Abstract
Additional exploration is required to determine how to treat patients with moderate aortic regurgitation (AR) in rheumatic mitral valve (MV) surgery. This study compared clinical outcomes in patients undergoing non-surgical treatment (NT) of aortic valve (AV), aortic valvuloplasty (AVP), and aortic valve replacement (AVR). This multicenter, retrospective observational cohort study included 338 moderate AR patients undergoing rheumatic MV surgery from January 2015 to January 2024. We followed up with patients for a median of 43.4 months. Furthermore, more-than-mild aortic valve dysfunction (AVD) in the follow-up period was our primary outcome. Our secondary outcomes were all-cause mortality and cardiac valve reoperation before discharge and during follow-up. We classified patients into three groups, namely, NT (n = 128), AVP (n = 91), and AVR (n = 119) groups, respectively. Moreover, 33.0%, 30.1%, and 3.9% of patients achieved our primary outcome, while 4.2%, 5.6%, and 9.6% of them attained our secondary outcomes in the three groups, respectively. We observed that fewer patients from the AVR group had achieved the primary outcome than those in the NT [adjusted relative risk (RR), 0.41; 95% confidence interval (CI), 0.21–0.68; p = 0.002] and AVP groups (adjusted RR, 0.14; 95% CI, 0.04–0.43; p = 0.002), respectively. The secondary outcomes did not significantly differ among the three groups. Without increasing surgical risks, the concurrent AVR significantly improves AV status in moderate AR patients undergoing rheumatic MV surgery throughout follow-up. None or mild AVD was observed in many patients from the NT group during the follow-up, thereby warranting the delay of surgery for AV.
Similar content being viewed by others
Introduction
Owing to substantial expenditure, rheumatic heart disease (RHD) remains a significant burden in low socio-demographic index regions1. In China, approximately 55.1% of cases of valvular heart disease are caused by RHD2. More than one-third of RHD patients exhibited combined mitral valve (MV) and aortic valve (AV) lesions3. The interdependence of the MV and AV makes surgical management of these patients a critical clinical challenge. The MV and AV demonstrate structural connections, characterized by the continuity of the anterior mitral annulus with the junction of the AV sinus, along with functional associations mediated through left atrial (LA) and left ventricular (LV) remodeling and alterations of the LA-LV pressure gradient, which collectively modulate their hemodynamic interactions4,5,6.
During MV surgery, aortic stenosis (AS) was more likely to require treatment than AR7,8,9. Many international guidelines uniformly recommend concurrent surgical intervention for moderate AS [Class of Recommendation (COR): IIa], but are inconsistent regarding the surgical management of moderate aortic regurgitation (AR) in conjunction with MV surgery10,11. The American Heart Association guidelines advocate synchronous AV surgery (COR: IIa) with a low level of evidence, whereas the lack of uniform surgical recommendations by the European Society of Cardiology reflects the scarcity of high-level evidence. Moreover, limited research is available on patients with moderate AR during rheumatic MV surgery.
Our previous single-center, retrospective study suggested that some patients with predominant moderate AR might experience partial resolution following an isolated rheumatic MV surgery7. However, this study was retrospective and had numerous confounding factors. This comprised moderate AS and AR, as well as the aggregation of aortic valvuloplasty (AVP) and aortic valve replacement (AVR) into a single category. Additionally, limited studies are available on the management of moderate AR during rheumatic MV surgery. Consequently, we compared clinical outcomes among patients managed with non-surgical treatment (NT), AVP, or AVR during rheumatic MV surgery to assess the need for concurrent AV surgery and determine an optimal approach. This was a multicenter, retrospective observational cohort study, which followed the STROCSS reporting guidelines12.
Materials and methods
Ethical approval
This study was performed as per the Declaration of Helsinki (revised in 2013). The ethics board of Beijing An Zhen Hospital approved our study protocol (Institutional Review Board document KS2022064). Our study’s clinical registration was ChiCTR2200067151 (registered on December 28, 2022). Informed consent was waived because of the retrospective nature.
