Abstract
The long-term prognosis of patients with distant metastasis of differentiated thyroid cancer (DM-DTC) after total thyroidectomy remains poorly defined. This study aims to assess the conditional survival (CS) of DM-DTC patients following total thyroidectomy and to analyze the key prognostic factors, with the objective of developing a more accurate survival prediction tool for clinical application. We retrospectively analyzed data from patients diagnosed with DM-DTC who underwent total thyroidectomy between 2004 and 2019, retrieved from the Surveillance, Epidemiology, and End Results (SEER) database. Overall survival (OS) was estimated using the Kaplan-Meier method. CS rates were calculated using the formula CS(y/x) = OS(y + x)/OS(x), where CS(y/x) represents the probability of surviving an additional y years after already surviving x years. Prognostic factors were identified using the least absolute shrinkage and selection operator (LASSO) regression and incorporated into a CS-nomogram model developed through multivariate Cox regression. The CS-nomogram was validated, and predictive factors were assigned point values. A risk stratification system was subsequently established using the optimal threshold of the total score. A total of 1,235 patients with DM-DTC who underwent total thyroidectomy were included, with a median follow-up of 51 months. Kaplan-Meier survival analysis revealed 3-year, 5-year, and 10-year OS rates of 81.7%, 70.4%, and 46.9%, respectively. CS analysis demonstrated a progressive increase in survival rates over time. The 10-year cumulative survival rate increased from 46.9% to 51.1%, 54.4%, 57.5%, 61.5%, 66.7%, 72.3%, 79.6%, and 85.5%, ultimately reaching 93.5% after surviving 1 to 9 years. Prognostic factors identified through LASSO regression and multivariate Cox regression analysis included age, sex, histological type, tumor size, and radioactive iodine therapy. A novel CS-nomogram for dynamic real-time survival prediction was successfully developed and validated, enabling identification of high- and low-risk patient groups. This study represents the first evaluation of CS patterns in DM-DTC patients following total thyroidectomy, demonstrating a gradual improvement in postoperative survival rates over time. A novel CS-nomogram model was successfully developed and validated, offering clinicians a personalized, dynamic, and real-time survival prediction tool. The risk stratification system derived from this model effectively distinguishes high- and low-risk patients, providing valuable guidance for follow-up and risk assessment.
Similar content being viewed by others
Introduction
Differentiated thyroid carcinoma (DTC) is the most common type of thyroid cancer, encompassing papillary thyroid carcinoma (PTC), follicular thyroid carcinoma (FTC), and oxyphilic thyroid carcinoma (OTC)1,2. The majority of DTC patients have favorable prognoses following standardized treatments, including surgery, radioactive iodine therapy (RAIT), and thyroid-stimulating hormone (TSH) suppression therapy3. However, approximately 6% to 34% of patients progress to distant metastatic differentiated thyroid carcinoma (DM-DTC)4,5. The common metastatic sites for DM-DTC are the lungs (63%), bones (25%), combined lung and bone (15%), and the central nervous system or other soft tissues (9%)6,7. According to the Surveillance, Epidemiology, and End Results (SEER) database, while the 5-year and 10-year survival rates for DTC patients are 96.8% and 95.8%, respectively, survival rates decline significantly once distant metastasis (DM) occurs, with 5-year and 10-year survival rates dropping to only 48.3% and 34.2%. Therefore, there is an urgent need for effective prognostic risk assessment methods and personalized follow-up strategies for DM-DTC patients.
Although several studies have conducted survival analysis and identified risk factors for DM-DTC patients, most of these studies rely on traditional survival analysis methods, which evaluate long-term prognosis from a static perspective8,9,10. However, these traditional approaches often fail to provide accurate prognostic assessments for patients with long-term survival and have not effectively improved follow-up adherence. Therefore, adopting real-time dynamic assessment methods to evaluate postoperative prognosis in DM-DTC patients is of particular importance. Conditional survival (CS), an emerging survival analysis method, dynamically evaluates the future survival probability based on the time a patient has already survived11,12,13,14. It has been widely applied in various types of cancer15,16,17. However, the CS analysis for postoperative DM-DTC patients has yet to be explored.
