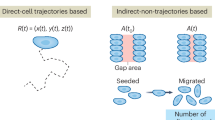
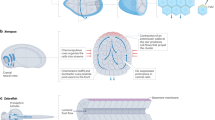
Abstract
Cell migration is a key cellular process that drives major developmental programs. To mimic and mechanistically understand cell migration in these different contexts, different assays have been developed. However, owing to the lack of practical guidelines, these different cell migration assays are often used interchangeably. This and the inherent dynamic nature of cell migration, which often requires sophisticated live-cell microscopy, may have caused cell migration to be notably less well understood than equally important cell functions, such as cell differentiation or proliferation. In this Review, we describe commonly used custom and commercial in vitro and in vivo cell migration assays and provide a comprehensive practical guide and decision tree outlining how to choose and implement an assay that best suits the biological question at hand. We hope this guidance spurs biological insights into this complex process and encourages future methods development.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Keller, R. Cell migration during gastrulation. Curr. Opin. Cell Biol. 17, 533–541 (2005).
Chuai, M., Hughes, D. & Weijer, C. J. Collective epithelial and mesenchymal cell migration during gastrulation. Curr. Genomics 13, 267–277 (2012).
Fu, X. et al. Mesenchymal stem cell migration and tissue repair. Cells 8, 784 (2019).
Wirtz, D., Konstantopoulos, K. & Searson, P. C. The physics of cancer: the role of physical interactions and mechanical forces in metastasis. Nat. Rev. Cancer 11, 512–522 (2011).
Du, W., Nair, P., Johnston, A., Wu, P. -H. & Wirtz, D. Cell trafficking at the intersection of the tumor–immune compartments. Annu. Rev. Biomed. Eng. 24, 275–305 (2022).
Friedl, P. & Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nat. Rev. Mol. Cell Biol. 10, 445–457 (2009).
Jain, S. et al. The role of single-cell mechanical behaviour and polarity in driving collective cell migration. Nat. Phys. 16, 802–809 (2020).
Sherratt, J. A. Chemotaxis and chemokinesis in eukaryotic cells: the keller-segel equations as an approximation to a detailed model. Bull. Math. Biol. 56, 129–146 (1994).
Shellard, A. & Mayor, R. Durotaxis: the hard path from in vitro to in vivo. Dev. Cell 56, 227–239 (2021).
Rens, E. G. & Merks, R. M. H. Cell shape and durotaxis explained from cell-extracellular matrix forces and focal adhesion dynamics. iScience 23, 101488 (2020).
Gruler, H. & Nuccitelli, R. Neural crest cell galvanotaxis: new data and a novel approach to the analysis of both galvanotaxis and chemotaxis. Cell Motil. Cytoskeleton 19, 121–133 (1991).
Huang, Y. -J. et al. Cellular microenvironment modulates the galvanotaxis of brain tumor initiating cells. Sci. Rep. 6, 21583 (2016).
Goswami, P. et al. Magnetotactic bacteria and magnetofossils: ecology, evolution and environmental implications. NPJ Biofilms Microbiomes 8, 43 (2022).
Gardel, M. L., Schneider, I. C., Aratyn-Schaus, Y. & Waterman, C. M. Mechanical integration of actin and adhesion dynamics in cell migration. Annu. Rev. Cell Dev. Biol. 26, 315–333 (2010).
Yamaguchi, H. & Condeelis, J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochim. Biophys. Acta 1773, 642–652 (2007).
Vicente-Manzanares, M., Ma, X., Adelstein, R. S. & Horwitz, A. R. Non-muscle myosin II takes centre stage in cell adhesion and migration. Nat. Rev. Mol. Cell Biol. 10, 778–790 (2009).
Yamada, K. M. & Sixt, M. Mechanisms of 3D cell migration. Nat. Rev. Mol. Cell Biol. 20, 738–752 (2019).
Bera, K. et al. Extracellular fluid viscosity enhances cell migration and cancer dissemination. Nature 611, 365–373 (2022).
Paul, C. D., Hung, W. C., Wirtz, D. & Konstantopoulos, K. Engineered models of confined cell migration. Annu. Rev. Biomed. Eng. 18, 159–180 (2016).