Patients
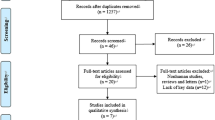
Inclusion criteria were adults aged ≥ 18 years undergoing surgical intervention for rheumatic mitral valve disease, with a confirmed diagnosis of moderate aortic regurgitation, either alone or in combination with mild aortic stenosis. Exclusion criteria encompassed patients with MV pathology of non-rheumatic etiologies, those requiring concomitant aortic root surgery, and individuals lacking preoperative echocardiographic assessment. We included 338 consecutive patients meeting the aforementioned inclusion and exclusion criteria at Beijing An Zhen Hospital and Guangdong Zhongshan People’s Hospital, China, from January 2015 to January 2024. We classified patients into three groups, namely, the NT (n = 128), AVP (n = 91), and AVR (n = 119) groups, respectively (Fig. 1). These patients occupied 15.2% (338/2226) of those undergoing rheumatic MV surgery at the above institutions during this period. We used transthoracic echocardiography (TTE) before the discharge and during the follow-up. Methods involving TTE for evaluating RHD grade have been previously described13. Especially, the criteria for moderate AR are as follows: a vena contracta width (VCW) of 3–6 mm, a Pressure Half Time of 200–500 milliseconds, an effective regurgitant orifice area of 0.10–0.29 cm2, a regurgitant volume of 30–59 ml, and a regurgitant fraction of 30–49%. The TTE images of several typical patients in the three groups were shown in Appendix 2.
Study design and patients included. AVD, aortic valve dysfunction; NT, non-surgical treatment; NT, non-surgical treatment; AVP, aortic valvuloplasty; AVR, aortic valve replacement.
Surgical technique
During MV and AV surgeries, a median sternotomy was performed utilizing the routine aorto-bicaval cannulation technique under the conditions of cold cardioplegic arrest and moderate hypothermia. More detailed information on MV repair has been provided14. As for the treatment of AV, the decision on performing NT, AVP, or AVR was made according to valvular disease severity evaluated previously, the method of cardioplegia perfusion, and the discretion of cardiac surgeons. Patients could choose the prosthetic valve type for usage after providing full informed consent. We routinely performed surgical ablation for permanent atrial fibrillation (AF) and tricuspid valve repair for mild or greater tricuspid regurgitation (TR).
The surgeons perform AVP on patients with relatively well-preserved valve conditions. AVP retains native AV tissue compared to AVR. However, its current clinical application remains limited to MVP15. Unlike degenerative AV lesions, rheumatic AR starts at the leaflet edges13, and primarily manifests as fibrotic proliferation. Valve body and annular fibrosis/calcification might occur later. In moderate rheumatic AR, fibrotic nodules at the coaptation margin induce distortion and stiffness. Therefore, partial fibrotic nodule resection could restore leaflet coaptation and improve AR16. However, the progression of the disease might cause AV thickening, fibrosis, commissural fusion, and calcification, thereby inducing complications in AVP by shrunken cusps9. Our AVP surgical approaches encompassed several repair techniques. These predominantly included shaving (58/89, 65.2%), along with commissurotomy (36/89, 40.4%), decalcification (7/89, 7.9%), commissural suspension (23/89, 25.8%), and AV neocuspidization (AVneo) (3/89, 3.4%). The first four methods are respectively employed to address thickened leaflets and margins, commissure adhesion, mild calcifications, and commissure avulsion. They can be applied individually or in combination, depending on the patient’s pathological conditions. Previous studies have also confirmed that the AVneo technique yields favorable mid-term outcomes17,18. The details of the AV repair approach for two patients in the AVP group were unavailable.
Double-valve surgery is far more complex than single-valve surgery. The conventional approach for double-valve replacement surgery in our center is as follows: following an incision in the aorta and the direct perfusion of the cardioplegia solution into the coronary ostia to induce cardiac arrest, the myocardial surface is cooled by applying ice chips. The mitral valve is then accessed through an approach involving the right atrium and the interatrial septum. The mitral valve leaflets, subvalvular apparatus, and the aortic valve leaflets are subsequently resected. Initially, a mitral valve prosthesis is implanted and tested, followed by the suturing of the interatrial septum. After that, an aortic valve prosthesis is implanted and tested, followed by the suturing of the aortic incision. If the patient has concomitant TR, a tricuspid valve annuloplasty may also be carried out, followed by the suturing of the right atrial incision.
Study endpoints
Our primary endpoint was more-than-mild aortic valve dysfunction (AVD), demonstrated using TTE in follow-up. Our secondary endpoints included all-cause mortality and cardiac reoperation before discharge and during the follow-up. The more-than-mild AVD was classified as native AVD in the NT and AVP groups, and as prosthetic AVD in the AVR group. The valve-related complications included major bleeding, new-onset stroke, valvular thrombosis, infective endocarditis, as well as severe arrhythmias requiring electrical cardioversion or ablation therapy.
