Abstract
The current investigation aims to decipher the comparative anti-feeding and insecticidal potential of the purified form of Allyl isothiocyanate (AITC), a byproduct of glucosinolate hydrolysis, and three different mustard oils (black, brown and white) against Aulacophora foveicollis (red pumpkin beetle) adults and Spodoptera litura (tobacco cutworm) larvae. The in silico investigation undertakes the interaction of AITC with two anti-oxidant enzymes found in vertebrates, as AITC is also known to be an anti-oxidant and anti-cancerous compound for humans. The study also explores the AITC and protein-protein interaction among pepsin and mustard seed proteins, cruciferin and napin, as mustard contains abundant protein content but remains bio-unavailable due to presence of similar anti-nutritional factors like glucosinolates potentially having insect deterring potential. Through in vitro bioassays, it was found that the AITC was having higher insect anti-feeding potential (ranging from 74.63 to 88.22%) than the three mustard oils (ranging from 40.18 to 78.92%) against two insect pests studied in present investigation. The brown mustard oil showed LC50 as 602.23 mg/mL and 251.99 mg/mL, black mustard showed LC50 as 677.18 and 429.82 mg/mL while, white mustard oil showed LC50 as 835.21 mg/mL and 620.31 mg/mL against A. foveicollis and S. litura, respectively. The LC50 of AITC was observed to be higher, 3990 mg/mL (A. foveicollis) and 3690 mg/mL (S. litura). The in silico analysis revealed that leucine and aspartic acid are key mediators of Glutathione-S-transferase (GST)-AITC and Sulfonyl transferase (SULT)-AITC interactions in humans, respectively. The study also showed a stronger binding between human digestive enzyme (pepsin) and seed storage proteins of mustard (napin and cruciferin). In terms of insect deterrence, brown mustard oil outperformed both black and white mustard oils due to its higher glucosinolate concentration. It also showed greater efficacy than AITC at lower doses, likely because of the presence of various fatty acids in the oil that are known to deter insect pests. In the near future, the metabolites of mustard may be explored as an economical bio-control agent thus elaborating their role in replacing harmful synthetic insecticides that are a hurdle in attaining the goal of sustainable agriculture.
Similar content being viewed by others
Introduction
Mustard is a plant with biochemicals of insect-deterring potential1. Among these biochemicals, glucosinolates are of primary importance having the biofumigation potential2. These are present within compartmentalized cells along with the enzyme myrosinase. By the enzymatic action of myrosinase on sinigrin (a type of glucosinolate), hydrolysis takes place and AITC, a type of major glucosinolate breakdown product, is formed3. Several studies suggest the utilization of AITC in deterring economically important insect pests4. The worldwide loss of crops is crucial to overcome for meeting food security and eradicating the ever-growing world’s hunger5. These crop losses capped by insect pests are followed by their diverse nature6,7. The Red Pumpkin beetle, A. foveicollis, belongs to the order Coleoptera and is specifically attributed for damaging Cucurbitaceous family plants worldwide8. More than 80% of crop damage is owed to this tiny beetle which is a serious problem as the cucurbits are consumed for their nutritive and health potential9. The Red Pumpkin beetle lays around 300–350 eggs in the soil and larvae start feeding on the leaves of cucurbits as soon as they emerge10. The tobacco cutworm, S. litura, (Lepidoptera: Noctuidae) is a polyphagous plant pest, that damages cereal crops, fruit crops as well as vegetable crops worldwide11. The larvae of cutworm hatch from egg mass and start consuming leaf until at its end, eventually making the whole plant parts disappear within a few days and the adult makes a fly to another plant, continuing the plant-eating cycle with rapid pace12. Humans can utilize mustard in a limited amount while insect are detrimental to the same biochemicals at even the lowest concentration. These biochemicals include glucosinolate and their breakdown product like AITC. Accordingly, the insect pests targeted by AITC are not limited to a single group of taxonomical hierarchy; rather the insect pests belonging to diverse Orders are deterred by AITC and its parent compound, the mustard oils4,13. Therefore, it becomes crucial to examine the differences between insect pests of diverse orders. In the current investigation (A) foveicollis (representing the largest Order Coleoptera) and S. litura (representing the second largest Order Lepidoptera) insect species were chosen so as to establish the differences in their deterring potential governed by AITC and three different mustard oils. Moreover, due to the variation in glucosinolate content of the seeds (majorly responsible for insect deterring potential) among three mustard species viz., Brassica juncea (brown), (B) nigra (black) and Sinapis alba (white)14,15,16,17 it becomes crucial to examine the diversity of insect deterring potential governed by these three mustard derived seed extracts (oil), hence chosen for the current investigation, as the oil extracted is globally utilized for the food consumption among humans in different continents of the world. Since, mustard oil containing AITC is consumed by humans but doesn’t pose a threat upon its consumption. Meanwhile, AITC even in its smallest concentrations is potently toxic to insects as is evident by the current investigation and as per previous reports18,19. The present investigation reveals lower binding affinities of Glutathione-S-transferase (GST) and Sulfonyl transferase (SULT) with AITC indicating involvement of specific amino acid residues showing how AITC present in mustard oil is perceived by two very important anti-oxidant enzymes of the human metabolic system. AITC itself is a strong anti-oxidant molecule and through molecular docking studies of the current investigation, it is revealed that AITC does not counteract the anti-oxidant potential of GST and SULT thus sustaining their enzymatic activities. The current study also describes the molecular interaction of digestive enzyme pepsin with two seed proteins found in mustard viz., napin and cruciferin. Docking scores achieved were effective raises the possibility of using mustard seed protein as food. However, the presence of glucosinolates especially AITC hinders the consumption of brown mustard seed meal as food as reported earlier1,20.
Overall, the current investigation reveals out the basis of AITC non-toxicity in humans through in silico studies while also describing the interaction of mustard seed proteins with key digestive enzyme pepsin and on the other hand reveals out the bio-efficacy of costly AITC against prominent insect pests compared to bio-efficacy governed by economic to produce three mustard oils viz., black, brown and white against the similar insect pests.
Materials and methods
The present study was conducted in G.B. Pant University of Agriculture and Technology (GBPUA&T), Pantnagar, Uttarakhand, India with work plan presented in Figure S1.
Chemicals and materials
The seeds of three mustard types were obtained from Norman E. Borlaug Crop Research Centre, (NEBCRC) GBPUA&T, Pantnagar. Black mustard (B. nigra, variety: Desi Kali Sarson), brown mustard (B. juncea, variety: Kranti) and white mustard (S. alba, variety: Desi Safed Sarson) were the three mustard types used in the study. The oil was extracted from seeds using soxhlet assembly at Lab 107 A, Metabolite Research Laboratory, Department of Biochemistry, GBPUA&T, Pantnagar. The standard of AITC was purchased from Sigma Aldrich (Product No. 36682, Purity 95%). Glassware and other required chemicals were procured from Borosil, Sigma, SRL and HiMedia.
