).
Hansen, S. G. et al. Effector memory T cell responses are associated with protection of rhesus monkeys from mucosal simian immunodeficiency virus challenge. Nat. Med. 15, 293–299 (2009).
Karrer, U. et al. Expansion of protective CD8+ T cell responses driven by recombinant cytomegaloviruses. J. Virol. 78, 2255–2264 (2004).
Klyushnenkova, E. N. et al. A cytomegalovirus-based vaccine expressing a single tumor-specific CD8+ T cell epitope delays tumor growth in a murine model of prostate cancer. J. Immunother. 35, 390–399 (2012).
Qiu, Z. et al. Cytomegalovirus-based vaccine expressing a modified tumor antigen induces potent tumor-specific CD8+ T cell response and protects mice from melanoma. Cancer Immunol. Res. 3, 536–546 (2015).
Snyder, C. M., Cho, K. S., Bonnett, E. L., Allan, J. E. & Hill, A. B. Sustained CD8+ T cell memory inflation after infection with a single-cycle cytomegalovirus. PLoS Pathog. 7, e1002295 (2011).
Dekhtiarenko, I. et al. Peptide processing is critical for T cell memory inflation and may be optimized to improve immune protection by CMV-based vaccine vectors. PLoS Pathog. 12, e1006072 (2016).
Hutchinson, S. et al. A dominant role for the immunoproteasome in CD8+ T cell responses to murine cytomegalovirus. PLoS ONE 6, e14646 (2011).
Torti, N., Walton, S. M., Brocker, T., Rulicke, T. & Oxenius, A. Non-hematopoietic cells in lymph nodes drive memory CD8+ T cell inflation during murine cytomegalovirus infection. PLoS Pathog. 7, e1002313 (2011).
Seckert, C. K. et al. Antigen-presenting cells of haematopoietic origin prime cytomegalovirus-specific CD8+ T cells but are not sufficient for driving memory inflation during viral latency. J. Gen. Virol. 92, 1994–2005 (2011).
Baumann, N. S. et al. Early primed KLRG1− CMV-specific T cells determine the size of the inflationary T cell pool. PLoS Pathog. 15, e1007785 (2019).
Baumann, N. S. et al. Tissue maintenance of CMV-specific inflationary memory T cells by IL-15. PLoS Pathog. 14, e1006993 (2018).
Snyder, C. M. et al. Memory inflation during chronic viral infection is maintained by continuous production of short-lived, functional T cells. Immunity 29, 650–659 (2008).
Welten, S. P. M., Baumann, N. S. & Oxenius, A. Fuel and brake of memory T cell inflation. Med. Microbiol. Immunol. https://doi.org/10.1007/s00430-019-00587-9 (2019).
Quinn, M. et al. Memory T cells specific for murine cytomegalovirus re-emerge after multiple challenges and recapitulate immunity in various adoptive transfer scenarios. J. Immunol. 194, 1726–1736 (2015).
Bottcher, J. P. et al. Functional classification of memory CD8+ T cells by CX3CR1 expression. Nat. Commun. 6, 8306 (2015).
Klenerman, P. The (gradual) rise of memory inflation. Immunol. Rev. 283, 99–112 (2018).
Remmerswaal, E. B. et al. Clonal evolution of CD8+ T cell responses against latent viruses: relationship among phenotype, localization and function. J. Virol. 89, 568–580 (2015).
Miron, M. et al. Human lymph nodes maintain TCF-1hi memory T cells with high functional potential and clonal diversity throughout Life. J. Immunol. 201, 2132–2140 (2018).
Brummelman, J. et al. High-dimensional single cell analysis identifies stem-like cytotoxic CD8+ T cells infiltrating human tumors. J. Exp. Med. 215, 2520–2535 (2018). This study demonstrates that TCF-1+ stem-like T cells are also found among tumor-infiltrating T cells in humans.
Miller, B. C. et al. Subsets of exhausted CD8+ T cells differentially mediate tumor control and respond to checkpoint blockade. Nat. Immunol. 20, 326–336 (2019).