Follow-up
Patients were followed up by telephone or outpatient services. The mean and median follow-up periods were 48.4 and 43.4 [Interquartile Range (IQR), 19.5–72.0] months, respectively. Subsequently, we achieved a follow-up rate of 95.0% (321/338).
Statistical analysis
Normally distributed continuous variables were summarized as mean ± SD and analyzed using Student’s t-test. Non-normally distributed continuous variables were summarized as median (IQR) and analyzed using the Mann-Whitney U-test. Categorical data were presented as frequencies and proportions and compared using the Chi-square test or Fisher’s exact test as appropriate. All missing data were left blank.
Poisson regression and Cox proportional hazard models helped to calculate relative risks (RR) and hazard ratios (HR), respectively. Multivariable Poisson regression and Cox regression analyses were conducted by adjusting for age, gender, AS, AV calcification, preoperative left ventricular end-diastolic dimension (LVEDD), and MV repair.
The survival curve was plotted by the Kaplan-Meier approach, while the groups’ survival rate was compared by the log-rank test. Statistical analysis was completed with R software version 4.2.2. All two-sided values of p < 0.05 denoted statistical significance.
Results
Patient features
We included 338 consecutive patients with moderate AR undergoing rheumatic MV surgery. Their average age at the time of surgery was 55.20 ± 10.71 years, with female patients occupying 66.6% of the total cohort (n = 225). The NT and AVP groups had a mild New York Heart Association (NYHA) functional class (p < 0.001), mild AS (p < 0.001), lower AV peak velocity (p < 0.001), lower AV calcification (p < 0.001), and enhanced MV repair rate (p < 0.001) than in AVR group, respectively. Moreover, the NT group showed a decreased regurgitation area of AV (p < 0.001), a decreased VCW of AV (p < 0.001), lower LVEDD value (p = 0.003), lower left ventricular end-systolic dimension (LVESD) value (p = 0.002) compared with AVP and the AVR group, respectively (Tables 1 and S1-1). VCW is a semi-quantitative echocardiographic parameter for assessing the severity of AR. There were only 7 (5.5%), 18 (19.8%), and 23 (19.3%) patients from the NT, AVP, and AVR groups with VCW ≥ 5 mm. The remaining basic characteristics did not significantly differ among the three groups. Additionally, AS grade was significantly related to AV calcification (χ2 = 48.3, p < 0.001). Table 1 shows the three groups’ basic characteristics, encompassing their demographic and clinical characteristics. Table S1-1 displays the pairwise comparison of the preoperative outcomes within the three groups. In the AVR group, 63.9% (76/119) of patients underwent mechanical valve replacement, while 36.1% (43/119) opted for biological valves. The average age of patients in the biological valve group was higher than that in the mechanical valve group, and the remaining baseline characteristics were comparable between the two groups.
Periopertative outcomes
Tables 2 and S1-2 display the perioperative data and pairwise comparisons, respectively. In our study, there was no in-hospital mortality. Adverse postoperative events, like cardiac reoperation in hospital (p = 0.75) and new-onset stroke (p = 0.79), did not significantly differ. The NT and AVP groups displayed an enhanced postoperative AR severity relative to the AVR group (p < 0.001, Table S1-2, Fig. 2). There was no severe patient-prothesis mismatch in the AVR group. During cardiopulmonary bypass (CPB), most patients (119/128) in the NT group underwent aortic root cardioplegia perfusion, while the rate for direct perfusion of cardioplegia was only 7.0% (9/128). The median CPB time and aortic cross clamp (ACC) time were 118.0 (IQR,98.3–137.8) min and 86.5 (IQR, 72.0–100.8) min, respectively. All patients in the AVP and AVR groups underwent direct perfusion of cardioplegia.
Severity of aortic regurgitation before operation, after operation and during follow-up for different surgical groups. NT, non-surgical treatment; AVP, aortic valvuloplasty; AVR, aortic valve replacement.
Predictors for postoperative AR and AVR
After adjustment through multivariable Poisson regression analyses, compared to NT group, undergoing AVP procedure was identified as an independent protective factor against a higher postoperative AR severity [relative risk (RR), 0.67; 95% confidence interval, 0.47–0.93; p = 0.02, Table S2-1]. Besides, a multivariable Poisson regression suggested that the severe NYHA functional class, AS, AV calcification, large LVEDD and VCW, and concurrent MV replacement were associated with a higher AVR feasibility, compared with NT and AVP groups (Table S2-2).