Lämmermann, T. et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008).
Lämmermann, T. & Sixt, M. Mechanical modes of ‘amoeboid’ cell migration. Curr. Opin. Cell Biol. 21, 636–644 (2009).
Wolf, K. et al. Compensation mechanism in tumor cell migration: mesenchymal–amoeboid transition after blocking of pericellular proteolysis. J. Cell Biol. 160, 267–277 (2003).
Marcadis, A. R. et al. Rapid cancer cell perineural invasion utilizes amoeboid migration. Proc. Natl Acad. Sci. USA 120, e2210735120 (2023).
Paňková, K., Rösel, D., Novotný, M. & Brábek, J. The molecular mechanisms of transition between mesenchymal and amoeboid invasiveness in tumor cells. Cell. Mol. Life Sci. 67, 63–71 (2010).
Loza, A. J. et al. Cell density and actomyosin contractility control the organization of migrating collectives within an epithelium. Mol. Biol. Cell 27, 3459–3470 (2016).
Tlili, S. et al. Collective cell migration without proliferation: density determines cell velocity and wave velocity. R. Soc. Open Sci. 5, 172421 (2018).
Kalluri, R. & Weinberg, R. A. The basics of epithelial-mesenchymal transition. J. Clin. Invest. 119, 1420–1428 (2009).
Dongre, A. & Weinberg, R. A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 20, 69–84 (2019).
Nair, P. R. et al. MLL1 regulates cytokine-driven cell migration and metastasis. Sci. Adv. 10, eadk0785 (2024).
Wu, P. H. et al. Methods to analyze cell migration data: fundamentals and practical guidelines. Nat. Methods https://doi.org/10.1038/s41592-025-02935-5 (2025).
Bananis, E., Murray, J. W., Stockert, R. J., Satir, P. & Wolkoff, A. W. Regulation of early endocytic vesicle motility and fission in a reconstituted system. J. Cell Sci. 116, 2749–2761 (2003).
Kiss, A., Horvath, P., Rothballer, A., Kutay, U. & Csucs, G. Nuclear motility in glioma cells reveals a cell-line dependent role of various cytoskeletal components. PLoS ONE 9, e93431 (2014).
Morris, N. R. Nuclear migration. From fungi to the mammalian brain. J. Cell Biol. 148, 1097–1101 (2000).
Lee, J. S., Chang, M. I., Tseng, Y. & Wirtz, D. Cdc42 mediates nucleus movement and MTOC polarization in Swiss 3T3 fibroblasts under mechanical shear stress. Mol. Biol. Cell 16, 871–880 (2005).
Jonkman, J. E. N. et al. An introduction to the wound healing assay using live-cell microscopy. Cell Adh. Migr. 8, 440–451 (2014).
Hulkower, K. I. & Herber, R. L. Cell migration and invasion assays as tools for drug discovery. Pharmaceutics 3, 107–124 (2011).
Alard, A. et al. Breast cancer cell mesenchymal transition and metastasis directed by DAP5/eIF3d-mediated selective mRNA translation. Cell Rep. 42, 112646 (2023).
Shimizu, T. et al. Indigo enhances wound healing activity of Caco-2 cells via activation of the aryl hydrocarbon receptor. J. Nat. Med. 75, 833–839 (2021).
Kim, J. H. et al. Propulsion and navigation within the advancing monolayer sheet. Nat. Mater. 12, 856–863 (2013).
Simpson, K. J. et al. Identification of genes that regulate epithelial cell migration using an siRNA screening approach. Nat. Cell Biol. 10, 1027–1038 (2008).
Chapnick, D. A. & Liu, X. Leader cell positioning drives wound-directed collective migration in TGFβ-stimulated epithelial sheets. Mol. Biol. Cell 25, 1586–1593 (2014).
Das, A. M., Eggermont, A. M. M. & ten Hagen, T. L. M. A ring barrier–based migration assay to assess cell migration in vitro. Nat. Protoc. 10, 904–915 (2015).
Wager, L. J., Murray, R. Z., Thompson, E. W. & Leavesley, D. I. A fence barrier method of leading edge cell capture for explorative biochemical research. Cell Adh. Migr. 11, 496–503 (2017).
