Abstract
Antimicrobial resistance has become a serious global issue, because these microbes produce beta-Lactamase that degrades antibiotics. To overcome this issue, worldwide researchers have focused on nanoparticles. It is because of their unique chemical and biological properties and their use as an antimicrobial and larvicidal agent to get rid of infectious and vector-borne diseases. Therefore, this study aimed to synthesize zinc oxide nanoparticles using Acorn fruit as a novel and phytochemical-rich plant and evaluated their antimicrobial and larvicide potentials. These ZnO NPs synthesis were confirmed by Visual observation of color change from yellowish to dark brown and UV visible analysis. Fourier transform infrared spectroscopy (FTIR) of prepared nanoparticles revealed a sharp peak at 600–450 cm− 1 indicating the presence of Zn-O stretching vibration. The prominent, sharp peaks in the X-ray diffraction pattern verified the crystalline structure of ZnO NPs. With scanning electron microscopy (SEM), ZnO nanoparticles exhibiting a tiny size indicated good surface morphology. In biological activities these ZnO NPs revealed outstanding antibacterial efficacy against E. coli (minimum zone of inhibition 15 ± 02 mm at 10 µg/ml and maximum 24 ± 05 mm at 80 µg/ml) and S.aureus (minimum zone of inhibition 17 ± 03 mm at 10 µg/ml and 26 ± 02 mm at 80 µg/ml). In the in-silico analysis, these nanoparticles demonstrated a considerable interaction with the Beta-Lactamase binding site residues with a docking score of -8.2 kcal/mol. It confirms the successful inhibition of the given enzyme, which catalyzes a lot of antibiotics. Thus, these nanoparticles combated microbes by blocking their resistive enzyme, which is the novel property of these ZnO NPs. Furthermore, these ZnO NPs showed high mortality rates against the Culex quinquefasciatus larvae and the LC 50 and LC 90 values were found as 13.6 ppm and 45 ppm respectively. In histopathological observation, they found that these nanoparticles caused microvilli destruction. It also initiated mid-gut columnar cell vacuolization, passage of epithelial cell contents into the mid-gut lumen and ultimately cell death. Hence, the above findings propose that the synthesized ZnO NPs may be used for various environmental and anti-pathogenic applications.
Similar content being viewed by others
Introduction
Antimicrobial resistance has become a serious global issue. New therapeutic agents are required to treat bacterial infections instead of antibiotics1. The better antibacterial potential of green synthesized nanoparticles derived from plant sources. It indicates that these nanoparticles also possess the additional feature of self-functionalization of molecular medicines2. Previous research has demonstrated that the surface area of a nanoparticle increases with decrease in size.it results in a higher level of antibacterial activity3. This is because small particles have the greater ability to directly interact with microbial pathogen cell walls4. Among them, ZnO nanoparticles are notable for their antibacterial properties because of their high surface area, which effectively dissolves and enters into bacterial cells4. The antibacterial effect of ZnO NPs was mostly due to the nanoparticles rather than the liberation of free Zn2 + ions5. Due to strong antibacterial properties, low toxicity to humans and durability in adverse conditions, zinc oxide nanoparticles are among the top studied polymeric metallic oxide nanoparticles6. Numerous investigations have proven the strong antibacterial property of ZnO NPs for various kinds of microorganisms and their potential to inhibit the formation of biofilms7. The ZnO NPs that have been synthesized in previous studies and tested against Salmonella typhimurium and Staphylococcus aureus demonstrated strong antibacterial activity8. The biological applications and identification of plant-mediated ZnO NPS have been the primary objectives of recent research9. Meanwhile, mosquito vectors can spread a number of pathogens, including arboviruses, protozoans and filariae, which result in infectious diseases of significant public health issues10. Mosquito-borne diseases, on the other hand, cause serious human diseases in practically every region of the world11. Among these, Culex quinquefasciatus may be a vector for Wuchereria bancrofti, lymphatic filariasis and Flavivirus.they might potentially spread bacterial diseases, rarely12, . The World Health Organization (WHO) states that the dissemination of innovative control techniques is crucial to lowering mosquito populations and the disease impact13. Synthetic pesticides are harmful to human beings and the ecosystem14. The advantage of ZnO NPs made from other wild plants against culex quinquefasciatus has been explored in the past, but no research has been done specifically on the use of ZnO NPs produced from acorn15. Acorns are regarded as an ecologically and commercially significant genus in the deciduous and evergreen forest ecosystems of the Northern Hemisphere16. They are members of the Fagaceae family. Acorns in Pakistan’s Himalayan and Hindu Kush regions have generated broad-leaved evergreen woods17. In Pakistan, the acorn species have not gotten as much attention as other economically significant plant species. They are mostly associated with traditional medicine and the identification of phytonutrients to assess their toxicological effects and bioactivities18. Numerous and varied health benefits of acorn trees have been recognized, and their various parts are utilized medicinally19. However, no nanoparticles have been synthesized from acorn. Considering this perspective, we attempted to synthesize ZnO nanoparticles from the acorn fruit aqueous extract and examine their antimicrobial and antilarvicidal potential against Culex quinquefasciatus. Therefore, these Zinc Oxide nanoparticles have been evaluated by an agar well diffusion method against E. coli and against S. aureus, in silico analysis of beta Lactamase and different larval instars of Culex quinquefasciatus. No previous research has been done on green synthesis of Zinc Oxide nanoparticles using Acorn Fruit. To the best of our knowledge, this is the first report to successfully synthesize ZnO nanoparticles using Acorn Fruit and evaluates their in vitro and in silico activities.