Follow-up of clinical outcomes
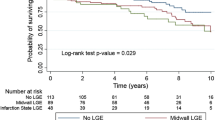
Although the total follow-up rate was 95.0% (321/338), 5.0% of patients could not be contacted again. Premature all-cause death during follow-up was reported in 2 (1.7%), 2 (2.2%), and 8 (7.0%) cases from the NT, AVP, and AVR groups, respectively. However, the survival rate was not significantly different (log rank p = 0.37, Figure S1-2). Furthermore, MV reoperation in follow-up was performed in 2 (1.7%), 2 (2.2%), 2 (1.8%) cases from the NT, AVP, and AVR groups, respectively. One patient (1.1%) from the AVP group underwent AV reoperation, whereas 2 (0.9%) from the AVR group had to undergo AV reoperation due to AV perivalvular leakage and aortic bioprosthetic valve destruction, respectively. Detailed information on valve reoperations during follow-up can be found in Table S3. The freedom from cardiac valve reoperation rate was also not significantly different among the three groups (log rank p = 0.94, Figure S1-3). Although the valve-related complications in the AVR group (20/114, 18.7%) was the highest, the rate of freedom from valve-related complications was not significantly different among the three groups (log rank p = 0.57, Figure S1-4). The follow-up duration of the three groups were comparable (p = 0.15, Tables 3 and S1-3).
Follow-up of echocardiographic outcomes
Furthermore, 287 (84.9%) patients had echocardiographic follow-up data. No significant difference was observed in MV or tricuspid valve dysfunction during the follow-up (p = 0.09, p = 0.77, respectively); the NT and AVP groups displayed more severe AR than the AVR group (p < 0.001, Table 4 and S1-3). Three (2.7%) and one (1.4%) cases from the NT and AVP groups developed severe AVD, respectively, whereas one (1.0%) case from the AVR group developed severe aortic paravalvular leak. Follow-up severe AVD did not significantly differ (p = 0.85). Besides, there were only 37 (33.0%), 22 (30.1%), and 4 (3.9%) patients from the NT, AVP, and AVR groups with more-than-mild AVD in follow-up, respectively. To sum up, the AVR group demonstrated significantly superior outcomes in terms of follow-up AR compared to both the NT and AVP groups (p < 0.001), while no statistically significant difference was observed between the AVP and NT groups (p = 0.78, Fig. 2, Table S1-3). Moreover, AVD conditions during follow-up in each group showed improvements relative to those observed preoperatively (Fig. 2).
Predictors for study endpoints
For primary endpoint, the AVR group displayed a reduced follow-up more-than-mild AVD risk relative to the NT (RR, 0.34; 95%CI, 0.19–0.54; p < 0.001) and AVP (RR, 0.13; 95% CI, 0.04–0.34; p < 0.001) groups, respectively; however, the AVP group was not significantly different from the NT group (RR, 0.91; 95% CI, 0.53–1.53; p = 0.73). After age, gender, AS, AV calcification, LVEDD, and MV repair adjustments, multiple Poisson regression analyses demonstrated that the AVR group showed a reduced follow-up more-than-mild AVD risk compared with the NT (adjusted RR, 0.41; 95%CI, 0.21–0.68; p = 0.002) and AVP (adjusted RR, 0.14; 95% CI, 0.04–0.43; p = 0.002) groups, while the AVP group did not significantly differ from the NT group (adjusted RR, 0.87; 95% CI, 0.49–1.50; p = 0.62).
Moreover, among patients who did not undergo AVR, using multivariable Poisson regression, we found that CAD served as the independent risk factor (RR, 2.13; 95% CI, 1.01–4.08; p = 0.032) of more-than-mild AVD in follow-up for NT and AVP groups (Table S2-3). As for the MV, multivariable Poisson regression showed that MV repair was an independent risk factor of the follow-up mitral regurgitation (MR) severity (RR, 6.29; 95% CI, 2.13–21.95; p = 0.003, Table S2-4).
However, secondary endpoints did not exhibit any significant difference among the three groups, as suggested by Cox proportional hazards regression analysis (Table 5). We obtained the same findings using the log-rank test. Event-free survival was not significantly different among the three groups during the ten-year follow-up (log-rank p = 0.62, Figure S1-1).