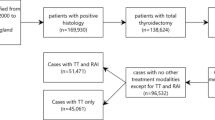
In this retrospective study, we included 1,235 postoperative DM-DTC patients from the SEER database (Fig. 1A) with the aim of assessing their CS, developing (Fig. 1B) and validating (Fig. 1C) a CS-based prognostic nomogram, and establishing a risk stratification system based on the predictive model (Fig. 1D). The primary objective was to determine the practical utility of this model in dynamic prognostic evaluation.
Overview of the research workflow. Patient selection (A).Development of the CS-based prognostic nomogram (B). Validation of the CS-based prognostic nomogram (C). Risk stratification system (D).
Methods
Patient selection
This study extracted data from the SEER Research Plus Data, 17 Registries (2000–2019), including patients diagnosed with DTC (ICD-10-CM code C73.9) and reporting DM. All patients underwent total thyroidectomy (RX Summ-Surg Prim Site (1998+) code 40 or 50). DM was reported at the initial staging based on Derived AJCC M (2004–2015), Derived SEER Combined M (2016–2017), and Derived EOD 2018 M (2018+). The SEER database, covering 34.6% of the U.S. population, includes data from 17 cancer registries across diverse geographic regions. Primary tumors were identified according to the International Classification of Diseases for Oncology, 3rd Edition (ICD-O-3), with histological codes 8050/3, 8260/3, 8290/3, 8330/3, 8331/3, 8335/3, 8339/3, 8340/3, 8341/3, 8342/3, 8343/3, 8344/3, 8350/3, 8450/3, 8452/3, 8453/3, and 8460/3. Patients with missing variables or survival records were excluded. Since the SEER database is publicly accessible and contains de-identified data, this study was exempted from ethical approval by the Ethics Committee of the Second Affiliated Hospital of Shenyang Medical College. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines18.
Clinical variables
We collected a variety of clinical variables for each patient, including gender, age at diagnosis, race, marital status, median household income, tumor size, histological type, regional nodes, and whether the patient received RAIT. For statistical analysis, age was categorized into two groups: <55 and ≥ 55 years. Tumor size was classified into three categories: ≤20 mm, 20–40 mm, and > 40 mm. Race was categorized as white, black, and other. Marital status was grouped into married, unmarried, and unknown. Median household income was divided into three brackets: <$60,000, $60,000–74,999, and >$75,000. Histological types included FTC, PTC, and OTC. Regional nodes was categorized as positive or negative. RAIT was classified as “yes” or “no.” The primary outcome was OS, defined as the time from diagnosis to death from any cause or the last follow-up, whichever occurred first.
Conditional survival analysis
The Kaplan-Meier method was used to estimate OS. CS was calculated using the formula CS(y/x) = OS(y + x)/OS(x)19, where CS(y/x) represents the probability that a patient survives an additional y years after already surviving x years of DM-DTC following total thyroidectomy; and OS(y + x) and OS(x) refer to the Kaplan-Meier estimates of OS for y + x years and x years, respectively.
CS-nomogram development and validation
The entire study cohort of DM-DTC patients who underwent total thyroidectomy was randomly divided into training and testing cohorts in a 7:3 ratio.First, in the training cohort, the Least Absolute Shrinkage and Selection Operator (LASSO) regression method was used to identify variables influencing postoperative survival in DM-DTC patients, combined with strict 10-fold cross-validation. The optimal predictor variables were selected based on the lambda-1se value. The selected predictors were then further validated through multivariate Cox regression analysis to assess their impact on survival. These independent prognostic factors were used to develop the CS-nomogram model. The model’s reliability and practicality were validated through the concordance index (C-index), calibration curve, receiver operating characteristic (ROC) curve, and decision curve analysis (DCA) results for both the training and testing cohorts. Based on the CS-nomogram model, risk scores for each patient were calculated, and the optimal cutoff value of the total score was determined to classify patients into high- and low-risk groups. Kaplan-Meier analysis was used to compare the prognostic differences between the risk groups.
Statistical analysis
Categorical data were expressed as frequencies and corresponding percentages. All statistical analyses were performed using R software (version 4.1.3), with a P-value of < 0.05 considered indicative of statistical significance.