Instrumentation
The oil extraction was done using Borosil’s SOX012 soxhlet assembly (Figure S8) using petroleum ether (60–80 °C) as a solvent (Figure S8). The Buchi Rotavapor R-300 rotary vapor evaporator was utilized to free the oil from petroleum ether. The Precisa Model XB 120 A weighing balance was used to measure insect weight for experimental analysis. The Shimadzu’s UV-1900i spectrophotometer was used to assess the content of total glucosinolates in freshly prepared seed meals.
Extraction of oils
Finely crushed and weighed seed material (10 g) was placed into a thimble and inserted into the Soxhlet extractor. A piece of cotton was also placed at the top to evenly distribute the solvent as it dripped on the sample during extraction. Petroleum ether in a volume two and a half times the capacity of the extractor was added to the flask. Oil extraction was carried out for a period of six hours (hrs). After extraction, the heating mantle was turned off and the apparatus was allowed to cool. The solvent was then evaporated using rotary vapor evaporator to remove any remaining traces and the extraction flask was air dried for 4 h. The flask was then weighed. The weight of the oil seed was noted as W(g), the weight of the flask plus glass beads as Z(g) and the total weight of the flask, beads and oil as Y(g). The weight of the oil was calculated as (Y − Z) g and the oil content (%) was calculated using the formula: (Y − Z)/W×10021.
Capturing, rearing and culturing of insects
The egg masses of S. litura collected from the field associated with the Biocontrol Laboratory were incubated under controlled laboratory conditions until hatching (Figure S2, S6 and S7) and adults of A. foveicollis were captured (Figure S4) from the Vegetable Research Center (VRC), GBPUA&T, Pantnagar (Figure S3). The newly hatched larvae were reared in sterilized plastic jars lined with clean filter paper and were fed with fresh castor leaves under laboratory-controlled conditions with temperature (25–28 °C), relative humidity (65–70%) and light: dark cycle as (14:10). Due to their vigorous feeding behavior, the leaves were replaced every 8 h to ensure continuous nourishment. The jars were cleaned regularly by removing insect waste and washing the containers thoroughly before each feeding session. Fresh filter paper and healthy castor leaves were placed in the jars after each cleaning cycle. This rearing process was maintained until the larvae reached the 4th instar stage, at which point they were selected and used for further experimentation.
Anti-feeding and insecticidal activity analysis of AITC and mustard oil seed extracts against insects
The dose groups of AITC (1000, 1500, 2000, 2500, 3000 and 3500 mg/mL) and mustard oil were used in concentrations ranging from 40, 80, 120, 160 and 200 mg/mL for anti-feeding activity analysis. The three different mustard oils viz., black, brown and white were assessed separately for bio-efficacy experiments. For, A. foveicollis the leaves of Lageneria siceraria (bottle-gourd) were cut into a section of (5 × 5) cm2 and against S. litura the leaves of Ricinus communis (castor bean) were cut into similar sections for bio-efficacy experimentation. The petri plates used were of size (100 × 50) mm each fitted with Sieve 20 Filter Paper soaked in double distilled water for moisture retaining. The cut leaf sections were rinsed for 5 min into respective concentrations of AITC and mustard oil as mentioned above. The double distilled water rinsed leaf sections of the same dimensions from both plants were used as control, viz., castor leaves were used as food for tobacco cutworm and bottle gourd leaves were used as food source for red pumpkin beetle assessment of bio-efficacy. The experimentation was done in 3 replications with 5 insects of a particular type in each petri plate and continued for 72 h After Feeding (HAF) (Figure S5)22. The data on Mean Leaf Area Consumed (MLAC) and anti-feeding activity was subjected to analysis done from SPSS software.
For insecticidal activity, the leaf sections and petri plates used were of similar dimensions. The experimentation time was noted from 6 HAF rather than 12 HAF. The 6, 12, 24, 36, 48, 72 and 96 HAF values were recorded and data were interpreted by using Probit analysis tool. The number of replications and insects was kept the same to that used in the anti-feeding activity bioassay. However, the concentrations were changed for AITC as 4000, 4500, 5000, 5500, 6000 and 6500 mg/mL and for mustard oils as 250, 350, 450, 550, 650 and 750 mg/ml. The mortality data was accounted by using Probit analysis software revealing the Lethal Dose 50 (LD50) and Lethal Time 50 (LT50) known for elucidating the insecticidal activity of the target compound23.
Estimation of phytochemicals
Total glucosinolate Estimation in seeds
The total glucosinolates were estimated spectrophotometrically in methanolic extracts by using sodium tetrachloropalladate. Absorbance was recorded at 425 nm24.
Estimation of AITC in seed meals & oil
Titration methods were used to analyze the concentration of AITC from different mustard defatted seed meals. The mustard seed meals were weighed (5 g) and vigorously mixed with 125 ml analytical grade absolute ethanol and 240 ml distilled water was then added to it. The proceeding for steam distillation the leftover 150 ml distillate was collected and 0.1 N silver nitrate (25 ml) and 10% ammonium hydroxide (10%) were added to it. Following filtration the titration was performed with standard ammonium thiocyanate solution25. The colorimetric estimation of AITC in mustard oils was determined14. This method is based on the color reaction between allyl thiourea and potassium ferricyanide solution in dilute acetic acid which measures the optical density of the color complex at 600 nm and it works in the range of 2.5 to 7.5 picogram of AITC per milliliter of the color solution.
Recognition of target protein and molecular Docking
The critical active site residues of GST and SULT were identified using UniProtKB with the UniProt IDs P0CG30 and Q06520, respectively. Both GST and SULT were derived from Homo sapiens (humans). The crystal structures corresponding to PDB IDs 4MPG (GST) and 1J99 (SULT), shown in Fig. 1, were retrieved from the Protein Data Bank (PDB) for structural analysis.
The crystal structures of GST and SULT with Uniprot ID and PDB ID.
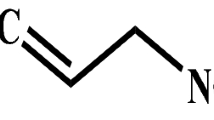
The proteins were selected based on criteria such as resolution, R-value, and structural coverage. The cruciferin and napin being seed proteins of mustard are limited in their bioavailability due to the presence of glucosinolates and other anti-nutritional factors. The conjugation reaction of GST with AITC enhances its anti-oxidant and anti-cancer effects. The physicochemical properties of these proteins were analyzed using the ProtParam tool (https://web.expasy.org/protparam)26. Molecular docking of AITC was performed to predict its interactions with GST and SULT. AITC (ZINC000001687017) and known ligands for GST and SULT, Glutathione (ZINC000003830891) and 3-Beta-0Hydroxy-5-Androsten-17-one (ZINC000003807917, Brand name, Astenile), respectively, shown in Fig. 2 were sourced from the ZINC15 database (https://zinc15.docking.org in SDF format. The SDF files were converted to PDB format using Open Babel 2.4.127. The protein structures were prepared in UCSF Chimera, where errors such as non-essential heteroatoms and water molecules were removed28. The prepared protein and ligand structures were imported into PyRx for docking simulations. Ligands were converted to PDBQT format and subjected to energy minimization using the default Gasteiger method in PyRx via Open babel. A docking grid was defined around the active sites of the proteins via http://www.scfbio-iitd.res.in/dock/ActiveSite.jsp29 and molecular docking was performed using PyRx30. Conformers of AITC were evaluated based on their binding affinities and acceptable RMSD values (≤ 2 Å). The chemical structure of AITC and Known Binders (Glutathione and Astenile) for GST and SULT are shown in Fig. 3.