Additional analysis
Given the pathophysiological interdependence between MV and AV, a stratified analysis was conducted to examine the impact of distinct MV lesion subtypes—specifically, predominant mitral stenosis (MS), predominant MR, and mixed MV lesions—on postoperative and follow-up AR in the NT group. Preoperative echocardiographic data revealed that patients in the MR-predominant subgroup demonstrated the largest LVEDD (p < 0.001); the mixed-lesion subgroup exhibited the lowest left ventricular ejection fraction (p = 0.01). The severity of AR was not significantly different among the three MV lesion subgroups in terms of postoperative and follow-up echocardiography, respectively (p = 0.71, p = 0.28, Table S4).
The subgroup analyses stratified by age, gender, AF, NYHA functional class, pulmonary artery hypertension (PAH), MV dysfunction, mild AS, and MV surgery suggested that the AVR group consistently displayed a decreased AVD risk (Figures S2-1–S2-3), without any interactions (Table S5).
To investigate whether different approaches of AVP had an impact on the echocardiographic outcomes, we conducted a univariable Poisson regression analysis. The results revealed that different surgical approaches of AVP had no significant effect on postoperative AR or follow-up more-than-mild AVD (Tables S6-1 and S6-2). Additionally, there were no significant differences in all-cause mortality (8.3% vs. 4.8%, p = 0.73) and the incidence of more-than-mild prosthetic AVD (4.8% vs. 2.6%, p = 0.98) during follow-up between patients undergoing mechanical AVR and those undergoing biological AVR. During the follow-up period, one patient in each group underwent reoperation for aortic valve due to paravalvular leakage of the mechanical valve and structural deterioration of the biological valve, respectively. The patient outcomes showed no significant differences based on the type of prosthesis used or the repair approach employed.
Discussion
In cardiac surgery, rheumatic MV disease with concomitant AVD represents a prevalent clinical scenario. Current guidelines recommend synchronous surgical intervention for severe AVD cases when other cardiac surgery is performed [Class of Recommendation (COR): I]10,11. However, for patients undergoing MV surgery with concomitant mild AVD, undertaking prophylactic AVR for mild AVD during MV surgery cannot be justified based on preventing delayed reoperations in some of those patients19. On one hand, mild AVD during rheumatic MV surgery exhibits an indolent natural history; a study has demonstrated a low 10-year risk of progression to severe AVD or need for AVR in untreated cases20. On the other hand, patients who underwent conservative treatment for mild AVD demonstrated comparable long-term clinical outcomes and echocardiographic parameters to those undergoing AVP or AVR modalities3.
Focusing on the issue of moderate AR during rheumatic MV surgery, we obtained the following primary results: (1) The AVR group outperformed the NT and AVP groups regarding the follow-up AVD; (2) Many patients from the NT group showed absence of or had mild AVD in the follow-up, comparable to those from the AVP group, and (3) All-cause mortality, cardiac valve reoperation rates, and valve-related complications did not significantly differ within the three groups.
Many studies have demonstrated that double-valve surgery carries higher risks compared to single-valve procedures21,22,23. However, the mortality and cardiac reoperation rates did not significantly differ postoperatively and during the follow-up period among the three groups (Table 3, Figure S1-1). Besides, with the development of surgical techniques, the in-hospital mortality of patients who underwent double-valve or triple-valve procedures was null in our cohort, which was lower than that reported before21,22,23.
Although there were no significant differences in clinical outcomes among the three groups, the AVR group outperformed the NT and AVP groups regarding the follow-up AVD. Following that, we investigated the influencing factors of follow-up AVD in the NT and AVP groups. Multiple Poisson regression demonstrated that CAD might predict the occurrence of more-than-mild AVD during the follow-up period in the NT and AVP groups (Table S2-3). These findings suggested that a comprehensive follow-up was required for CAD patients postoperatively. Besides, stratified analysis was performed to evaluate the impact of MV lesions on follow-up AVD in the NT group. The types of MV lesions can influence the hemodynamic effects and clinical manifestations of AR. The coexistence of MR and AR might impose significant hemodynamic burdens24,25, while MS mitigates the increased AR-induced LV volume overload. Theoretically, isolated intervention for MS alone might augment LV volume loading26. However, stratified analysis demonstrated that the type of preoperative MV pathology showed no significant impact on follow-up AR in the NT group (Table S4).