Results
Baseline characteristics
A total of 1235 DM-DTC patients who underwent total thyroidectomy were enrolled in the study, with 864 patients assigned to the training cohort and 371 to the validation cohort. The cohort was predominantly female (55.8%), with 44.2% male, and 63.6% of patients were aged 55 years or older. PTC was the most prevalent histological type, comprising 82.6% of the cases, followed by FTC (13.6%) and OTC (3.8%). Tumor size distribution was as follows: 24.8% of patients had tumors smaller than 20 mm, 35.0% had tumors ranging from 20 to 40 mm, and 40.2% had tumors larger than 40 mm. Furthermore, 84.3% of patients had positive regional lymph nodes, and 76.8% received RAIT. The demographic and clinical characteristics of the participants are summarized in Table 1.
Overall and conditional survival
Of the 1235 patients, 452 (36.6%) died during the follow-up period, with a median follow-up time of 51 months. Traditional survival analysis revealed OS rates of 81.7%, 70.4%, and 46.9% at 3, 5, and 10 years, respectively (Fig. 2). CS analysis demonstrated a progressive increase in cumulative survival over time. The 10-year cumulative survival rate rose incrementally from an initial 46.9% to 51.1%, 54.4%, 57.5%, 61.5%, 66.7%, 72.3%, 79.6%, and 85.5%, ultimately reaching 93.5% after surviving 1 to 9 years (Fig. 2).
Conditional survival of DM-DTC patients following total thyroidectomy estimated using the Kaplan-Meier method after surviving for 0–9 years.
Construction a prognostic CS nomogram
In the training cohort, the LASSO regression method, coupled with rigorous 10-fold cross-validation, was employed to identify variables influencing postoperative survival in DM-DTC patients. The optimal predictor variables, including age, sex, histology type, tumor size, and RAIT, were selected based on the lambda-1se value, which minimizes the cross-validated prediction error plus one standard error, thereby yielding a more regularized and streamlined model compared to lambda.min (Fig. 3A, B). Multivariate Cox regression analysis further highlighted the significant impact of these predictors on OS (Fig. 3C, P < 0.05) and was subsequently used to construct the CS-nomogram model (Fig. 4).
Identification of prognostic factors. The least absolute shrinkage and selection operator (LASSO) regression was applied to screen for prognostic factors (A, B). A multivariate Cox regression forest plot illustrates the impact of these predictors on the OS of DM-DTC patients following total thyroidectomy (C). Abbreviations: FTC, follicular thyroid carcinoma; OTC, oxyphilic thyroid carcinoma; PTC, papillary thyroid carcinoma.
A conditional survival-based nomogram model successfully developed for predicting 10-year CS in DM-DTC patients following total thyroidectomy. Abbreviations: FTC, follicular thyroid carcinoma; OTC, oxyphilic thyroid carcinoma; PTC, papillary thyroid carcinoma; OS, overall survival; CS, conditional survival.
CS-nomogram validation
The prognostic CS-nomogram model for predicting postoperative survival in DM-DTC patients was rigorously validated and evaluated in both the training and validation cohorts. In the training cohort, the model achieved a C-index of 0.707 (95% CI: 0.675–0.739), and in the validation cohort, it was 0.713 (95% CI: 0.669–0.759), demonstrating its robust discriminative power in predicting postoperative survival. Calibration curve analysis further confirmed the model’s reliability, with the survival prediction curves for 3, 5, and 10 years in both cohorts closely aligning with the ideal 45-degree reference line, indicating excellent agreement between the predicted and actual outcomes (Fig. 5A, B). Moreover, the model’s predictive capacity was further corroborated by the AUC values from the ROC curves. In the training cohort, the AUCs at 3, 5, and 10 years were 0.724, 0.727, and 0.802, respectively, while in the validation cohort, the AUCs were 0.738, 0.706, and 0.793 (Fig. 5C, D). Finally, clinical DCA demonstrated significant net benefits at 3, 5, and 10 years, further underscoring the model’s clinical utility and potential value (Fig. 5E, F).
Evaluation of the discriminatory power, calibration, and clinical utility of the CS-nomogram. Calibration curves for the CS-nomogram in the training (A) and validation cohorts(B), demonstrating the agreement between predicted and observed 3-, 5-, and 10-year survival outcomes. ROC curves and AUC values for the CS-nomogram in both the training (C) and validation cohorts (D), highlighting its predictive accuracy. Decision curve analysis (DCA) for the CS-nomogram in the training (E) and validation cohorts (F), showcasing its clinical utility and net benefit across different threshold probabilities. Abbreviations: AUC, area under the curve.