Siddiqui, I. et al. Intratumoral TCF-1+PD-1+CD8+ T cells with stem-like properties promote tumor control in response to vaccination and checkpoint blockade immunotherapy. Immunity 50, 195–211 (2019).
Gattinoni, L. et al. Wnt signaling arrests effector T cell differentiation and generates CD8+ memory stem cells. Nat. Med. 15, 808–813 (2009).
Gattinoni, L. et al. A human memory T cell subset with stem cell-like properties. Nat. Med. 17, 1290–1297 (2011).
Pilipow, K. et al. Antioxidant metabolism regulates CD8+ T memory stem cell formation and antitumor immunity. JCI Insight https://doi.org/10.1172/jci.insight.122299 (2018).
Fraietta, J. A. et al. Determinants of response and resistance to CD19 chimeric antigen receptor T cell therapy of chronic lymphocytic leukemia. Nat. Med. 24, 563–571 (2018).
Sade-Feldman, M. et al. Defining T cell states associated with response to checkpoint immunotherapy in melanoma. Cell 175, 998–1013 (2018).
Liu, B. et al. Temporal single-cell tracing reveals clonal revival and expansion of precursor exhausted T cells during anti-PD-1 therapy in lung cancer. Nat. Cancer 3, 108–121 (2022).
Thommen, D. S. et al. A transcriptionally and functionally distinct PD-1+ CD8+ T cell pool with predictive potential in non-small-cell lung cancer treated with PD-1 blockade. Nat. Med. 24, 994–1004 (2018).
Clarke, J. et al. Single-cell transcriptomic analysis of tissue-resident memory T cells in human lung cancer. J. Exp. Med. 216, 2128–2149 (2019).
Zhang, Y. et al. Single-cell analyses reveal key immune cell subsets associated with response to PD-L1 blockade in triple-negative breast cancer. Cancer Cell 39, 1578–1593 (2021).
Duhen, T. et al. Coexpression of CD39 and CD103 identifies tumor-reactive CD8+ T cells in human solid tumors. Nat. Commun. 9, 2724 (2018).
Li, H. et al. Dysfunctional CD8+ T cells form a proliferative, dynamically regulated compartment within human melanoma. Cell 176, 775–789 (2019).
Simoni, Y. et al. Bystander CD8+ T cells are abundant and phenotypically distinct in human tumour infiltrates. Nature 557, 575–579 (2018).
Oliveira, G. et al. Phenotype, specificity and avidity of antitumour CD8+ T cells in melanoma. Nature https://doi.org/10.1038/s41586-021-03704-y (2021).
Caushi, J. X. et al. Transcriptional programs of neoantigen-specific TIL in anti-PD-1-treated lung cancers. Nature https://doi.org/10.1038/s41586-021-03752-4 (2021).
Yost, K. E., Chang, H. Y. & Satpathy, A. T. Recruiting T cells in cancer immunotherapy. Science 372, 130–131 (2021).
Spitzer, M. H. et al. Systemic immunity is required for effective cancer immunotherapy. Cell 168, 487–502 (2017).
Huang, A. C. et al. A single dose of neoadjuvant PD-1 blockade predicts clinical outcomes in resectable melanoma. Nat. Med. 25, 454–461 (2019).
Huang, A. C. et al. T cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 545, 60–65 (2017).
Yost, K. E. et al. Clonal replacement of tumor-specific T cells following PD-1 blockade. Nat. Med. 25, 1251–1259 (2019).
Connolly, K. A. et al. A reservoir of stem-like CD8+ T cells in the tumor-draining lymph node preserves the ongoing antitumor immune response. Sci. Immunol. 6, eabg7836 (2021).
Schenkel, J. M. et al. Conventional type I dendric cells maintain a reservoir of proliferative tumor-antigen specific TCF-1+ CD8+ T cells in tumor-draining lymph nodes. Immunity https://doi.org/10.1016/j.immuni.2021.08.026 (2021).