The long-term durability and indications of AVP remain controversial27,28. Moreover, many studies lack comparative data between NT and AVP groups in rheumatic moderate AR cases, we found that AVP ameliorated AVD to some extent and that AVP was an independent protective factor against more severe postoperative AR compared to NT (Table S2-1). But it showed no significant superiority over NT cases in terms of follow-up more-than-mild AVD (Table 5). However, in this study, AVP still demonstrated potential benefits. Firstly, its early and follow-up clinical outcomes showed no significant differences compared to the NT and AVR groups. Secondly, compared to AVR, AVP exhibited a slightly lower rate of valve-related complications during the follow-up period and could avoid the long-term risks of anticoagulation therapy and the disadvantage of bioprosthetic valve deterioration in the distant future29. Thirdly, compared to the NT group, the preoperative VCW in the AVP group was significantly higher, indicating more severe valve pathology in the AVP group. Moreover, AVP was an independent protective factor for postoperative AR. Thus, AVP is a reasonable option for patients with suitable AV anatomy, whose AV need concurrent surgical intervention as assessed by surgeons, and who underwent MVP. However, the long-term durability of AVP warrants further investigation, and further studies are required to validate the efficacy of AVP.
Our study’s conclusions support the guideline recommendation that for patients with concomitant moderate AR, AVR therapy should be considered, as it can more definitively improve the patients’ AR condition. However, our research also indicates that NT and AVP are also viable approaches for some patients with moderate AR. The selection among the three methods primarily depends on the surgeon’s clinical experience. Based on the aforementioned results, we have also summarized the experience of our center as followed: (1) on the premise of ensuring adequate myocardial protection, aortic root cardioplegia perfusion might be initiated without AV intervention. (2) In cases where the perfusion of cardioplegia was inadequate, characterized by low cardioplegia infusion pump pressure, LV distension, incomplete cardiac arrest, or poor reflux of solution at the coronary sinus ostia, it was important to promptly perform an incision in the ascending aorta to expose the coronary artery ostia and switch to direct coronary perfusion of the cardioplegia solution. Following the thorough evaluation of the AV, most of the patients underwent AVP or AVR based on the degree of AV lesions, while only a small portion of patients did not undergo any AV intervention. (3) patients who had worse cardiac function, underwent mitral valve replacement, exhibit AS or AV calcification, and especially those with VCW ≥ 5 mm were more likely to receive AV surgery. the aforementioned conclusions should be considered more cautiously for these patients.
In summary, AR conditions were slightly alleviated in most of the patients of the NT group. During mid-term follow-up, the risk of progression to severe AR remained low at 2.7% in the NT group. This holds greater clinical significance in the era of transcatheter aortic valve implantation. Besides, asymptomatic severe AVD also typically does not require AVR10,11,30. However, the AVP group could not show superior clinical outcomes compared with the NT group during follow-up. This suggested that the AVP procedure, which primarily focused on leaflet thinning and commissurotomy, required additional improvement and research in the management of moderate rheumatic AR. Overall, AR improved to different degrees post-treatment of rheumatic MV disease. This might be attributed to LV remodeling and the use of diuretics7.
Limitations
Our study had a few limitations. Firstly, our follow-up period was insufficient. Given the slow progression of AVD, further follow-up is necessary to assess the long-term prognosis of all three groups. Moreover, we included only the patients’ recent TTE findings. Additional TTE data at multiple time points during follow-up were needed to evaluate the progression of AVD. Secondly, while rheumatic alterations constitute the primary etiology underlying concurrent AR, degenerative and functional pathological elements nonetheless also play a small role in the development of AR in certain patients. Notably, there exists a deficiency in precise analysis concerning the causative factors of moderate AR. Thirdly, although all patients included in this study had moderate AR, the severity of moderate AR in the NT group was generally mild overall. Despite conducting multivariable regression analysis to adjust for baseline conditions, there may still be a risk of selection bias, which could lead us to overestimate the clinical prognosis of the NT group.
Conclusions
For moderate AR patients in rheumatic MV surgery, concurrent AVR significantly improves AV conditions during follow-up without increasing the surgical risks. Most patients from the NT group had none or mild AVD in the follow-up period. Therefore, delaying surgical management for AV and undergoing regular follow-up examinations can be a safe and reasonable strategy.