The 2-D representations of protein-ligand interactions of GST, SULT with AITC and KBs via LigPlot.
Chemical structure of AITC and Known Binders (Glutathione and Astenile) for GST and SULT.
Protein- protein molecular Docking
The software High-accuracy Docking (HDOCK) was employed to perform molecular docking to identify potential binding sites between pepsin, the napin and cruciferin proteins, aiming to determine the most favorable protein–protein complex pose. Pepsin, napin and cruciferin proteins were identified using UniProtKB with the UniProt IDs P0DJD7, P84529 andQ7XB53, respectively31. The crystal structures corresponding to PDB IDs 1QRP (Fig. 4), 2SS1 and 3KGLwere retrieved from the Protein Data Bank (PDB) for structural analysis. The refined crystal structure of human pepsin provides valuable structural data for understanding the PPI interface. Protein 1QRP has a molecular weight of 35.39 kDa, contains 2,625 atoms, and comprises a single unique chain with 326 residues. The crystal structure was refined to a resolution of 1.96 Å, yielding an R factor of 20.0%. This structural insight is crucial in mapping the interaction sites and contributes to understanding the conformational adaptability and binding mechanisms across related proteinase families (https://www.rcsb.org/structure/1QRP). HDOCK was utilized to predict the best docking pose for the modeled complex based on a structural template32. The predicted model of napin and cruciferin, acting as a ligand, was docked with Pepsin, the receptor protein. The 3D structure of pepsin showing its active site is shown in Fig. 4.
The 3D Structure of Pepsin showing its active site.
Statistical analysis
The Statistical Package for Social Sciences (SPSS) and Probit software were used to analyze the anti-feeding and insecticidal activity, respectively. The experimental values of replicates were analyzed by ANOVA at p < 0.05.
Results
Total glucosinolates and AITC determination
The current presented a comparative analysis of three mustard varieties and their key insect deterring bioactive compounds viz., total glucosinolates and AITC. Brown mustard exhibited the highest concentration of total glucosinolates in seed meals at 92.67 ± 0.54 µmol/g, which significantly exceeded both black mustard (62.78 ± 0.74 µmol/g) and white mustard (52.35 ± 0.62 µmol/g) (Table 1). This represented 48% higher glucosinolate content in brown mustard compared to black mustard, and a 77% higher content compared to white mustard. The total glucosinolates in seed meals were measured using UV-Vis Spectrophotometer, AITC in seed meals was assessed through titration measures and AITC in mustard oils was assessed through colorimetric methods. AITC content in seed meals followed a similar pattern, with brown mustard containing the highest concentration at 0.88 ± 0.012%, followed by black mustard at 0.69 ± 0.016%, and white mustard at 0.56 ± 0.023%. This trend continued in the oil extracts, where brown mustard oil contained 0.57 ± 0.08% AITC, black mustard oil contained 0.42 ± 0.02%, and white mustard oil contained 0.31 ± 0.05%. The AITC concentration was consistently lower in oils compared to seed meals across all three varieties, with reductions that ranged from 35% to 45%.
Anti-feeding bioassay of AITC and mustard oils
When comparing the effectiveness of these mustard-based compounds against insect pests (Table 2), AITC emerges as the clear superior option, demonstrating 88.22% and 86.30% anti-feeding activity against A. foveicollis and S. litura respectively at 3500 mg/mL, which is significantly higher than any mustard oil tested here. Among the three mustard oils evaluated at identical concentrations, brown mustard oil consistently outperformed the others, achieving 78.92% and 73.41% anti-feeding activity at 200 mg/mL compared to black mustard oil’s 70.15% and 68.07%, and white mustard oil’s 68.14% and 63.12% at the same concentration against A. foveicollis and S. litura, respectively. The performance gap widens further at lower concentrations, where brown mustard oil maintains relatively strong effectiveness (78.37% and 74.63% at 1000 mg/mL equivalent comparison against A. foveicollis and S. litura, respectively), while white mustard oil drops to its lowest performance levels of 44.12% and 40.18% at 40 mg/mL against A. foveicollis and S. litura, respectively. Notably, A. foveicollis appears slightly more susceptible to these compounds than S. litura across most treatments, with consistently higher anti-feeding percentages and lower mean feeding rates. The dose-response relationship is most pronounced with AITC, showing a dramatic drop from 88.22% (3500 mg/mL) to 78.37% (1000 mg/mL) against A. foveicollis, while the mustard oils show more gradual declines, suggesting different mechanisms of action or potency thresholds between these natural compounds.
The anti-feeding activity of A. foveicollis in AITC (Table S1) ranged from 78.37 at 1000 mg/mL to 88.22% at 3500 mg/mL concentration. The anti-feeding activity of S. litura in AITC (Table S2) ranged from 74.63% at 1000 mg/mL to 86.30% at 3500 mg/mL concentration. The anti-feeding activity of A. foveicollis in Brown Mustard Oil (Table S3) ranged from 61.04% at 40 mg/mL to 78.92% at 200 mg/mL concentration. The anti-feeding activity of S. litura in Brown Mustard Oil (Table S4) ranged from 50.29% at 40 mg/mL to 73.41% at 200 mg/mL concentration. The anti-feeding activity of A. foveicollis in Black Mustard Oil (Table S5) ranged from 49.03% at 40 mg/mL to 70.15% at 200 mg/mL concentration. The anti-feeding activity of S. litura in Black Mustard Oil (Table S6) ranged from 44.23% at 40 mg/mL to 68.07% at 200 mg/mL concentration. The anti-feeding activity of A. foveicollis in White Mustard Oil (Table S7) ranged from 44.12% at 40 mg/mL to 68.14% at 200 mg/mL concentration. The anti-feeding activity of S. litura in White Mustard Oil (Table S8) ranged from 40.18% at 40 mg/mL to 63.12% at 200 mg/mL concentration.