Kramer, N. et al. In vitro cell migration and invasion assays. Mutat. Res. 752, 10–24 (2013).
Yarrow, J. C., Perlman, Z. E., Westwood, N. J. & Mitchison, T. J. A high-throughput cell migration assay using scratch wound healing, a comparison of image-based readout methods. BMC Biotechnol. 4, 21 (2004).
Kam, Y., Guess, C., Estrada, L., Weidow, B. & Quaranta, V. A novel circular invasion assay mimics in vivo invasive behavior of cancer cell lines and distinguishes single-cell motility in vitro. BMC Cancer 8, 198 (2008).
Acosta, S., Canclini, L., Galarraga, C., Justet, C. & Alem, D. Lab-made 3D printed stoppers as high-throughput cell migration screening tool. SLAS Technol. 27, 39–43 (2022).
Cappiello, F., Casciaro, B. & Mangoni, M. L. A novel in vitro wound healing assay to evaluate cell migration. J. Vis. Exp. 17, 56825 (2018).
Boyden, S. The chemotactic effect of mixtures of antibody and antigen on polymorphonuclear leucocytes. J. Exp. Med. 115, 453–466 (1962).
Albini, A. et al. A rapid in vitro assay for quantitating the invasive potential of tumor cells. Cancer Res. 47, 3239–3245 (1987).
Oner, A. & Kobold, S. Transwell migration assay to interrogate human CAR-T cell chemotaxis. STAR Protoc. 3, 101708 (2022).
Knieke, K. et al. CD152 (CTLA-4) determines CD4 T cell migration in vitro and in vivo. PLoS ONE 4, e5702 (2009).
Albini, A. & Benelli, R. The chemoinvasion assay: a method to assess tumor and endothelial cell invasion and its modulation. Nat. Protoc. 2, 504–511 (2007).
Pfeifer, C. R. et al. Constricted migration increases DNA damage and independently represses cell cycle. Mol. Biol. Cell 29, 1948–1962 (2018).
Xia, Y. et al. Rescue of DNA damage after constricted migration reveals a mechano-regulated threshold for cell cycle. J. Cell Biol. 218, 2545–2563 (2019).
Jacobson, E. C. et al. Migration through a small pore disrupts inactive chromatin organization in neutrophil-like cells. BMC Biol. 16, 142 (2018).
Zhang, F. et al. Astrocyte elevated gene-1 interacts with β-catenin and increases migration and invasion of colorectal carcinoma. Mol. Carcinog. 52, 603–610 (2013).
Tang, C. L. et al. Focal adhesion kinase signaling is necessary for the cyclosporin A-enhanced migration and invasion of human trophoblast cells. Placenta 33, 704–711 (2012).
Gillespie, J. L., Anyah, A., Taylor, J. M., Marlin, J. W. & Taylor, T. A. A versatile method for immunofluorescent staining of cells cultured on permeable membrane inserts. Med. Sci. Monit. Basic Res. 22, 91–94 (2016).
Kreiseder, B. et al. α-Catulin downregulates E-cadherin and promotes melanoma progression and invasion. Int. J. Cancer 132, 521–530 (2013).
Wu, P. -H., Giri, A., Sun, S. X. & Wirtz, D. Three-dimensional cell migration does not follow a random walk. Proc. Natl Acad. Sci. USA 111, 3949–3954 (2014).
Wu, P. -H., Giri, A. & Wirtz, D. Statistical analysis of cell migration in 3D using the anisotropic persistent random walk model. Nat. Protoc. 10, 517–527 (2015).
Gómez-de-Mariscal, E. et al. Use of the p-values as a size-dependent function to address practical differences when analyzing large datasets. Sci. Rep. 11, 20942 (2021).
Hilsenbeck, O. et al. Software tools for single-cell tracking and quantification of cellular and molecular properties. Nat. Biotechnol. 34, 703–706 (2016).
Jaqaman, K. et al. Robust single-particle tracking in live-cell time-lapse sequences. Nat. Methods 5, 695–702 (2008).
Wu, P. -H., Gilkes, D. M. & Wirtz, D. The biophysics of 3D cell migration. Annu. Rev. Biophys. 47, 549–567 (2016).