Data availability
The anonymous raw data that support the findings of this study are available from corresponding author (Wenjian Jiang, email: [jiangwenjian@ccmu.edu.cn](mailto: jiangwenjian@ccmu.edu.cn) ) but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are available from the authors upon reasonable request and with permission of Beijing An Zhen Hospital.
Abbreviations
- ACC:
-
Aortic cross clamp
- AF:
-
Atrial fibrillation
- AR:
-
Aortic regurgitation
- AS:
-
Aortic stenosis
- AV:
-
Aortic valve
- AVD:
-
Aortic valve dysfunction
- AVP:
-
Aortic valvuloplasty
- AVR:
-
Aortic valve replacement
- CAD:
-
Coronary artery disease
- CI:
-
Confidence interval
- COR:
-
Class of recommendation
- CPB:
-
Cardiopulmonary bypass
- HR:
-
Hazard ratios
- LA:
-
Left atrium
- LV:
-
Left ventricular
- LVEDD:
-
Left ventricular end-diastolic dimension
- LVESD: Left ventricular end-systolic dimension MR:
-
Mitral regurgitation
- MS:
-
Mitral stenosis
- MV:
-
Mitral valve
- NT:
-
Non-surgical treatment
- NYHA:
-
New York Heart Association
- OR:
-
Odds ratio
- PAH:
-
Pulmonary artery hypertension
- RHD:
-
Rheumatic heart disease
- RR:
-
Relative risk
- SD:
-
Standard deviation
- TR:
-
Tricuspid regurgitation
- TTE:
-
Transthoracic echocardiography
- VCW:
-
Vena contracta width
References
Chen, Q. F. et al. Global, regional, and national burden of valvular heart disease, 1990 to 2021. J. Am. Heart Assoc. 13(24), e037991 (2024).
Yang, Y. et al. Current status and etiology of valvular heart disease in china: a population-based survey. BMC Cardiovasc. Disord. 21(1), 339 (2021).
Hwang, H. Y., Kim, K. H. & Ahn, H. Attitude after a mild aortic valve lesion during rheumatic mitral valve surgery. J. Thorac. Cardiovasc. Surg. 147(5), 1540–1546 (2014).
Ho, S. Y. Anatomy of the mitral valve. Heart 88(Suppl 4(Suppl 4), iv5–10 (2002).
Unger, P. & Tribouilloy, C. Aortic stenosis with other concomitant valvular disease: aortic regurgitation, mitral regurgitation, mitral stenosis, or tricuspid regurgitation. Cardiol. Clin. 38(1), 33–46 (2020).
Mantovani, F. et al. When aortic stenosis is not alone: Epidemiology, pathophysiology, diagnosis and management in mixed and combined valvular disease. Front. Cardiovasc. Med. 8, 744497 (2021).
Liu, C. et al. Attitude towards a moderate aortic valve dysfunction during rheumatic mitral valve surgery: a retrospective cohort study. J. Thorac. Dis. 17(1), 187–197 (2025).
Weingarten, N. et al. Aortic valve replacement for moderate aortic stenosis during mitral surgery improves survival free from severe stenosis. Asian Cardiovasc. Thorac. Ann. 31(6), 475–482 (2023).
Afifi, A., Hosny, H. & Yacoub, M. Rheumatic aortic valve disease-when and who to repair? Ann. Cardiothorac. Surg. 8(3), 383–389 (2019).
Otto, C. M. et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 143(5), e72–e227 (2021).
Vahanian, A. et al. 2021 ESC/EACTS guidelines for the management of valvular heart disease. Eur. Heart J. 43(7), 561–632 (2022).
Mathew, G. et al. STROCSS 2021: strengthening the reporting of cohort, cross-sectional and case-control studies in surgery. Int. J. Surg. 96, 106165 (2021).
Pandian, N. G. et al. Recommendations for the use of echocardiography in the evaluation of rheumatic heart disease: A report from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 36(1), 3–28 (2023).
Fu, J. et al. Outcomes of mitral valve repair compared with replacement for patients with rheumatic heart disease. J. Thorac. Cardiovasc. Surg. 162(1), 72–82 (2021). e7.
Kunihara, T. Aortic valve repair for aortic regurgitation and preoperative echocardiographic assessment. J. Med. Ultrason. (2001). 46(1), 51–62 (2019).