Insecticidal bioassay of AITC and mustard oils
The comparative toxicity of oil extracts of Black, Brown and White Mustard against A. foveicollis and S. litura was analyzed (Table 3). The AITC was observed to be more toxic to S. litura with 3.55 of LT50 than LT50 of 8.07 in the case of A. foveicollis (Table 4). The study evaluated mortality rates at two different concentrations (250 mg/mL and 750 mg/mL) and determined the lethal concentrations (LC50) and lethal times (LT50) for each treatment. Brown mustard oil demonstrated the highest toxicity with mortality rates of 40.00% at 250 mg/mL and 60.00% at 750 mg/mL, achieving an LC50 of 602.23 mg/mL and LT50 of 16.71 h against A. foveicollis. The chi-square value was 0.952 with a regression equation of y = 0.773x − 2.849. Black mustard oil showed moderate effectiveness with 20.00% mortality at 250 mg/mL and 60.00% at 750 mg/mL, resulting in an LC50 of 677.18 mg/mL and LT50 of 24.40 h (chi-square: 0.817, regression: y = 1.447x + 0.902). White mustard oil exhibited the lowest toxicity against this species, with mortality rates of 26.67% and 53.33% at the respective concentrations, leading to an LC50 of 835.21 mg/mL and LT50 of 31.83 h (chi-square: 0.954, regression: y = 1.393x + 0.929). The toxicity pattern differed significantly against S. litura. Brown mustard oil maintained strong effectiveness with mortality rates of 53.30% and 73.30% at 250 mg/mL and 750 mg/mL respectively, achieving the lowest LC50 of 251.99 mg/mL and remarkably quick LT50 of 4.03 h (chi-square: 0.975, regression: y = 1.069x − 2.431). Black mustard oil showed improved performance against this species compared to A. foveicollis, with 40.00% and 66.67% mortality rates, resulting in an LC50 of 429.82 mg/mL and LT50 of 11.50 h (chi-square: 0.936, regression: y = 1.550x + 0.916). White mustard oil demonstrated enhanced toxicity against S. litura with mortality rates of 26.67% and 60.00%, achieving an LC50 of 620.31 mg/mL and LT50 of 14.93 h (chi-square: 0.989, regression: y = 1.667x + 0.342). The data revealed that brown mustard oil consistently exhibited the highest toxicity against both insect species, with S. litura being significantly more susceptible than A. foveicollis. The chi-square values ranged from 0.817 to 0.989, indicating good model fit for the dose-response relationships across all treatments.
Molecular docking
Molecular docking simulations are valuable tools for predicting the orientation and binding affinity of ligands within a protein’s active site. The grid box was defined to encompass the active site residues of the GST and SULT protein, ensuring accurate ligand-target interactions. The grid box dimensions of GST and SULT were set at 12.028 Å (X-axis), 63.028 Å (Y-axis), 1.419 Å (Z-axis) and 23.914Å (X-axis), −51.025Å (Y-axis), 42.208Å (Z-axis) respectively, centering the box round the binding pocket. These dimensions were optimized to fully cover the active site, facilitating a precise analysis of the binding interactions between AITC with GST and SULT (Table 5). The grid box parameters were chosen based on the spatial distribution of the key binding residues, ensuring comprehensive docking coverage are shown in Fig. 5. The 2-D representations of protein-ligand interactions of GST, SULT with AITC and KBs via LigPlot are shown in Fig. 2.
The HDOCK results for the protein - protein docking between napin and pepsin provided the docking score and confidence score of −247.90 and 0.8763 indicating a strong binding affinity between napin and pepsin (Table 6). Lower (more negative) docking scores typically signify a more favorable interaction, suggesting that napin forms a stable complex with pepsin. Confidence Score reflects a high level of reliability in the predicted model. The ligand RMSD value of 92.59 Å is unusually high, which may indicate significant conformational changes or flexibility in the ligand, or it might suggest potential challenges in predicting an accurate binding mode. Generally, RMSD values below 2 Å indicate well-aligned structures, so this deviation points to variability in the docked poses. The LG score of 5.378 indicates a confident prediction for the global quality of the docking pose. LG scores above 4.0 typically suggest that the model is reliable. A MaxSub value of 0.197 is relatively low, suggesting moderate alignment between the predicted docked structure and the reference structure, which could imply some level of structural deviation or flexibility in the interaction. Overall, the docking results suggest a strong binding affinity between napin and pepsin, with a confident prediction of the binding poses. The complex stability model of protein-protein interaction via HDOCK is shown in Fig. 6.
The complex stability model of protein-protein interaction via HDOCK.
The current investigation also opens gateways of futuristic research aimed at elucidating the molecular mechanism of AITC and other glucosinolate breakdown products in insect metabolic systems. The biochemical behavior of AITC is studied in silico in humans and in vitro in insects in addition to three mustard oils. The involvement of leucine and aspartic acid in AITC-GST and AITC-SULT interactions will be helpful in elucidating the AITC interaction with different proteins. In near future, the defined concentration levels of AITC along with three distinct mustard oils shall prove beneficial in controlling the insect pests while attaining the goal of sustainable agriculture. The studies of insect pest control with AITC and mustard oil against wide range of insect pests are in the need of hour to overcome excessive synthetic pesticide usage.
Discussion
Natural pesticides are safe for the environment and are thus the best alternative in comparison to industry-generated synthetic chemicals used as pesticides which otherwise deteriorate soil health and are found toxic to humans and other animals as well33. Previous research studies have enlisted chemical compounds and identified properties in AITC and naturally derived mustard oils that are held responsible for insect-deterring potential4,16,18,34. Investigations into the pest control potential of AITC have revealed promising results across multiple economically important insect species. Studies targeting the melon fly, Zeugodacus cucurbitae (Diptera: Tephritidae), showed that incorporating AITC into artificial diets at doses between 5 and 200 mg/mL resulted in increased mortality rates among first through third instar larvae. Additionally, developmental timelines were significantly extended for larval, pupal, and overall maturation phases when compared to control groups treated with dimethyl sulfoxide34. The compound’s vapor-phase activity has also been established through experiments with grain storage pests, specifically the rice weevil Sitophilus oryzae and maize weevil S. zeamais (Coleoptera: Curculionidae). Exposure to AITC concentrations ranging from 0.25 to 1.25 µg/mL resulted in suppression of critical enzymatic functions, including catalase, glutathione-S-transferase, acetylcholine esterase, and cytochrome C oxidase activities35. Additional research involving the cigarette beetle Lasioderma serricorne (Coleoptera: Ptinidae) and confused flour beetle Tribolium confusum (Coleoptera: Tenebrionidae) revealed AITC’s ability to impair reproductive success. Exposure to AITC at concentrations approaching the LC50 threshold of 4.50 µL/L induced developmental abnormalities across multiple life stages in confused flour beetles36,37. Developmental stage sensitivity to AITC varies between species, as demonstrated in studies with the fungus gnat Bradysia odoriphaga (Diptera: Sciaridae), where mature insects showed higher susceptibility than immature forms. Field trials using application rates of 4,000–6,000 ml per 667 m² resulted in population suppression ranging from 45 to 71% compared to untreated areas38. Building on knowledge that mustard-derived glucosinolate metabolites exhibit insect-deterrent properties25, researchers examined larvicidal activity of seed meals from various mustard cultivars (B. juncea and S. alba) against mosquito populations. Comparative toxicity assessments revealed that synthetic benzyl isothiocyanate achieved the highest larval mortality after 24 h exposure (LC50 = 5.29 mg/mL), outperforming both AITC (LC50 = 19.35 mg/mL) and 4-hydroxybenzyl isothiocyanate (LC50 = 55.41 mg/mL)39.