Konstantopoulos, K., Wu, P. H. & Wirtz, D. Dimensional control of cancer cell migration. Biophys. J. 104, 279–280 (2013).
Zaman, M. H. et al. Migration of tumor cells in 3D matrices is governed by matrix stiffness along with cell-matrix adhesion and proteolysis. Proc. Natl Acad. Sci. USA 103, 10889–10894 (2006).
Fraley, S. I., Feng, Y., Giri, A., Longmore, G. D. & Wirtz, D. Dimensional and temporal controls of three-dimensional cell migration by zyxin and binding partners. Nat. Commun. 3, 719 (2012).
Phillip, J. M. et al. Fractional re-distribution among cell motility states during ageing. Commun. Biol. 4, 81 (2021).
Guilbert, M. et al. Highlighting the impact of aging on type I collagen: label-free investigation using confocal reflectance microscopy and diffuse reflectance spectroscopy in 3D matrix model. Oncotarget 7, 8546–8555 (2016).
Maska, M. et al. Quantification of the 3D collagen network geometry in confocal reflection microscopy. In 2015 IEEE International Conference on Image Processing (ICIP) 1791–1794 (IEEE, Quebec City, 2015).
Steinwachs, J. et al. Three-dimensional force microscopy of cells in biopolymer networks. Nat. Methods 13, 171–176 (2016).
Chen, B. -C. et al. Lattice light-sheet microscopy: imaging molecules to embryos at high spatiotemporal resolution. Science 346, 1257998 (2014).
Fritz-Laylin, L. K. et al. Actin-based protrusions of migrating neutrophils are intrinsically lamellar and facilitate direction changes. eLife 6, e26990 (2017).
Zhao, Z. et al. Organoids. Nat. Rev. Methods Primers 2, 94 (2022).
Drost, J. & Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 18, 407–418 (2018).
LeSavage, B. L., Suhar, R. A., Broguiere, N., Lutolf, M. P. & Heilshorn, S. C. Next-generation cancer organoids. Nat. Mater. 21, 143–159 (2022).
Costa, E. C. et al. 3D tumor spheroids: an overview on the tools and techniques used for their analysis. Biotechnol. Adv. 34, 1427–1441 (2016).
Hofer, M. & Lutolf, M. P. Engineering organoids. Nat. Rev. Mater. 6, 402–420 (2021).
Verduin, M., Hoeben, A., De Ruysscher, D. & Vooijs, M. Patient-derived cancer organoids as predictors of treatment response. Front. Oncol. 11, 641980 (2021).
Veninga, V. & Voest, E. E. Tumor organoids: opportunities and challenges to guide precision medicine. Cancer Cell 39, 1190–1201 (2021).
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013).
Broutier, L. et al. Culture and establishment of self-renewing human and mouse adult liver and pancreas 3D organoids and their genetic manipulation. Nat. Protoc. 11, 1724–1743 (2016).
Ootani, A. et al. Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat. Med. 15, 701–706 (2009).
Gjorevski, N. et al. Designer matrices for intestinal stem cell and organoid culture. Nature 539, 560–564 (2016).
Hernandez-Gordillo, V. et al. Fully synthetic matrices for in vitro culture of primary human intestinal enteroids and endometrial organoids. Biomaterials 254, 120125 (2020).
Zhang, S., Cheng, G., Zhu, S., Lin, D. & Wu, C. Protocol for the generation, characterization, and functional assays of organoid cultures from normal and cancer-prone human esophageal tissues. STAR Protoc. 5, 103316 (2024).
Russo, G. C. et al. E-cadherin interacts with EGFR resulting in hyper-activation of ERK in multiple models of breast cancer. Oncogene 43, 1445–1462 (2024).
Lee, M. -H. et al. Multi-compartment tumor organoids. Mater. Today 61, 104–116 (2022).
Nakamura, H. et al. Organoid culture containing cancer cells and stromal cells reveals that podoplanin-positive cancer-associated fibroblasts enhance proliferation of lung cancer cells. Lung Cancer 134, 100–107 (2019).
Tsai, S. et al. Development of primary human pancreatic cancer organoids, matched stromal and immune cells and 3D tumor microenvironment models. BMC Cancer 18, 335 (2018).