Li, Y. et al. Mid-term clinical outcome analysis of aortic valve repair for rheumatic aortic valve disease. Chin. J. Thorac. Cardiovasc. Surg. 37(8), 467–471 (2021).
Ozaki, S. et al. Midterm outcomes after aortic valve neocuspidization with glutaraldehyde-treated autologous pericardium. J. Thorac. Cardiovasc. Surg. 155(6), 2379–2387 (2018).
Benedetto, U. et al. Aortic valve neocuspidization with autologous pericardium in adult patients: UK experience and meta-analytic comparison with other aortic valve substitutes. Eur. J. Cardiothorac. Surg. 60(1), 34–46 (2021).
Antunes, M. J. Commentary: The aortic valve after rheumatic mitral valve surgery. Remarkably stable! J. Thorac. Cardiovasc. Surg. 165(2), 630–631 (2023).
Kim, H. R. et al. The fate of aortic valve after rheumatic mitral valve surgery. J. Thorac. Cardiovasc. Surg. 165(2), 622–629 (2023). e2.
Vassileva, C. M. et al. Outcome characteristics of multiple-valve surgery: comparison with single-valve procedures. Innovations 9(1), 27–32 (2014).
Rankin, J. S. et al. The society of thoracic surgeons risk model for operative mortality after multiple valve surgery. Ann. Thorac. Surg. 95(4), 1484–1490 (2013).
Iung, B. et al. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. Eur. Heart J. 24(13), 1231–1243 (2003).
Hagendorff, A. et al. Expert proposal to analyze the combination of aortic and mitral regurgitation in multiple valvular heart disease by comprehensive echocardiography. Clin. Res. Cardiol. 113(3), 393–411 (2024).
Tribouilloy, C., Bohbot, Y. & Unger, P. Mitral regurgitation in patients with severe aortic regurgitation: when misery loves company. J. Am. Coll. Cardiol. 76(3), 247–250 (2020).
Chen, C. R. et al. Percutaneous balloon mitral valvuloplasty for mitral stenosis with and without associated aortic regurgitation. Am. Heart J. 125(1), 128–137 (1993).
Tamer, S. et al. Late results of aortic valve repair for isolated severe aortic regurgitation. J. Thorac. Cardiovasc. Surg. 165(3), 995–1006e3 (2023).
Zhao, M. et al. Aortic valve repair for the treatment of rheumatic aortic valve disease: a systematic review and meta-analysis. Sci. Rep. 12(1), 674 (2022).
Chen, C. Y. et al. Bioprosthetic versus mechanical mitral valve replacements in patients with rheumatic heart disease. J. Thorac. Cardiovasc. Surg. 165(3), 1050–1060e8 (2023).
Kaul, S. Aortic valve replacement for asymptomatic severe aortic stenosis: is enthusiasm exceeding the evidence? J. Am. Coll. Cardiol. 85(14), 1511–1514 (2025).
Acknowledgements
None.
Funding
Beijing Anzhen Hospital Major Science and Technology Innovation Fund (No. KCZD202203, KCQY202201). National Science Foundation of China (No. 82422007, 82241205, 82170487), Beijing Natural Science Foundation.
Author information
Authors and Affiliations
Contributions
(I) Conception and design: Wen-jian Jiang, Chuang Liu; (II) Administrative support: Hong-jia Zhang, Wen-jian Jiang, Xiao-long Wang, and Mao-zhou Wang; (III) Provision of study materials or patients: Xiao-long Wang, Wen-jian Jiang, and Hong-jia Zhang; (IV) Collection and assembly of data: Chuang Liu, Zhou Liu, Jia-jun Liang, Song-hao Jia, Mao-zhou Wang, and Pei-yi Liu; (V) Data analysis and interpretation: Chuang Liu, Zhou Liu, Jia-jun Liang, Ming-xuan Zhang, Mao-zhou Wang; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors.
Corresponding authors
Ethics declarations
Consent for publication
Informed consent was waived because of the retrospective nature.
Ethical approval and consent to participate
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by ethics board of Beijing An Zhen Hospital (Institutional Review Board document KS2022064). The clinical registration number of this study is ChiCTR2200067151. Informed consent was waived because of the retrospective nature.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, C., Liu, Z., Liang, Jj. et al. Clinical outcomes and management strategies for moderate aortic regurgitation in patients undergoing rheumatic mitral valve surgery. Sci Rep 16, 7059 (2026). https://doi.org/10.1038/s41598-026-37045-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37045-5