In our current research, AITC displayed insecticidal properties with LT50 values of 8.07 h and 3.55 h against A. foveicollis and S. litura, respectively.
The anti-feeding and larvicidal activity of S. litura and Culex quinquefasciatus was assessed against bioactive compounds isolated from entomopathogenic fungi Penicillium spp. The ethyl acetate extract of Penicillium spp., showed larval mortality analyzed after 48 h exposure with LC50: 72.205 mg/ml: LC90: 282.783 mg/ml and LC50: 94.701 mg/ml: LC90:475.049 mg/ml against both these insect pests40. Bioassay analysis of S. litura larvae showed a mortality rate of 33.33 to 46.7% against Broccoli cultivar extracts41. However, in the current study, the mortality rates varied from 53.30 to 73.30% in S. litura against brown mustard oil, indicating superior efficacy compared to previously tested plant extracts, though these controlled laboratory conditions may not reflect the complex field environment where factors such as UV degradation, rainfall, and temperature fluctuations could significantly reduce effectiveness.
The first of its kind assessment done in the current research analysis of insecticidal analysis against A. foveicollis (in response to three different mustard oils) showed variation from 40% to 60% at 250 and 750 mg/mL, respectively. In other research finding, the 50% of anti-feeding activity was reported against A. foveicollis in response to Azadirachta indica leaf extracts42. Anti-feeding activity of 81.1 and 93.7% for A. foveicollis were reported at 3200 and 6400 µg/ml concentrations against leaf extracts containing triterpenoids in Mordica charantia43. The analysis of insect pest anti-feeding activity from other plant extracts in other studies, as compared to mustard seed extracts has shown a significant variation44. In the current study, a range of 63.12% (white mustard oil against S. litura) to 73.41% (brown mustard oil against S. litura) anti-feeding activity was observed, though these laboratory results may not translate directly to field conditions where insects have alternative food sources and behavioral adaptations. The anti-feeding activity in response to brown mustard oil was noted highest as 78.92% against A. foveicollis and 73.41% against S. litura. However, AITC showed highest anti-feeding activity as 88.22% (against A. foveicollis) and 86.30% (against S. litura) (Table 2) and highest mortality rates of 80.00% (against A. foveicollis) and 86.67% (against S. litura) at 6500 mg/mL in comparison to brown mustard oil’s 60.00% (against A. foveicollis) and 73.40% (against S. litura) at 750 mg/mL (Tables 3 and 4). When compared to synthetic insecticides, these mortality rates are notably lower than conventional organophosphates or pyrethroids, which typically achieve > 90% mortality at much lower concentrations, and field studies with neem-based formulations have shown that laboratory efficacy often drops by 30–50% under field conditions due to environmental degradation and pest behavioral adaptations.
Earlier studies exploring the fumigation effects of mustard oil against the maize weevil S. zeamais identified eggs as the most vulnerable developmental stage, attributed to the presence of AITC. Mortality rates in eggs achieved LT50/LT95 time intervals of 16.72/77.77 h when exposed to 1.25 µL/L concentrations and 16.52/60.86 h at 1.87 µL/L doses. Pupal development phases showed intermediate susceptibility with corresponding LT50/LT95 values of 16.39/38.70 h and 15.58/32.92 h at the respective concentrations. Larval instars proved most susceptible, displaying LT50/LT95 intervals of 6.19/29.27 h at 1.25 µL/L and 2.64/23.96 h at 1.87 µL/L45. In our research, brown mustard oil exhibited feeding deterrent activity of 61.04–78.92% when tested against A. foveicollis and 50.29–73.41% against S. litura. This same oil variant also showed lethal activity with median survival times (LT50) of 16.71 h for A. foveicollis and 4.03 h for S. litura. Black mustard oil achieved LT50 values of 24.40 h and 11.50 h for A. foveicollis and S. litura, respectively, while white mustard oil demonstrated the weakest performance with LT50 values reaching 31.83 h for A. foveicollis and 14.93 h for S. litura. Studies conducted with S. litura larvae using broccoli variety extracts from the Brassicaceae family achieved mortality percentages between 33.33% and 46.70%. These findings highlighted broccoli extracts’ substantial pest control capabilities and reinforced the necessity for ongoing investigations to characterize and purify active compounds for practical pest management strategies41. In contrast, our experimental work with three mustard oil types revealed that brown mustard seed extracts produced the highest lethality rates. Mortality in S. litura larvae varied from 53.30% to 73.30%, while adult A. foveicollis mortality ranged between 40.00% and 60.00%. Separate research evaluated white mustard oil’s toxicity through feeding trials against the diamond black moth Cydia pomonella (Lepidoptera: Tortricidae), pine processionary moth Dendrolimus pini (Lepidoptera: Lasiocampidae), and beet armyworm Spodoptera exigua (Lepidoptera: Noctuidae). Calculated LC50 concentrations were 0.422 mg/ml for C. pomonella, 11.74 mg/ml for D. pini, and 11.66 mg/ml for S. exigua, indicating maximum potency against C. pomonella46.
In our present study, brown mustard oil showed LC50 values of 602.23 mg/mL against A. foveicollis and 251.99 mg/mL against S. litura, respectively. The black mustard oil showed LC50 values of 677.18 mg/mL against A. foveicollis and 429.82 mg/mL against S. litura, respectively. The white mustard oil showed LC50 values of 835.21 mg/mL against A. foveicollis and 620.31 mg/mL against S. litura, respectively.
Further investigations examined water-based mustard seed extracts for their feeding inhibition capabilities against the rose aphid Macrosiphum roseiformis (Hemiptera: Asphididae), western flower thrips Scirtothrips dorsalis (Thysanoptera: Thripidae), and two-spotted spider mite Tetranychus urticae (Trombidiformes: Tetranychidae). Both preference and no-preference bioassays were conducted using rose leaves either treated with extracts or left untreated. Results confirmed feeding suppression activity against all target pest species47. Chemical analysis using Gas Chromatography-Mass Spectrometry (GC-MS) has identified the main bioactive components in mustard oils, specifically fatty acids and glucosinolates, as the primary compounds responsible for their pest-repelling characteristics13,48.