Tomasova, L., Guttenberg, Z., Hoffmann, B. & Merkel, R. Advanced 2D/3D cell migration assay for faster evaluation of chemotaxis of slow-moving cells. PLoS ONE 14, e0219708 (2019).
Grognot, M. & Taute, K. M. A multiscale 3D chemotaxis assay reveals bacterial navigation mechanisms. Commun. Biol. 4, 669 (2021).
Cockx, M. et al. Neutrophils from patients with primary ciliary dyskinesia display reduced chemotaxis to CXCR2 ligands. Front. Immunol. 8, 1126 (2017).
Kamakura, S. et al. The cell polarity protein mInsc regulates neutrophil chemotaxis via a noncanonical G protein signaling pathway. Dev. Cell 26, 292–302 (2013).
Larasati, R. A. et al. The role of butyrate on monocyte migration and inflammation response in patient with type 2 diabetes mellitus. Biomedicines 7, 74 (2019).
Chakrabarti, J. et al. Sonic hedgehog acts as a macrophage chemoattractant during gastric epithelial regeneration in response to injury. FASEB J. 33, 869.827 (2019).
Huang, Y. et al. Three-dimensional hydrogel is suitable for targeted investigation of amoeboid migration of glioma cells. Mol. Med. Rep. 17, 250–256 (2018).
Pepperell, E. E. & Watt, S. M. A novel application for a 3-dimensional timelapse assay that distinguishes chemotactic from chemokinetic responses of hematopoietic CD133+ stem/progenitor cells. Stem Cell Res. 11, 707–720 (2013).
Zengel, P. et al. μ-Slide chemotaxis: a new chamber for long-term chemotaxis studies. BMC Cell Biol. 12, 21 (2011).
Tien, J. et al. Matrix pore size governs escape of human breast cancer cells from a microtumor to an empty cavity. iScience 23, 101673 (2020).
Wolf, K. et al. Physical limits of cell migration: control by ECM space and nuclear deformation and tuning by proteolysis and traction force. J. Cell Biol. 201, 1069–1084 (2013).
Charras, G. & Sahai, E. Physical influences of the extracellular environment on cell migration. Nat. Rev. Mol. Cell Biol. 15, 813–824 (2014).
Doyle, A. D., Carvajal, N., Jin, A., Matsumoto, K. & Yamada, K. M. Local 3D matrix microenvironment regulates cell migration through spatiotemporal dynamics of contractility-dependent adhesions. Nat. Commun. 6, 8720 (2015).
Miermont, A., Lee, S. W. L., Adriani, G. & Kamm, R. D. Quantitative screening of the effects of hyper-osmotic stress on cancer cells cultured in 2- or 3-dimensional settings. Sci. Rep. 9, 13782 (2019).
Paul, C. D., Mistriotis, P. & Konstantopoulos, K. Cancer cell motility: lessons from migration in confined spaces. Nat. Rev. Cancer 17, 131–140 (2017).
Holle, A. W. et al. Cancer cells invade confined microchannels via a self-directed mesenchymal-to-amoeboid transition. Nano Lett. 19, 2280–2290 (2019).
Tong, Z. et al. Chemotaxis of cell populations through confined spaces at single-cell resolution. PLoS ONE 7, e29211 (2012).
Hung, W. C. et al. Confinement sensing and signal optimization via piezo1/PKA and myosin II pathways. Cell Rep. 15, 1430–1441 (2016).
Stroka, K. M. et al. Water permeation drives tumor cell migration in confined microenvironments. Cell 157, 611–623 (2014).
Li, Y., Yao, L., Mori, Y. & Sun, S. X. On the energy efficiency of cell migration in diverse physical environments. Proc. Natl Acad. Sci. USA 116, 23894–23900 (2019).
Yang, Z. et al. High throughput confined migration microfluidic device for drug screening. Small 19, 2207194 (2023).
Zanotelli, M. R. et al. Energetic costs regulated by cell mechanics and confinement are predictive of migration path during decision-making. Nat. Commun. 10, 4185 (2019).
Zhao, R. et al. Cell sensing and decision-making in confinement: the role of TRPM7 in a tug of war between hydraulic pressure and cross-sectional area. Sci. Adv. 5, eaaw7243 (2019).