Method and materials
Sample collection
The fresh fruits of Acorn were collected during the month of November 2024 from Drosh District Chitral, Khyber Pakhtunkhwa, Pakistan. It was in compliance with institutional, national, international guidelines and legislation. Collection permission was granted by the Forest and Wildlife Department, Government of Khyber Pakhtunkhwa (Permit No. FD/CHR/AC-2024/11). The species was identified by the plant’s taxonomist Prof. Jawad Ali, Department of Botany, Government Degree College Shabqadar, Charsadda, and a voucher specimen (Voucher No. GDC-SH-BOT.43.11.24) has been deposited in the college herbarium for reference. These acorn fruits were rinsed carefully with double-distilled water to eliminate the soil and other residues. The acorn fruits were dried under shade conditions and ground in a free-air circulated grinding mill. Finally, the ground material was stored in airtight jars for further processing.
Collection and identification of larvae
The fourth instar, Culex quinquefasciatus larvae were collected from a drainage, septic tank and contaminated water from Mazara, Shabqadar. Muhammad Umar, HOD Zoology at Govt. College Shabqadar, identified them as Culex quinquefasciatus. The general protocol of the World Health Organization was followed for experiment. At 25–27 °C temperature and 75–85% humidity, the larvae were kept with water and enough food in a sterilized flask.
Extract preparation
Aqueous extract of acorn fruit was prepared with the modified method of Umar et al., 202320. The acorn fruits were dried under shade conditions and ground in a free-air circulated grinding mill. 25gm of powder was put in 100 ml distilled water at 60 °C for 15 min. The same was filtered using Whatman filter paper and stored at 4 °C for further use.
Synthesis of nanoparticles
Acorn extract was added to a 0.25 M solution of Zinc Chloride in the 9:1 ratio and the mixture was heated for 24 h to enable nanoparticles synthesis. For two hours at 65–70 °C, the resultant mixture was swirled vigorously in a magnetic stirrer. Change in the color of the solution from yellowish to dark brown confirmed the occurrence of ZnO nanoparticles. The resulting liquid was kept at room temperature and in a dark chamber to prevent the Zinc Chloride from auto-oxidizing. The solution centrifuged at 10,000 rpm for 30 min using a high-speed centrifuge. After that, the supernatants were eliminated. The pellets were washed three times through double-distilled water. It passed through another centrifugation step to eliminate any traces of boundless plant compounds. The same pellets were collected in a Petri dish, dried at ambient temperature and kept for further process. A brief ZnO NPs Synthesis and Characterizations process has shown in the Fig. 1.
Shows the ZnO NPs Synthesis and Characterizations Real lab Diagram.
Characterization of synthesized ZnO NPs
Nanoparticles behavior, bio-dispersal and efficacy are all affected by their physicochemical characteristics. Thus, it is crucial to characterize ZnO NPs in order to assess the functional characteristics of the produced particles. Characterization is performed using a variety of analytical techniques such as UV-vis spectroscopy, X-ray diffractometry (XRD), Fourier transform infrared spectroscopy (FT-IR), Zeta potential and scanning electron microscopy (SEM).
UV–Vis spectrum analysis
UV-vis absorption spectra were recorded using a PerkinElmer (Waltham, MA, USA) Lambda 950 UV-Vis spectrophotometer. The ZnO NPs are dispersed in water at a concentration of 0.1 wt% and then the solution is used to perform the UV–Vis measurement. Triplicates of the sample were used and their absorption was measured at wavelengths between 200 and 800 nm with a 2-nm difference at 1-hour interval and recorded.
Fourier transform infrared spectroscopy (FT-IR)
The dried powder of the synthesized zinc oxide nanoparticles was used in an FTIR spectrometer (vector 22, Bruker, Germany). It is to analyze the spectra properties of the particles using Fourier transform infrared spectroscopy (FTIR). At ambient temperature, the pellets are scanned at a resolution of 400–4000 cm− 1.
X-ray diffraction (XRD)
The dried Zinc Oxide Nanoparticles were subjected to X-ray diffraction (XRD) examinations with CuKo radiation and an XPert PRO diffractometer (Holland) at a detector voltage of 40 kV and a current of 30 mA. With a scanning rate of 6°/min, the reported range of 2\(\:\theta\:\) was 10–80°. The Debye-Scherrer formula was used to estimate the particle sizes.
Where D is the average crystallite domain size perpendicular to the reflecting planes, λ is the x-ray wavelength, \(\:{\upbeta\:}\) is the full width at half maximum (FWHM), and \(\:\theta\:\) is the diffraction angle.
Zeta potential
Zeta potential, a crucial characteristic for colloids or nanoparticles in suspension.There is no reliable method for determining the surface charge of tiny particles in liquid. The standard procedure is to locate a particle’s electric potential anywhere in the diffuse layer, away from the particle surface. The sliding or shear plane is the term used to describe this area in relation to particle movement in liquid is Zeta potential.
Scanning electron microscopy (SEM)
The TM-1000 Scanning Electron Microscope (SEM) from Hitachi, Japan, was used to quantify and analyze the structural morphology of zinc oxide nanoparticles. The SEM grid’s film was dried by putting it under a mercury lamp for five minutes after an aliquot of each sample was fixed on a carbon-coated copper grid. To confirm the existence of nanoparticles, the device was outfitted with an energy dispersive spectrum.
Antibacterial activities
Gram-positive bacteria Staphylococcus aureus and gram-negative bacteria E. coli were subjected to the antibacterial activity of bio-synthesized ZnO NPs using the well diffusion method21. The pathogenic microbes were cultured for 12 h at 37 °C in a different nutritional broth. Following an overnight growth period in a petri dish, test pathogenic cultures were swabbed on Muller-Hinton agar (MHA) plates. The wells were then filled with 10, 20, 40, and 80 µg/ml of ZnO NPs, Acorn crude extract, ZnCl2 and amoxillin to compare their antimicrobial potentials. The plates were incubated for a full day at 37 °C and the inhibitory zones were determined and compared.