Our present study revealed that even the most effective mustard oil variant (brown mustard oil) necessitated substantially higher application rates compared to pure AITC to achieve comparable insecticidal results. When tested against A. foveicollis, brown mustard oil achieved 40.00% lethality at 250 mg/mL concentration and 60% at 750 mg/mL, while AITC produced 46.67% mortality at 4000 mg/mL and reached 80.00% at 6500 mg/mL. Mustard oil applications resulted in 53.30% mortality at 250 mg/mL and 73.30% at 750 mg/mL concentrations, while AITC achieved 53.33% mortality at 4000 mg/mL and 86.67% at 6500 mg/mL for S. litura trials. Pure AITC exhibited accelerated toxic action, demonstrating reduced median lethal times (LT50) of 8.07 h against A. foveicollis and 3.55 h against S. litura compared to brown mustard oil, which recorded LT50 values of 16.71 h and 4.03 h for the same respective species.
The GST and SULT play crucial roles in detoxification and oxidative stress regulation processes that are considered vital for cellular defense across species. GSTs are multifunctional enzymes that catalyze the conjugation of glutathione (GSH) to electrophilic compounds, including xenobiotics and reactive oxygen species (ROS), converting these into less toxic metabolites for excretion49. This detoxification mechanism protects cellular components like Deoxyribose Nucleic Acid (DNA), proteins, and lipids from oxidative damage, which can otherwise lead to mutations and disease. Additionally, GSTs contribute to oxidative stress management by conjugating and eliminating reactive aldehydes such as 4-hydroxynonenal (4HNE), a lipid peroxidation byproduct associated with apoptosis and neurodegenerative disorders. SULTs detoxify compounds by transferring sulfonate groups to drugs, hormones and toxins, increasing their solubility for excretion and supporting the body’s defense against toxicity50,51. The functional conservation of both GSTs and SULTs across species highlights their evolutionary significance in cellular detoxification and stress regulation, which are essential for maintaining homeostasis and preventing cellular damage52. To elucidate the role of GST and SULT against AITC, the protein (GST and SULT) and ligand (AITC) interactions were studied in the current investigation and it was found that leucine and aspartic acid are the key amino acids in mediating GST-AITC and SULT-AITC interaction, respectively. This information in near future will prove helpful in undermining the more deep mechanisms of AITC interaction within the metabolic system of humans involving different proteins with these two common amino acid residues. Though this molecular docking analysis represents theoretical binding interactions that may not accurately predict in vivo detoxification efficiency the alternative explanations for the observed insecticidal activity could include membrane disruption, interference with neurotransmitter function, or inhibition of essential metabolic enzymes beyond GST and SULT pathways.
The selection of pepsin, napin and procruciferin for Protein-Protein Interaction (PPI) analysis is based on their roles in digestion and nutrient absorption, which are crucial for understanding their efficacy. Pepsin is a vital digestive enzyme that catalyzes the breakdown of proteins into smaller peptides, facilitating their absorption in the intestine53. Napin and cruciferin are key seed storage proteins found in oilseeds like rapeseed (B. napus). Napin, a 2 S albumin with a molecular weight of about 14 kDa, exhibits high solubility across various pH levels, particularly in acidic conditions and contains multiple disulfide bonds that enhance its stability. Its rich composition of basic residues, such as arginine and lysine, makes napin valuable for emulsification and foaming in food applications21. Cruciferin, classified as an 11 S globulin with a molecular weight ranging from 20 to 40 kDa, forms hexameric structures characterized by a predominantly β-sheet secondary structure, providing essential amino acids during seed germination. However, cruciferin’s solubility is limited at low pH, which can affect its functionality in food systems54. Pepsin is essential for digestion, while napin and cruciferin are critical seed storage proteins with unique properties that enhance their utility in food applications55. The PPI analysis of pepsin with napin and procruciferin provides insights into the digestive interactions in pests exposed to mustard oils, helping to clarify how these oils and their bioactive components might disrupt pest nutrition and physiology. The AITC acts as fumigant and is known to inhibit electron transport chain thereby generating reactive oxygen species, resulting ultimately in the death of insects. However, defined dose of AITC makes insects inactive yet al.ive, by which insects develop anti-feeding behavior25,56.
Conclusion
The present investigation outlines coherently the different concentration levels of AITC and three mustard oils required to attain insect pest resistance against two diverse insect species belonging to different taxonomical Orders and their respective Families, reflecting an effective range of metabolites responsible over the Class Insecta. This opens a gateway of deeper analysis of research required in the near future aiming to tackle the heavy crop economic losses caused to farmers by global pests, A. foveicollis and S. litura and also discusses how glucosinolate breakdown products in the human metabolic system interact with the key antioxidant enzymes, GST and SULT. Advanced bioinformatics tools shall reveal the clearer basis of mustard metabolite interactions in humans and other animals. However, both GST and SULT are also present in insects but they cannot not detoxify glucosinolate breakdown products and thus lead to anti-feeding behavior or, the death of insects. Moreover, certain fatty acids present in mustard oils also account for insect pest resistance. The low-level concentration of glucosinolate breakdown products leads to anti-feeding behavior while higher concentrations accompanied by fatty acids lead to the death of the insects. However, the individual involvement of AITC although in higher concentration, reflects higher insect deterring potential than the three mustard oils studied here distinctly and the comparable insect toxicity effects are generated. This also reflects the advantage of utilizing mustard oils against insect pests as they are easy and economical to produce as compared to AITC which is a highly costly and purified metabolic product synthesized industrially, though practical application faces significant formulation challenges including volatility, oxidation susceptibility, and the need for sophisticated stabilization technologies that could substantially increase production costs. Several study limitations warrant acknowledgment, including controlled laboratory conditions that may not reflect field environments, short-term exposure effects without addressing potential resistance development, and lack of evaluation of effects on beneficial insects and soil microorganisms. Future research should prioritize field trials to validate laboratory findings, investigate formulation strategies to enhance stability and efficacy, and conduct comprehensive economic analysis comparing mustard oil-based biopesticides with existing synthetic pest management options to accurately assess their true potential as sustainable pest management tools. Specifically, the generation of data on AITC homology modeling with human ingestion of mustard proteins is crucial (not done here) for deeper analysis of the largely unknown biochemical interactions, and also field trials to assess the efficacy of AITC and mustard oils against global insect pests are urgently needed.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Lietzow, J. Biologically active compounds in mustard seeds: a toxicological perspective. Foods. 10(9), 2089 (2021).
Nguyen, V. P. T., Stewart, J., Lopez, M., Ioannou, I. & Allais, F. Glucosinolates: natural Occurrence, Biosynthesis, Accessibility, Isolation, Structures, and biological activities. Molecules 25 (19), 4537 (2020).
Barba, F. J. et al. Bioavailability of glucosinolates and their breakdown products: impact of processing. Front. Nutr. 3, 24 (2016).