Dammeijer, F. et al. The PD-1/PD-L1-checkpoint restrains T cell Immunity in tumor-draining lymph nodes. Cancer Cell 38, 685–700 (2020).
Francis, D. M. et al. Blockade of immune checkpoints in lymph nodes through locoregional delivery augments cancer immunotherapy. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aay3575 (2020).
van der Leun, A. M., Thommen, D. S. & Schumacher, T. N. CD8+ T cell states in human cancer: insights from single-cell analysis. Nat. Rev. Cancer 20, 218–232 (2020).
Sabatino, M. et al. Generation of clinical-grade CD19-specific CAR-modified CD8+ memory stem cells for the treatment of human B cell malignancies. Blood 128, 519–528 (2016).
van der Waart, A. B. et al. Inhibition of Akt signaling promotes the generation of superior tumor-reactive T cells for adoptive immunotherapy. Blood 124, 3490–3500 (2014).
Verma, V. et al. MEK inhibition reprograms CD8+ T lymphocytes into memory stem cells with potent antitumor effects. Nat. Immunol. 22, 53–66 (2021).
Weber, E. W. et al. Transient rest restores functionality in exhausted CAR T cells through epigenetic remodeling. Science https://doi.org/10.1126/science.aba1786 (2021).
Highton, A. J. et al. Single-cell transcriptome analysis of CD8+ T cell memory inflation. Wellcome Open Res. 4, 78 (2019).
Carmona, S. J., Siddiqui, I., Bilous, M., Held, W. & Gfeller, D. Deciphering the transcriptomic landscape of tumor-infiltrating CD8 lymphocytes in B16 melanoma tumors with single-cell RNA-seq. Oncoimmunology 9, 1737369 (2020).
Pauken, K. E. et al. Single-cell analyses identify circulating anti-tumor CD8+ T cells and markers for their enrichment. J. Exp. Med. https://doi.org/10.1084/jem.20200920 (2021).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587 2021).
Durinck, S., Spellman, P. T., Birney, E. & Huber, W. Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat. Protoc. 4, 1184–1191 (2009).
Acknowledgements
D.Z. is supported by a European Research Council consolidator grant (ToCCaTa) and grants from the German Research Foundation (SFB1054 and SFB1371). A.O. is supported by the Swiss National Science Foundation (grant no. IZHRZ0_180552 and grant no. 310030B_185374). E.L. is a CRI Lloyd J. Old STAR (CRI award 3914) and is supported by the Associazione Italiana per la Ricerca sul Cancro (AIRC IG 20676 and AIRC 5×1000 UniCanVax 22757). R.T. is supported by CRC/TRR 179-Project 01 and CRC 1160-Project A02.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
D.Z. has a consulting agreement and research collaboration agreement with Pieris Pharmaceuticals related to manipulation of Tpex cells. E.L. receives research grants from Bristol Myers Squibb and is inventor on a patent describing methods for the generation and isolation of Tscm cells.
Peer review
Peer review information
Nature Immunology thanks Axel Kallies and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: L. A. Dempsey, in collaboration with the Nature Immunology team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zehn, D., Thimme, R., Lugli, E. et al. ‘Stem-like’ precursors are the fount to sustain persistent CD8+ T cell responses. Nat Immunol 23, 836–847 (2022). https://doi.org/10.1038/s41590-022-01219-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41590-022-01219-w
This article is cited by
-
Regulators of CD8+ T cell exhaustion
Nature Reviews Immunology (2026)
-
Epigenetic regulation of T cell exhaustion in cancer
Nature Reviews Cancer (2026)
-
High efficiency CRISPR knock-in demonstrates that TCF1 is insufficient to reverse T cell exhaustion
Nature Communications (2026)
-
Inhibitory PD-1 axis maintains high-avidity stem-like CD8+ T cells
Nature (2026)
-
Fine-tuning BACH2 dosage balances stemness and effector function to enhance antitumor T cell therapy
Nature Immunology (2026)