Molecular docking analysis
Beta-Lactamase 3D structure (PDB ID: 1Z4G) was obtained from the Protein Databank Database (PDB). After target crystal structure retrieval, ligands removed, water molecules eliminated and saved as PDB format using Discovery Studio Visualizer 2016 (DSV 2016). Synthesized ZnO NP (nanoparticle) structure coordinates were prepared in Chem Sketch and converted into PDB format. Both target and protein were docked using PyRx. Finally, DSV 2016 was employed for the ligand-protein complex interaction analysis22.
Larvicidal activity procedure
To make a stock solution, the dried nanoparticles were re-suspended in deionized water. The toxicity of different doses of biosynthesized ZnO NPs (5, 15, 30, 45, and 60 ppm) was examined against different larval instars of culex quinquefasciatus. Briefly, 20 larvae were put into a beaker of 200 mL dechlorinated water and covered with net. Screening was done on all larval instars for each ZnO NPs concentration. A control group, free of ZnO NPs, was conducted alongside each experiment23. Only deionized water was used to treat the control groups. The mortality rates (percentage) after 24 h were determined following the formula below.
% Larval mortality = No. of dead larvae / No. of treated larvae×100.
Histopathological slides preparation
The fourth-instar larvae of Cx. quinquefasciatus (both treated and untreated) were separated and preserved in Bouin’s solution for 48 h. The material was implanted and sliced with glass knives in a rotating microtome following dehydration in a succession of graded ethanol. Haematoxylin-eosin staining, analysis and photomicroscope photography were performed on the Sect24. The midgut was fixed in a solution comprising 2.5% glutaraldehyde and 4% paraformaldehyde in a 0.1 M phosphate buffer (pH 7.3) for microscopic examinations after 24 and 48 h. After that, it was dehydrated in a graded acetone solution, post-fixed in a 1% osmium tetroxide solution in the same buffer and embedded. Ultra-thin slices were stained with uranyl acetate and lead citrate before examination.
Statistical analysis
The ZnO NPs antibacterial activity against the investigated bacterial strains and larvicide activity against Culex quinquefasqiatus statistically analyzed using Graph Pad Prism version 7.04 for Windows in order to identify any significant differences.
Results and analysis
UV-Visible spectrophotometry
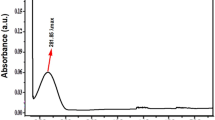
The UV-Vis spectrophotometer was used to analyze the optical properties of the generated ZnO NPs solution. Figure 2 shows a clear absorption peak at 270 nm in the spectra of the pure Zinc Oxide nanoparticles. This indicated that the nanoparticles had formed in the solution. This behavior agrees with previous results25. The results clearly showed that zinc ions were successfully reduced by the acorn fruit extract.
Shows UV-visible absorption peak (270 nm) of Acorn fruit mediated ZnO NPs.
FT-IR analysis
The Fourier transform infrared spectra of the ZnO NPs were analyzed. In order to determine which functional groups are connected to these reductive biomolecules. It is also to find out which ones helped reduce ZnO into nanoparticles. The behavior is found in line with previous research26. The results clearly showed that the acorn fruit extract successfully reduced zinc ions. In (Fig. 3), the bond at 3400 cm− 1 OH stretching, indicating the presence of phenol and flavonoids as capping agents27. The peaks at 2920 cm− 1 and 1540–1630 cm− 1 correspond to C-H stretching, indicating organic moieties on the Nanoparticles and N–H bonds of primary and secondary amides and –C–N-stretching vibration of amides or –C–O– stretching of alcohols, ether, carboxylic acids and anhydrides28. The peaks at 1380 cm− 1 and 1020 to 1100 cm− 1 indicate the presence of C-H/C-N groups and C-O stretching respectively. The sharp absorption peak at 600–450 cm− 1 indicates the presence of Zn-O stretching vibration. Flavonoids, more complex polyphenolic chemicals, including proanthocyanidins and simple phenolics and their derivatives are all present in acorn fruit extract19. These substances might be in charge of reducing metal or metal oxide ions and creating nanoparticles29.
Shows FT-IR spectrum of Acorn fruit mediated ZnO NPs.
X-ray diffraction (XRD) analysis
X-ray diffraction analysis (Fig. 4) shown the 2θ distinctive peaks of ZnO NPs at 31.70°, 47.5°, 54°, 56.60°, 68.8° and 75.20° for (112), (126), (200), (202), (004) and (002) planes of the crystal lattice, correspondingly. The minor peak at 28.5° may be occur due to organic moieties from the green synthesis process. These peaks suggest the presence of the hexagonal Wurtzite type of Zinc Oxide Nanoparticles and were consistent with JCPDS Card No. 36-1451. Strong and narrow diffraction peaks indicate the ZnO NPs ideal crystalline structure. Moreover, the size of particles was found 29.6 nm using Debye-Scherrer formula30.
Shows the X-ray diffraction peaks of Acorn fruit mediated ZnO NPs.
Zeta potential
The potential stability of the particles in the colloidal suspension was measured by zeta potential as shown in Fig. 5. The majority of ZnO nanoparticles have a negative charge (-25.9 mV). The results indicate that the ZnO NPs synthesized from the aqueous extract of Acorn fuit do not combine at all, which had a negative charge and were stable at room temperature.
Shows the Zeta potential graph of Acorn fruit ZnO NPs.
SEM analysis
In the SEM examination, the external morphology of the bio-synthesized zinc oxide nanoparticles was primarily described; the subsequent image is shown in Fig. 6. The majority of ZnO NPs have been found to be nanometer-sized. Interestingly, the majority of ZnO NPs have the same dimensions and there are not enough immense particles. Furthermore, the ZnO NPs exhibit a minor agglomeration, which is characteristic of green synthesis nanoparticles. This occurs because of the biosynthetic ZnO NPs larger surface area and their long-lasting affinity for one another, which leads to agglomeration or aggregation31. It can be said that ZnO NPs stability and agglomeration are significantly influenced by ecological factors. Consequently, during the nanoparticle synthesis process, the ZnO NPs adhere to one another and spontaneously form asymmetrical clusters32.