Du, Y., Grodowitz, M. J. & Chen, J. Insecticidal and Enzyme Inhibitory Activities of Isothiocyanates against Red Imported Fire Ants, Solenopsis invicta. Biomolecules, 10(5), 716 (2020).
Cerda, R. et al. Primary and secondary yield losses caused by pests and diseases: assessment and modeling in coffee. PLoS One. 12 (1), e0169133 (2017).
Cates, R. G. Host plant predictability and the feeding patterns of oligophagous and insect herbivores. Oecologia 48 (3), 319–326 (1981).
Hardy, N. B. & Otto, S. P. Specialization and generalization in the diversification of phytophagous insects: tests of the musical chairs and Oscillation hypotheses. Proc. Royal Soc. B: Biol. Sci. 281 (1795), 20132960 (2014).
Shukla, G. S. & Singh, J. P. Studies on the rate of excretion of Aulacophora foviecollis Lucas., the red pumpkin beetle (Coleoptera: Chrysomelidae). Experientia, 26(3), 272 (1970).
Rolnik, A. & Olas, B. Vegetables from the cucurbitaceae family and their products: positive effect on human health. Nutrition 78, 110788 (2020).
Khan, M. M. H., Alam, M. Z., Rahman, M. M., Miah, M. I. H. & Hossain, M. M. Influence of weather factors on the incidence and distribution of pumpkin beetle infesting cucurbits. Bangladesh J. Agricultural Res. 37 (2), 361–367 (2012).
Vengateswari, G., Arunthirumeni, M. & Shivakumar, M. S. Effect of food plants on Spodoptera Litura (Lepidoptera: Noctuidae) larvae immune and antioxidant properties in response to Bacillus Thuringiensis infection. Toxicol. Rep. 7, 1428–1437 (2020).
Shekhawat, S. S., ShafiqAnsari, M. & Basri, M. Effect of host plants on life table parameters of Spodoptera Litura. Indian J. Pure Appl. Biosci. 6 (1), 324–332 (2018).
Garg, S., Nain, P., Joshi, R., Punetha, H. & Srivastava, R. M. Bio-efficacy of mustard seed extracts against Bihar hairy caterpillar Spilosoma obliqua (Erebidae: Lepidoptera) & assessment of mustard allelo-chemicals in response to mustard aphid Lipaphis erysimi (Aphididae: Hemiptera) infestation. South. Afr. J. Bot. 171, 156–163 (2024).
Mukhopadhyay, S. & Bhattacharyya, D. K. Colorimetric Estimation of allyl isothiocyanate content in mustard and rapeseed oils. Fette Seifen Anstrichm. 85 (8), 309–311 (1983).
Garg, S., Pant, U., Nain, P. & Punetha, H. Nutritional & Anti-Nutritional and Anti-Oxidative profiling of globally utilized diverse seed coat color mustards. Bioscience Forum – Int. J. 15 (4), 1261–1267 (2023).
Garg, S., Punetha, H., Chaudhary, D. & Srivastava, R. M. Comparative bio efficacy of allyl isothiocyanate and brown mustard oil against polyphagous insect pests Aulacophora foveicollis (Coleoptera: Chrysomelidae) and Spodoptera Litura (Lepidoptera: Noctuidae). International J. Trop. Insect Science (2025).
Das, G. et al. Glucosinolates and Omega-3 fatty acids from mustard seeds: phytochemistry and Pharmacology. Plants (Basel). 11 (17), 2290 (2022).
Cerón, D. A. C., de Alencar, E. R., Faroni, L. R. D., Silva, M. V. A. & Salvador, D. V. Toxicity of allyl isothiocyanate applied in systems with or without recirculation for controlling Sitophilus zeamais, Rhyzopertha dominica, and Tribolium castaneum in corn grains. J. Sci. Food. Agric. 103 (13), 6373–6382 (2023).
Gou, Y. P. et al. Responses of fungi maggot (Bradysia impatiens Johannsen) to allyl isothiocyanate and high CO2. Front. Physiol. 13, 879401 (2022).
Garg, S., Gairola, K., Punetha, H. & Gangola, S. An exploration of the biochemistry of mustard seed meals: A phytochemical and in Silico perspective. Foods 13 (24), 4130 (2024).
Ntone, E. et al. Napins and cruciferins in rapeseed protein extracts have complementary roles in structuring emulsion-filled gels. Food Hydrocoll. 125, 1–11 (2022).
Isman, B., Koul, O., Lucyzynski, A. & Kaminski, J. Insecticidal and antifeedant bioactivities of Neem oils and their relationship to Azadirachtin content. J. Agric. Food Chem. 38 (6), 1407–1411 (1997).
Duraipandiyan, V., Ignacimuthu, S. & Gabriel Paulraj, M. Antifeedant and larvicidal activities of Rhein isolated from the flowers of Cassia fistula L. Saudi J. Biol. Sci. 18 (2), 129–133 (2011).
Mawlong, I., Kumar, M. S. S., Gurung, B., Singh, K. H. & Singh, D. A simple spectrophotometric method for estimating total glucosinolates in mustard de-oiled cake. Int. J. Food Prop. 20 (S3), 3274–3281 (2017).
Tsao, R., Peterson, C. J. & Coats, J. R. Glucosinolate breakdown products as insect fumigants and their effect on carbon dioxide emission of insects. BMC Ecol. 2 (1), 1–7 (2002).
Garg, V. K. et al. K. MFPPI-multi FASTA ProtParam interface. Bioinformation 12 (2), 74–77 (2016).
Yoshikawa, N., Hutchison, G. R. & Fast Efficient fragment-based coordinate generation for open babel. J. Cheminform. 11 (1), 1–9 (2019).
Guterres, H. et al. CHARMM-GUI high‐throughput simulator for efficient evaluation of protein-ligand interactions with different force fields. Protein Sci. 31 (11), e4413 (2022).
Chatterjee, A., Roy, U. K. & Halder, D. Protein active site structure prediction strategy and algorithm. Int. J. Curr. Eng. Technol. 7 (3), 1092–1096 (2017).
Li, H. et al. Multi-ligand molecular docking, simulation, free energy calculations and wavelet analysis of the synergistic effects between natural compounds Baicalein and cubebin for the Inhibition of the main protease of SARS-CoV-2. J. Mol. Liq. 374, 121253 (2023).
Rahman, M. et al. In silico, molecular Docking and in vitro antimicrobial activity of the major rapeseed seed storage proteins. Front. Pharmacol. 11, 1340 (2020).
Yan, Y., Tao, H., He, J. & Huang, S. Y. The HDOCK server for integrated protein-protein Docking. Nat. Protoc. 15 (6), 1829–1852 (2020).
Souto, A. L. et al. Plant-Derived pesticides as an alternative to pest management and sustainable agricultural production: Prospects, applications and challenges. Molecules 26 (16), 4835 (2021).