Vedula, S. R. K. et al. Emerging modes of collective cell migration induced by geometrical constraints. Proc. Natl Acad. Sci. USA 109, 12974–12979 (2012).
Jiang, X., Bruzewicz, D. A., Wong, A. P., Piel, M. & Whitesides, G. M. Directing cell migration with asymmetric micropatterns. Proc. Natl Acad. Sci. USA 102, 975–978 (2005).
Fink, A. et al. Area and geometry dependence of cell migration in asymmetric two-state micropatterns. Biophys. J. 118, 552–564 (2020).
Bernard, A. et al. Printing patterns of proteins. Langmuir 14, 2225–2229 (1998).
Chen, C. S., Mrksich, M., Huang, S., Whitesides, G. M. & Ingber, D. E. Micropatterned surfaces for control of cell shape, position, and function. Biotechnol. Prog. 14, 356–363 (1998).
Fink, J. et al. Comparative study and improvement of current cell micro-patterning techniques. Lab Chip 7, 672–680 (2007).
Du, W., Hong, S., Scapin, G., Goulard, M. & Shah, D. I. Directed collective cell migration using three-dimensional bioprinted micropatterns on thermoresponsive surfaces for myotube formation. ACS Biomater. Sci. Eng. 5, 3935–3943 (2019).
Kim, S. E. et al. Cell migration according to shape of graphene oxide micropatterns. Micromachines 7, 186 (2016).
Mahmud, G. et al. Directing cell motions on micropatterned ratchets. Nat. Phys. 5, 606–612 (2009).
Pittet, M. J. & Weissleder, R. Intravital imaging. Cell 147, 983–991 (2011).
Flesken-Nikitin, A., Williams, R. M., Zipfel, W. R., Webb, W. W. & Nikitin, A. Y. Use of multiphoton imaging for studying cell migration in the mouse. Methods Mol. Biol. 294, 335–345 (2005).
Kedrin, D., Wyckoff, J., Sahai, E., Condeelis, J. & Segall, J. E. Imaging tumor cell movement in vivo. Curr. Protoc. Cell Biol. 19, Unit 19.17 (2007).
Supatto, W., McMahon, A., Fraser, S. E. & Stathopoulos, A. Quantitative imaging of collective cell migration during Drosophila gastrulation: multiphoton microscopy and computational analysis. Nat. Protoc. 4, 1397–1412 (2009).
Lucas, L., Gilbert, N., Ploton, D. & Bonnet, N. Visualization of volume data in confocal microscopy: comparison and improvements of volume rendering methods. J. Microsc. 181, 238–252 (1996).
Hoover, E. E. & Squier, J. A. Advances in multiphoton microscopy technology. Nat. Photonics 7, 93–101 (2013).
Sahai, E. et al. Simultaneous imaging of GFP, CFP and collagen in tumors in vivo using multiphoton microscopy. BMC Biotechnol. 5, 14 (2005).
Jain, R. et al. Visualizing murine breast and melanoma tumor microenvironment using intravital multiphoton microscopy. STAR Protoc. 2, 100722 (2021).
Miller, M. J., Wei, S. H., Cahalan, M. D. & Parker, I. Autonomous T cell trafficking examined in vivo with intravital two-photon microscopy. Proc. Natl Acad. Sci. USA 100, 2604–2609 (2003).
Benninger, R. K. P. & Piston, D. W. Two-photon excitation microscopy for the study of living cells and tissues. Curr. Protoc. Cell Biol. 4, 4.11.1–4.11.24 (2013).
Svoboda, K. & Yasuda, R. Principles of two-photon excitation microscopy and its applications to neuroscience. Neuron 50, 823–839 (2006).
Durr, N. J., Weisspfennig, C. T., Holfeld, B. A. & Ben-Yakar, A. Maximum imaging depth of two-photon autofluorescence microscopy in epithelial tissues. J. Biomed. Opt. 16, 026008 (2011).
Kasprzycka, W., Szumigraj, W., Wachulak, P. & Trafny, E. A. New approaches for low phototoxicity imaging of living cells and tissues. Bioessays 46, 2300122 (2024).