Reveals scaning electron microscopy photograph of Zinc oxide nanoparticles.
Antibacterial activity
The well diffusion method was used to evaluate the antibiogram potentials of ZnO NPs at different concentrations (10, 20, 40 and 80 µg ml− 1) against the pathogenic strains. The zones of inhibitions were measured in millimeters. In Fig. 7a, b and c a graphical description of the antibiogram assessment is given. ZnO NPs show a high level of antibacterial activity against S. aureus (Table 1) and E. coli (Table 2). In comparison, the standard drug Amoxicillin (in the same concentrations as ZnO NPs) shown low antibacterial efficacy against S. aureus and E. coli. In this study, it is observed that as ZnO NPs concentrations increased, the zones of inhibitions would also increase (Fig. 7d and e).
(a) Represents the zones of inhibition of ZnO NPs and Amoxicillin against E. coli, (b) Represents the zones of inhibition of ZnO NPs and Amoxicillin against S. aureus, (c) Represents the zones of inhibition of Acorn Extract and ZnCl2 against E.coli and S. aureus, (d) Shows the comparative antibacterial potential of ZnCl2, Crude Extract, Amoxicillin and ZnO NPs against E. coli, (e) Shows the comparative antibacterial potential of ZnCl2, Crude Extract, Amoxicillin and ZnO NPs S. aureus.
Molecular Docking analysis
Docking studies of ZnO NPs revealed a variety of probable interactions with the binding site of the substrate of the receptor protein (Fig. 8a); the binding site residues entangled in the interactions include ARG275, ARG244, SER235, LYS234, ALA237, GLY238, ASN170, GLU104, GLU166, ASN132, SER70, SER130 and LYS73. The docking score and interaction details are enlisted in Table 3. The synthesized nanoparticle demonstrated a considerable interaction with the beta-Lactamase binding site residues having a docking score of -8.2 kcal/mol. Furthermore, amoxicillin was also docked to the active site of targets ASN170, ASN132, SER70, SER235, VAL216, ALA237 and ARG2449 (Fig. 8,b, c and d). The binding affinity of amoxicillin with beta lactamase is (-6 kcal/mol) in comparison to the nanoparticle (-8.2 kcal/mol), which is in agreement with the experimental findings.
(a) Crystal structure of beta lactamase (PDB ID: 1ZG4), (b) Shows docking results of ZnO NPs (Zinc oxide nanoparticles) into active site of beta lactamase target. Best scoring posse (A) with target (binding affinity -8.2 kcal/mol), (B) solvent accessible surface (SAS) and interactions (C), (c) Show docking results of standard drug (amoxicillin) into active site of beta lactamase target. Best scoring posse (A) with target (binding affinity -6 kcal/mol) and (B) solvent accessible surface (SAS), (d) Show molecular interactions of standard drug (amoxicillin) with beta lactamase target (C) and 2D diagram (D).
Larvicidal bioassay
Concentrations of 5, 15, 30, 45 and 60 ppm were used to check the larvicidal properties of biosynthesized ZnO NPs against Culex larvae. The results showed the percent mortality as 20 ± 1.23%, 55 ± 2.01%, 70 ± 1.67%, 90 ± 3.10% and 100% respectively. According to data analysis, the larvicidal potential of ZnO NPs depended on both time and dose. Furthermore, for the IV instar, the LC50 (the concentration of ZnO NPs that results in 50% death) and LC90 (the concentration of ZnO NPs that results in 90% mortality) were found 13.6 ppm and 45 ppm, respectively (Fig 9).
Reveals the affects of ZnO NPs on % mortality of Culex quinquefasciatus .
Histological studies
The normal control group showed the stomach caecum anatomy normal. The layer of epithelial cells in the untreated larvae of Cx. quinquefasqiatus stomach caecum was well-preserved. The center of the cell contains the ovoid nuclei (Fig. 10A). At ZnO NPs dose 5ppm, the caecum and stomach showed the first symptoms of intoxication. The histopathology of Cx. quinquefasciatus larvae revealed significant damage to the epithelial columnar cells as well as morphological changes at this level (Fig. 10B). At a ZnO NPs dose of 15ppm, there is a noticeable onset of vacuolization at the apical level and some cells appear mildly hypertrophied. The cells were infiltrated by these vacuoles. We occasionally observed an expansion of the intercellular gaps (Fig. 10C). We observed a cytoplasmic rejection of cell material that was associated with the food column at a ZnO NPs dose of 30 ppm, and the gastric caeca epithelial cells began to rupture (Fig. 10D). The treated larvae with a ZnO NPs dose of 45 ppm, a consistent microvilli border is seen in the gastric lumen. Some cells displayed because of lysis and degraded (Fig. 10E). The majority of the epithelial cells vacuolated and degraded at a ZnO NPs dose of 60 ppm of treatment. Based on the concentration, histopathological results show that ZnO NPs have various harmful effects on the two distinct areas of the midgut epithelium. Large vacuoles of various sizes with ruptured membranes at the apical side of the epithelial cells and widespread cellular microvillar disruption were the main cytopathological changes (Fig. 10F).
The effects of ZnO nanoparticles the mid gut of Culex Quinquefasciatus. (A) Normal control group; (B) group treated with ZnO NPs dose 5ppm; (C) Treated group with ZnO NPs dose 15 ppm ; (D) Treated group with ZnO NPs dose 30 ppm; (E) Treated group with ZnO NPs dose 45 ppm; (F) Treated group with ZnO NPs dose 60 ppm.