Singh, S., Diksha, E. & Mahajan, E. Appraisal of growth inhibitory, biochemical and genotoxic effects of allyl isothiocyanate on different developmental stages of Zeugodacus cucurbitae (Coquillett) (Diptera: Tephritidae). Sci. Rep. 12 (1), 10363 (2022).
Wu, H., Liu, X., Yu, D., Zhang, X. & Feng, J. Effect of allyl isothiocyanate on ultra-structure and the activities of four enzymes in adult Sitophilus Zeamais. Pestic. Biochem. Physiol. 109, 12–17 (2014).
Worfel, R. C., Schneider, K. S. & Yang, T. C. Suppressive effect of allyl isothiocyanate on populations of stored grain insect pests. J. Food Process. Preserv. 21 (1), 9–19 (1997).
Santos, J. C., Faroni, L. R. A., Sousa, A. H. & Guedes, R. N. C. Fumigant toxicity of allyl isothiocyanate to populations of the red flour beetle Tribolium castaneum. J. Stored Prod. Res. 47 (4), 238–243 (2011).
Shi, C. H. et al. Control of bradysia Odoriphaga (Diptera: Sciaridae) with allyl isothiocyanate under field and greenhouse conditions. J. Econ. Entomol. 110 (3), 1127–1132 (2017).
Flor-Weiler, L. B. et al. Bioactivity of brassica seed meals and its compounds as ecofriendly larvicides against mosquitoes. Sci. Rep. 13 (1), 3936 (2023).
Arunthirumeni, M., Vinitha, G. & Shivakumar, M. S. Antifeeding and larvicidal activity of bioactive compounds isolated from entomopathogenic fungi Penicillium sp. for the control of agricultural and medically important insect pest (Spodoptera Litura and Culex quinquefasciatus). Parasitol. Int. 92, 102688 (2023).
Gupta, S., Chaudhary, A., Singh, S., Arora, S. & Sohal, S. K. Broccoli (Brassica Oleracea L. var. italica) cultivars, Palam Samridhi and Palam Vichitra affect the growth of Spodoptera Litura (Fabricius) (Lepidoptera: Noctuidae). Heliyon 7 (8), e07612 (2021).
Gujar, G. T. & Mehrotra, K. N. Biological activity of Neem against the red pumpkin beetle, Aulacophora foveicollis. Phytoparasitica 16 (4), 293–302 (1988).
Chandravadana, M. V. Identification of triterpenoid feeding deterrent of red pumpkin beetles (Aulacophora foveicollis) from Momordica Charantia. J. Chem. Ecol. 13 (7), 1689–1694 (1987).
Jan, Q. et al. Comparative conventional preventive strategies for insect pest of Okra. Saudi J. Biol. Sci. 29 (5), 3114–3121 (2022).
Pae, J. L., Faroni, L. R. D. A., Dhingra, O. D., Cecon, P. R. & Silva, T. A. Insecticidal fumigant action of mustard essential oil against Sitophilus Zeamais in maize grains. Crop Prot. 34, 56–58 (2012).
Konecka, E. et al. Insecticidal activity of Brassica alba mustard oil against lepidopteran pests Cydia pomonella (Lepidoptera: Tortricidae), Dendrolimus pini (Lepidoptera: Lasiocampidae), and Spodoptera exigua (Lepidoptera: Noctuidae). J. Plant Prot. Res. 58(2), 206–209 (2018).
Gupta, G., Kaur, G., Yadav, R. & Kumar, N. R. Repellent effects of aqueous extracts of mustard seeds Brassica juncea, on three major pests of horticultural crops. CJAAS 1 (1), 16–24 (2021).
House, H. L. & Graham, A. R. Capric acid blended into food stuff for control of an insect pest, Tribolium confusum (Coleoptera:tenebrionidae). Can. Entomol. 99 (9), 994–999 (1967).
Dasari, S., Ganjayi, M. S., Oruganti, L., Balaji, H. & Meriga, B. Glutathione S-transferases detoxify endogenous and exogenous toxic agents-minireview. J. Dairy. Veterinary Anim. Res. 5 (4), 1–3 (2017).
Bauer-Marinovic, M., Taugner, F., Florian, S. & Glatt, H. Toxicity studies with 5-hydroxymethylfurfural and its metabolite 5-sulphooxymethylfurfural in wild-type mice and Transgenic mice expressing human sulphotransferases 1A1 and 1A2. Arch. Toxicol. 86 (5), 701–711 (2012).
Pedersen, L. C., Yi, M., Pedersen, L. G. & Kaminski, A. M. From steroid and drug metabolism to glycobiology, using sulfotransferase structures to understand and tailor function. Drug Metab. Dispos. 50 (8), 1027–1041 (2022).
Sandamalika, W. G., Priyathilaka, T. T., Lee, S., Yang, H. & Lee, J. Immune and xenobiotic responses of glutathione S-Transferase theta (GST-θ) from marine invertebrate disk abalone (Haliotis discus discus): with molecular characterization and functional analysis. Fish Shellfish Immunol. 91, 159–171 (2019).
Bhutta, Z. A., Sadiq, K. & Aga, T. Protein digestion and bioavailability. Encyclopedia Hum. Nutr. 4, 116–122 (2013).
Perera, S. P., McIntosh, T. C. & Wanasundara, J. P. Structural properties of cruciferin and napin of Brassica napus (canola) show distinct responses to changes in pH and temperature. Plants, 5(3), 1–24 (2016).
Sarker, A. K., Saha, D., Begum, H., Zaman, A. & Rahman, M. M. Comparison of cake compositions, Pepsin digestibility and amino acids concentration of proteins isolated from black mustard and yellow mustard cakes. AMB Express. 5 (1), 22–26 (2015).
Haouel Hamdi, S., Hedjal-Chebheb, M., Kellouche, A., Khouja, M. L. & Boudabous, A. Mediouni Ben Jemaa, J. Management of three pests’ population strains from Tunisia and Algeria using Eucalyptus essential oils. Industrial Crops Prod. 74, 551–556 (2015).
Funding
No funding received.
Author information
Authors and Affiliations
Contributions
SG: Conceptualization, Methodology, Writing – original draft, Writing – review & editing. HP: Supervision, Writing – review & editing. SaG: Supervision, Writing – review & editing AV.: Validation, Writing – review & editing. MT: Writing – review & editing. MJ: Supervision, Writing – review & editing. AM: Supervision, Writing – review & editing. FM: MT: Writing – review & editing, Supervision. AMA: Supervision, Writing – review & editing. MBBH: Funding acquisition, Validation, Supervision. NA: Funding acquisition, Writing – review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Garg, S., Punetha, H., Gangola, S. et al. Mustard derived compounds as insecticides and modulators of human metabolism. Sci Rep 16, 5783 (2026). https://doi.org/10.1038/s41598-026-35536-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-35536-z