Hallaji, Z. et al. An insight into the potentials of carbon dots for in vitro live-cell imaging: recent progress, challenges, and prospects. Mikrochim. Acta 189, 190 (2022).
Soha, S. A. et al. Improved imaging and preservation of lysosome dynamics using silver nanoparticle-enhanced fluorescence. Mol. Biol. Cell 34, ar96 (2023).
Harada, T. et al. An antioxidant screen identifies ascorbic acid for prevention of light-induced mitotic prolongation in live cell imaging. Commun. Biol. 6, 1107 (2023).
Seker, U. et al. Trolox is more successful than allopurinol to reduce degenerative effects of testicular ischemia/reperfusion injury in rats. J. Pediatr. Urol. 16, 465.e461–465.e468 (2020).
Vorobjeva, N. V. & Pinegin, B. V. Effects of the antioxidants Trolox, Tiron and Tempol on neutrophil extracellular trap formation. Immunobiology 221, 208–219 (2016).
Zahra, M., Abrahamse, H. & George, B. P. Flavonoids: antioxidant powerhouses and their role in nanomedicine. Antioxidants 13, 922 (2024).
Song, J. et al. SNR enhanced high-speed two-photon microscopy using a pulse picker and time gating detection. Sci. Rep. 13, 14244 (2023).
Pérez-Vizcaíno, J. et al. Dispersion management in two-photon microscopy by using diffractive optical elements. Opt. Lett. 38, 440–442 (2013).
Guan, H. et al. Deep-learning two-photon fiberscopy for video-rate brain imaging in freely-behaving mice. Nat. Commun. 13, 1534 (2022).
He, Y. et al. Self-supervised deep-learning two-photon microscopy. Photon. Res. 11, 1–11 (2023).
Wan, Y., McDole, K. & Keller, P. J. Light-sheet microscopy and its potential for understanding developmental processes. Annu. Rev. Cell Dev. Biol. 35, 655–681 (2019).
Lu, C. -H. et al. Lightsheet localization microscopy enables fast, large-scale, and three-dimensional super-resolution imaging. Commun. Biol. 2, 177 (2019).
Kaur, P., Saunders, T. E. & Tolwinski, N. S. Coupling optogenetics and light-sheet microscopy, a method to study Wnt signaling during embryogenesis. Sci. Rep. 7, 16636 (2017).
Weber, M. & Huisken, J. Light sheet microscopy for real-time developmental biology. Curr. Opin. Genet. Dev. 21, 566–572 (2011).
Schmied, C. & Tomancak, P. Sample preparation and mounting of drosophila embryos for multiview light sheet microscopy. Methods Mol. Biol. 1478, 189–202 (2016).
Khairy, K., Lemon, W. C., Amat, F. & Keller, P. J. Light sheet-based imaging and analysis of early embryogenesis in the fruit fly. Methods Mol. Biol. 1189, 79–97 (2015).
Hillman, E. M. C., Voleti, V., Li, W. & Yu, H. Light-sheet microscopy in neuroscience. Annu Rev. Neurosci. 42, 295–313 (2019).
Acknowledgements
We acknowledge the following sources of support: U54AR081774 (to D.W.), U54CA268083 (to D.W.), R01CA300052 (to D.W.), UG3CA275681 (to P.-H.W.) and UH3CA275681 (to P.-H.W.) all from the National Cancer Institute; and R35-GM157099 (to J.M.P.) from the National Institute of General Medical Sciences and the American Federation for Aging Research Glenn Foundation Junior Faculty Award (to J.M.P.).
Author information
Authors and Affiliations
Contributions
D.W. conceived the outline. W.D., P.R.N., A.F., J.M.P., P.-H.W. and D.W. wrote the paper. W.D. produced the figures.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Methods thanks Li Yang, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Rita Strack, in collaboration with the Nature Methods team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–9 and Supplementary Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Du, W., Nair, P.R., Forjaz, A. et al. Selecting the optimal cell migration assay: fundamentals and practical guidelines. Nat Methods 23, 30–42 (2026). https://doi.org/10.1038/s41592-025-02890-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41592-025-02890-1
This article is cited by
-
Methods to analyze cell migration data: fundamentals and practical guidelines
Nature Methods (2026)