Discussion
The success of nanotechnology has changed the productivity and scope of research because of its uses in many fields33. Nanoparticles have been synthesized via various methods, such as chemical, biological and physical ones. As compared to other synthesis processes, biological synthesis is less hazardous, less expensive. It is safer and easier to utilize, which makes it more important34. For many years, metal oxide nanoparticles have been used extensively for medicinal applications. Several nanoparticles, including Fe3O4, TiO2, CuO and ZnO have been investigated through various biological activities35. Particularly among these, ZnO NPs have shown good potential use in medicine for drug delivery as well as larvicidal, antibacterial, antioxidant and anticancer purposes. In daily life, bacterial strains’ adhesion and reproductive activity result in severe illnesses in both humans and animals36. The development of microbial resistance to commercial antibiotics in the medicinal zone is a significant issue. Consequently, a new medication must be developed in order to solve the issue. Nanotechnology uses medicinal plants as essential sources for eradicating microorganisms37. Conyza canadensis was used to synthesize ZnO NPs and the bioflavonoid rutin molecule showed strong antibacterial potential against human bacterial pathogens in a previous study38. Similarly, mosquitoes pose a threat to millions of people and animals around the world due to their role as vectors of germs that cause a wide range of terrible diseases39. Dengue and Japanese encephalitis are two of the most common diseases that they spread. Since there are now no viable vaccines or medications for mosquito-borne diseases40. Chemical pesticides such as carbamates, organophosphates, diflubenzuron and organochlorines have been used as an efficient way to control insects41. However, these pesticides are sloppy, dangerous and frequent use causes mosquitoes to become resistant. Since mosquito resistance poses a serious risk to human beings and the ecosystem, it is imperative to develop target-specific, biodegradable, environmentally benign and economically viable anti-mosquito medications42. In the current decade, nanotechnology opens up new possibilities for a variety of uses in the sectors of agriculture, electronics and medicine43. Plant-mediated nanoparticles are more valuable than chemical and physical approaches. Researchers have been focusing on zinc oxide nanoparticles lately because of their many uses9. Nanoparticles made from plants demonstrated remarkably larvicidal effects44. The acorn fruits have been used from ancient times to treat many diseases. Recent research found that acorn fruits contain tannins, polyphenol, saponins, flavonoids and terpinoids that exhibit strong antimicrobial activities and reducing in nature. Due to their synergistic effects, Acorn Fruit-based ZnO nanoparticles exhibit unique and novel antibacterial and larvicide activities. Therefore, in the current study, Acorn Fruit mediated ZnO nanoparticles were synthesized and characterized through various techniques like UV, FTIR, XRD, zeta potential and SEM. Furthermore, their activities were assessed through well diffusion method on the pathogenic bacteria and larval instars of Culex quinquefasciatus. In the results of the ZnO nanoparticles, the UV spectrum showed that the highest absorbance values were at 270 nm, indicating that the appropriate nanoparticles had formed in the solution (Fig. 2). The observed absorption of ZnO NPs agrees with previous studies45. The results clearly showed that zinc ions were successfully reduced by the acorn fruit extract. In the FTIR graph (Fig. 3), the bond at 3400 cm− 1 OH stretching, indicating the presence of phenol and flavonoids as capping agents27. The peaks at 2920 cm− 1 and 1540–1630 cm− 1 correspond to C-H stretching.it indicates organic moieties on Nanoparticles and N–H bonds of primary and secondary amides. The aforementioned ranges represent –C–N-stretching vibration of amides or –C–O– stretching of alcohols, ether, carboxylic acids and anhydrides28. The bands at 1380 cm− 1 and 1020 to1100 cm− 1 indicate the presence of C-H/C-N groups and C-O stretching, respectively. The sharp absorption peak at 600–450 cm− 1 indicates the presence of Zn-O stretching vibration46. These substances may cause the reduction of metal or metal oxide into ions and the formation of nanoparticles47. X-ray diffraction analysis (Fig. 4) revealed the 2θ characteristic peaks of ZnO NPs at 31.70°, 47.5°, 54°, 56.60°, 68.8° and 75.20° for (112), (126), (200), (202), (004) and (002) planes of the crystal lattice, correspondingly. The minor peak at 28.5° may be occur due to organic moieties from the green synthesis process. These peaks suggested the presence of the hexagonal Wurtzite type of ZnO NPs and were consistent with previous studies48. The ZnO NPs crystalline structure is indicated by strong, narrow diffraction peaks. This result supported the existence of crystals of given ZnO NPs and this is in agreement with the previously reported green synthesized ZnO NPs used for biomedical applications49. In the zeta potential the majority of ZnO nanoparticles have a negative charge (-25.9 mV). The results indicate that the ZnO NPs synthesized from the aqueous extract of Acorn fuit do not combine at all and were stable at room temperature (Fig. 5). In the SEM image, (Fig. 6) ZnO NPs are found in a small-sized. Remarkably, there are not enough big particles and the majority of ZnO NPs have the same dimensions. Additionally, the ZnO NPs are slightly clumped together. This is due to the fact that biosynthetic ZnO NPs have a larger surface area and that their long-lasting affinity results in agglomeration or aggregation50. It can be said that ecological considerations have a significant impact on nanoparticle stability and agglomeration. As a result, the nanoparticles stick to one another and instinctively form asymmetrical clusters51. These ZnO NPs demonstrated tremendous antibacterial activity against E. coli (with zone of inhibition of 15 ± 02 mm,18 ± 04 mm, 21 ± 03 mm and 24 ± 05 mm) and S.aureus (with zone of inhibition of 17 ± 03 mm, 20 ± 06 mm, 22 ± 03 mm and 26 ± 02 mm). Amoxicillin (standard drug) had antibacterial potential against S. aureus (with zones of inhibition of 8 ± 04 mm, 10 ± 02 mm, 12 ± 05 mm, and 14 ± 06 mm) and E. coli (with zones of inhibition of 7 ± 03 mm, 10 ± 05 mm, 12 ± 03 mm, and 16 ± 05 mm) Tables 1 and 2. This outcome was supported by a prior study that assessed zinc oxide nanoparticles based on Moringa oleifera gum52. Molecular docking was performed to re-check and confirm the biological interaction between the beta-lactamase and ZnO nanoparticles. Molecular docking was performed. Docking studies of ZnO NPs revealed a variety of probable interactions with the binding site of substrate of receptor protein; the binding site residues entangled in the interactions include ARG275, ARG244, SER235, LYS234, ALA237, GLY238, ASN170, GLU104, GLU166, ASN132, SER70, SER130 and LYS73. The docking score and interaction details are enlisted in Table 3. The synthesized nanoparticles demonstrated a considerable interaction with the Beta-Lactamase binding site residues having a docking score of -8.2 kcal/mol. Furthermore, the standard drug Amoxicillin was also docked to the active site of target ASN170, ASN132, SER70, SER235, VAL216, ALA237 and ARG244. The binding affinity of amoxicillin with beta-lactamase is relatively high (-6 kcal/mol) in comparison to the nanoparticle, which is in agreement with the experimental findings (Fig. 8). At the doses of 5, 15, 30, 45, and 60 ppm, the larvae mortality was 20 ± 1.23%, 55 ± 2.01%, 70 ± 1.67%, 90 ± 3.10% and 100% respectively. Furthermore, the LC50 and LC90 values were 13.6 ppm and 45 ppm, respectively (Fig. 9). These findings are consistent with previously reported findings52. In Histological studies Normal Control group shown the stomach caecum anatomy is normal. The layer of epithelial cells in the untreated larvae of Cx. quinquefasqiatus stomach caecum was well-preserved. The center of the cell contains the ovoid nuclei (Fig. 10A). At ZnO NPs dose 5ppm, the caecum and stomach showed the first symptoms of intoxication. The histopathology of Cx. quinquefasciatus larvae revealed significant damage to the epithelial columnar cells as well as morphological changes at this level (Fig. 10B). At a ZnO NPs dose of 15ppm, there is a noticeable onset of vacuolization at the apical level and some cells appear mildly hypertrophied. The cells were infiltrated by these vacuoles. We occasionally observed an expansion of the intercellular gaps (Fig. 10C). We observed a cytoplasmic rejection of cell material that was interspersed with the food column at a ZnO NPs dose of 30 ppm, and the stomach ceacae epithelial cells began to rupture (Fig. 10D). The treated larvae with a ZnO NPs dose of 45 ppm, a consistent microvilli border is seen in the gastric lumen. Some cells displayed because of lysis and degraded (Fig. 10E). These cells are well-developed. The majority of the epithelial cells vacuolated and degraded at a ZnO NPs dose of 60 ppm of treatment. Based on the concentration, histopathological results show that ZnO NPs have various harmful effects on the two distinct areas of the midgut epithelium. Large vacuoles of various sizes with ruptured membranes at the apical side of the epithelial cells and widespread cellular microvillar disruption were the main cytopathological changes (Fig. 10F). These results are supported by a prior study in which Melia azedarach extract was used53. The histopathological effects of zinc oxide nanoparticles as a bio-insecticide against Cx. quinquefasciatus larvae are reported for the first time in this article. According to the current study, the antibacterial and larvicidal activity increased as the nanoparticle concentration increased. This suggests that ZnO NPs can be employed as an eco-friendly alternative to conventional antibacterial and larvicidal remedies.
Conclusions
In the current study, the Acorn Fruit mediated ZnO NPs have been synthesized. The characterization showed that they are spherical and irregular in shape. The activity of these nanoparticles against bacterial pathogens resulted in a higher zone of inhibition as compared to amoxicillin. In the in-silico analysis, these nanoparticles demonstrated a considerable interaction with the Beta-Lactamase binding site residues with a docking score of -8.2 kcal/mol that confirmed the successful inhibition of the given enzyme which catalyzes a lot of antibiotics. Thus, these nanoparticles combated microbes by blocking their resistive enzyme, which is the novel property of these ZnO NPs. Moreover, they revealed effective larvicidal effects against the tested Cx. quinquefasciatus. This study therefore concludes that ZnO NPs mediated by acorn fruit may be used as effective approaches to combat microorganisms, control mosquito larval populations and might have applications in the biomedical and pharmaceutical fields.
Recommendations and future perspectives
The use of only two bacterial strains, one mosquito species and the other one is inhibition of beta lactamase enzyme from the Acorn Fruit extract limits this research. Further studies with a broader range of microorganisms and other mosquito species and enzyme-based experiments are recommended.
Data availability
The data presented in this study are available on request from the corresponding author.
References
Salam, M. A. et al. July. Antimicrobial resistance: a growing serious threat for global public health. Healthcare 11, 13, 1946. MDPI. (2023).
Parvathy, C. R. & Praseetha, P. K. Evaluation of anti-diabetic potential of anti-microbial carbon quantum Dots from vitis vinifera seeds. Nano Biomed. Eng. 15 (1), 28–35 (2023).
Menichetti, A., Mavridi-Printezi, A., Mordini, D. & Montalti, M. Effect of size, shape and surface functionalization on the antibacterial activity of silver nanoparticles.J. Functional Biomaterials, 14 (5), 244. (2023).
Camenzind, T., Mason-Jones, K., Mansour, I., Rillig, M. C. & Lehmann, J. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nat. Geosci. 16 (2), 115–122 (2023).
Vitasovic, T. et al. Antibacterial action of Zn2 + ions driven by the in vivo formed ZnO nanoparticles. ACS Appl. Mater. Interfaces. 16 (24), 30847–30859 (2024).
Ijaz, M., Zafar, M., Islam, A., Afsheen, S. & Iqbal, T. A review on antibacterial properties of biologically synthesized zinc oxide nanostructures. J. Inorg. Organomet. Polym Mater. 30 (8), 2815–2826 (2020).
Bianchini Fulindi, R. et al. P.I. and Zinc-based nanoparticles reduce bacterial biofilm formation. Microbiol, Spectrum, 11 (2), .e04831. (2023).
Mohd Yusof, H., Rahman, A., Mohamad, N. A., Hasanah Zaidan, R. & Samsudin, A. A. U. and Antibacterial potential of biosynthesized zinc oxide nanoparticles against poultry-associated foodborne pathogens: an in vitro study. Animals, 11(7), 2093. (2021).
Adeyemi, J. O., Oriola, A. O., Onwudiwe, D. C. & Oyedeji, A. O. Plant extracts mediated metal-based nanoparticles: synthesis and biological applications. Biomolecules, 12 (5), 627. (2022).
Nebbak, A., Almeras, L., Parola, P. & Bitam, I. Mosquito vectors (Diptera: Culicidae) and mosquito-borne diseases in North Africa. Insects,13 (10), p.962. (2022).
Saleem, M. A. & Lobanova, I. Mosquito-borne diseases. In Dengue Virus Disease (57–83). Academic. (2020).
Paul, J. Blood and lymphatic infections. In Disease Causing Microbes (247–314). Cham: Springer International Publishing. (2024).
Kan, H. World health organization air quality guidelines 2021: implication for air pollution control and climate goal in China. Chin. Med. J. 135 (5), 513–515 (2022).
Singh, S., Gupta, A., Waswani, H., Prasad, M. & Ranjan, R. Impact of pesticides on the ecosystem. In Agrochemicals in Soil and Environment: Impacts and Remediation (157–181). Singapore: Springer Nature Singapore. (2022).
Vinotha, V. & Vaseeharan, B. Bio-fabricated zinc oxide and cry protein nanocomposites: Synthesis, characterization, potentiality against Zika, malaria and West nile virus vector’s larvae and their impact on non-target organisms. Int. J. Biol. Macromol. 224, 699–712 (2023).
Inácio, L. G., Bernardino, R., Bernardino, S. & Afonso, C. Acorns: From an Ancient Food to a Modern Sustainable Resource. Sustainability, 16 (22), 9613. (2024).
Muhammad, N., Castillejo, M. Á., Rey, M. D. & Jorrín-Novo, J. V. An overview of oak species in Pakistan: Past, present, and future research perspectives. Forests, 14 (4), p.777. (2023).
Umair, M., Altaf, M., Bussmann, R. W. & Abbasi, A. M. Ethnomedicinal uses of the local flora in Chenab riverine area, Punjab province Pakistan. Journal of ethnobiology and ethnomedicine, 15 (1), 7. (2019).
Szabłowska, E. & Tańska, M. Acorns as a source of valuable compounds for food and medical applications: A review of Quercus species diversity and laboratory studies. Applied Sciences,14 (7), 2799. (2024).
Umar, M. et al. Green synthesis of iron oxide NPs (IONPs) by using aqueous extract of parthenium hysterophorus Linnaeus for the In-vitro antidiabetic and antiinflammatory activities. J. Pure Appl. Microbiol., 18(4). (2024).
Aboelnga, A., Salaheldin, H. & Elsayed, A. Antibacterial activity using eco-friendly bio-synthesized zinc nanoparticles. Egypt. J. Chem. 67 (2), 555–562 (2024).
Umar, M. et al. Green synthesis of silver nanoparticles using Azadirachta indica seeds aqueous extract and evaluation of their Anti-diabetic potentials through in vitro and in Silico analysis. Indus J. Bioscience Res. 3 (10), 28–36 (2025).
Mohammad, A. M. M., Shaker, A. M. M., Salem, O. M. M., Elmashay, A. H. & Hassani, R. Screening the larvicidal activity of ZnO, CuO nanoparticles, and Neem seed oil extract against the full armyworm spodoptera Frugiperda (JE Smith)(Lepidoptera, Noctuidae). Emirates J. Food Agric. 36, 1–9 (2024).
Braga, C. Q. et al. de Avila Botton, S. and Pereira, D.I.B., Exposure of Culex quinquefasciatus to the oomycete Pythium insidiosum: A protocol for in vitro studies. Fungal Biology, 127 (4), 969–974. (2023).
Abdelghani, G. M., Ahmed, A. B. & Al-Zubaidi, A. B. Synthesis, characterization, and the influence of energy of irradiation on optical properties of ZnO nanostructures. Sci. Rep., 12(1), 20016. (2022).
Jothibas, M., Paulson, E., Mathivanan, A., Srinivasan, S. & Kannan, K. S. Biomolecules influences on the physiochemical characteristics of ZnO nanoparticles and its enhanced photocatalysis under solar irradiation. Nanatechnol. Environ. Eng. 8 (2), 511–533 (2023).
Yadav, S., Nadar, T., Lakkakula, J. & Wagh, N. S. Biogenic synthesis of nanomaterials: bioactive compounds as reducing, and capping agents. In Biogenic Nanomaterials for Environmental Sustainability: Principles, Practices, and Opportunities (147–188). Cham: Springer International Publishing. (2024).
Pasieczna-Patkowska, S., Cichy, M. & Flieger, J. Application of Fourier transform infrared (FTIR) spectroscopy in characterization of green synthesized nanoparticles. Molecules, 30 (3), 684. (2025).
Kumawat, G. et al. Characterization of green-synthesized zinc oxide nanoparticles and its influence on post-harvest shelf-life of garlic against black mold disease caused by Aspergillus niger. Frontiers in Microbiology, 16, 1532593. (2025).
Jain, D. et al. Microbial fabrication of zinc oxide nanoparticles and evaluation of their antimicrobial and photocatalytic properties. Front. Chem. 8, 778 (2020).
Sena, S., Ochatt, S. J. & Kumar, V. Application of green synthesized nanoparticles in medicinal plant research: revisiting an emerging eco-friendly approach. Plant. Cell. Tissue Organ. Cult. (PCTOC). 155 (2), 345–384 (2023).
ALBERT, H. M. Nanotechnology: Physicochemical and Green Synthesis, Characterizations and Applications (Perfect Writer Publishing, 2023).
Haleem, A., Javaid, M., Singh, R. P., Rab, S. & Suman, R. Applications of nanotechnology in medical field: a brief review. Global Health J. 7 (2), 70–77 (2023).
Kar, S., Sanderson, H., Roy, K., Benfenati, E. & Leszczynski, J. Green chemistry in the synthesis of pharmaceuticals. Chem. Rev. 122 (3), 3637–3710 (2021).
Murthy, S., Effiong, P. & Fei, C. C. Metal oxide nanoparticles in biomedical applications. In Metal Oxide Powder Technologies (233–251). Elsevier. (2020).
Alamri, A. A. et al. Chemical Composition of Anabasis articulata, and Biological Activity of Greenly Synthesized Zinc Oxide Composite Nanoparticles (Zn-NPs): Antioxidant, Anticancer, and Larvicidal Activities. Agronomy, 14(8), 1742. (2024).
Mubeen, B. et al. Nanotechnology as a novel approach in combating microbes providing an alternative to antibiotics. Antibiotics, 10(12), 1473. (2021).
Zhang, S. et al. Green synthesis, biomedical effects, and future trends of Ag/ZnO bimetallic nanoparticles: An update. Nanotechnology Reviews, 14(1), 20250186. (2025).
Socha, W., Kwasnik, M., Larska, M., Rola, J. & Rozek, W. Vector-borne viral diseases as a current threat for human and animal health One Health perspective. Journal of Clinical Medicine, 11(11), 3026. (2022).
Pichl, T., Wedderburn, C. J., Hoskote, C., Turtle, L. & Bharucha, T. A systematic review of brain imaging findings in neurological infection with Japanese encephalitis virus compared with dengue virus. Int. J. Infect. Dis. 119, 102–110 (2022).
Sabzghabaei, G. & Raisi, M. The effects of important groups of chemical pesticides (Organochlorine, organophosphate and Carbamate) on environment and humans. Hum. Environ. 19 (3), 69–78 (2021).
Zaller, J. G. What is the problem? Pesticides in our everyday life. In Daily Poison: Pesticides-an Underestimated Danger (1–125). Cham: Springer International Publishing. (2020).
Malik, S., Muhammad, K. & Waheed, Y. Nanotechnology: a revolution in modern industry. Molecules, 28(2), 661. (2023).
Onen, H. et al. Mosquito-borne diseases and their control strategies: an overview focused on green synthesized plant-based metallic nanoparticles. Insects, 14 (3), 221. (2023).
Goswami, M., Adhikary, N. C. & Bhattacharjee, S. Effect of Annealing Temperatures on the Structural and Optical Properties of Zinc Oxide Nanoparticles Prepared by Chemical Precipitation Method 158, 1006–1015 (Optik, 2018).
Sedky, A., Afify, N., Ali, A. M. & Algarni, H. Structural, FTIR, optical and photoluminescence investigation of Zn1-xRExO nanoparticles for optical and power operation devices. J. Electroceram. 49 (2), 33–52 (2022).
Soltys, L., Olkhovyy, O., Tatarchuk, T. & Naushad, M. Green synthesis of metal and metal oxide nanoparticles: Principles of green chemistry and raw materials. Magnetochemistry, 7(11), 145. (2021).
Chen, M. A Composite System Using Ultra High-Performance Fibre-Reinforced Concrete and Cross-Laminated Timber (University of Toronto (Canada), 2016).
Vishunumurthy, G. & Bhaskar, A. A comprehensive study on the impact of aluminum doping on X-Ray diffraction peak profile Analysis, Structural, Morphological, and optical properties of ZnO nanoparticles synthesized by Sol–Gel Auto-Combustion. J. Electron. Mater. 54 (3), 2146–2166 (2025).
Kulkarni, D. et al. Biofabrication of nanoparticles: sources, synthesis, and biomedical applications. Front. Bioeng. Biotechnol. 11, 1159193 (2023).
Yeap, S. P., Lim, J., Ooi, B. S. & Ahmad, A. L. Agglomeration, colloidal stability, and magnetic separation of magnetic nanoparticles: collective influences on environmental engineering applications. J. Nanoparticle Res., 19 (11), 368. (2017).
Sarwar, K., Nazli, Z. I. H., Munir, H., Aslam, M. & Khalofah, A. Biosynthesis of zinc oxide nanoparticles using Moringa oleifera leaf extract, probing antibacterial and antioxidant activities.Sci. Rep., 15 (1), p.20413. (2025).
Kausar, S., Jabeen, F., Latif, M. A. & Asad, M. Characterization, dose dependent assessment of hepatorenal oxidative stress, hematological parameters and histopathological divulging of the hepatic damages induced by Zinc oxide nanoparticles (ZnO-NPs) in adult male Sprague Dawley rats. Saudi journal of biological sciences, 30(9), p.103745. (2023).
Acknowledgements
All the authors are thankful to the Deanship of Scientific Research at Imam Mohammed Ibn Saud University (IMSIU).
Funding
This work has supported and funded by the Deanship of Scientific Research at Imam Mohammed Ibn Saud University (grant number IMSIU-DDRSP2601).
Author information
Authors and Affiliations
Contributions
“Conceptualization, M.U. and M.S.; methodology, H.A; software, M.U.; validation, M.U., A.A.C and M.A.; formal analysis, A.K.; investigation, M.S, A.K and H.A.; resources, M.U.; data curation, M.A. M.N and A.K; writing original draft preparation, M.U.; writing review and editing, A.A.C M.A M.N.; visualization, M.S., H.A and A.K; supervision, M.U.; project administration, M.U. and M.A; funding acquisition, A.A.C. and M.A .M.N. All authors have read and agreed to the published version of the manuscript”.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Umar, M., Ahmad, M., Sadeeq, M. et al. Green synthesis and characterizations of zinc oxide nanoparticles using acorn fruit extract for antimicrobial, larvicidal and in silico activities. Sci Rep 16, 7072 (2026). https://doi.org/10.1038/s41598-026-36137-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-36137-6