CS-Nomogram-Based risk stratification
Based on the CS-nomogram model, the total OS score for each patient was calculated. Using the optimal cut-off value of 83.48 derived from the training cohort, patients were stratified into low-risk (total score < 83.48) and high-risk (total score > 83.48) groups (Fig. 6A, B). Kaplan-Meier survival analysis was then conducted to compare survival outcomes between the high-risk and low-risk groups in both the training and validation cohorts. The results revealed significant survival differences between the two groups (Fig. 6C, D). These findings further underscore the utility of the CS-nomogram model in effectively stratifying patient risk and predicting survival outcomes.
Development and validation of a risk stratification system using the CS-nomogram. (A) Distribution of risk scores derived from the CS-nomogram; (B) Identification of the optimal cut-off points using standardized log-rank statistics; Kaplan-Meier survival analysis with log-rank tests comparing survival outcomes across stratified risk groups in both the training (C) and validation (D) cohorts.
Discussion
To our knowledge, this is the first study to assess CS in patients with DM-DTC following total thyroidectomy. In this retrospective study involving 1,235 patients, CS analysis revealed a significant increase in survival rates as the duration of survival extended.Based on five key prognostic factors, including age, sex, histological type, tumor size, and RAIT, we constructed a CS-nomogram model. The model demonstrated excellent predictive performance through various evaluation methods and successfully established a risk stratification system.
In the 8th edition of the American Joint Committee on Cancer (AJCC) TNM staging system, DM in thyroid cancer are classified as stage IV, significantly correlating with reduced survival rates20. However, traditional survival analyses, based on fixed time points such as 3- or 5-year survival rates, provide limited prognostic information, particularly for long-term postoperative survivors. In contrast, CS offers a dynamic, time-dependent survival prediction that allows for continuous assessment of prognosis, delivering a more personalized and adaptive prognostic evaluation21,22,23. In this study, we assessed the actual and CS of DM-DTC patients following total thyroidectomy. Consistent with findings in other malignancies24,25,26, the cumulative survival rates of DM-DTC patients following total thyroidectomy significantly improved with prolonged survival time. Specifically, the likelihood of achieving a 10-year survival progressively increased from an initial 46.9%, ultimately reaching 93.5% after surviving nine years. This real-time, dynamic survival prediction holds promise in alleviating anxiety for patients and their families while providing clearer and more optimistic prognostic insights for long-term survivors.
Studies have demonstrated that total thyroidectomy combined with RAIT significantly improves the 10-year survival rate in DM-DTC patients (HR = 0.67; 95% CI: 0.52–0.87). In contrast, patients who did not undergo surgery and were treated with external radiation therapy and/or chemotherapy exhibited a significantly increased risk of mortality (HR = 3.33; 95% CI: 2.42–4.59)27. Given that this study focuses on assessing the long-term survival prognosis of patients who have received standard treatments, we concentrated exclusively on those who underwent total thyroidectomy.Numerous retrospective studies have validated the survival benefits of RAIT in DM-DTC patients. An analysis by Yang et al. of 11,832 DM-DTC patients from the National Cancer Database found that those not receiving RAIT had nearly double the all-cause mortality rate at 5 and 10 years compared to those who received RAIT28. A large-scale retrospective study involving over 100,000 patients found that RAIT significantly improved disease-specific survival (CSS) in DM-DTC patients, particularly in metastatic PTC, where RAIT notably enhanced the 50-month CSS29. Another large-scale retrospective study based on the SEER database confirmed that RAIT substantially increased the 5-year CSS in DM-DTC patients (83.6% vs. 74.1%)4.Our findings further corroborate these results, as we observed that RAIT significantly reduced the mortality risk in DM-DTC patients who underwent total thyroidectomy (HR = 0.57; 95% CI: 0.44–0.73), reinforcing the crucial role of RAIT in improving patient prognosis.
Previous studies on DM-DTC have identified several factors that potentially influence the OS of affected patients. Among these, age stands out as a critical prognostic factor, with older age strongly associated with a higher risk of mortality and poorer survival outcomes30,31. In this study, we found that patients aged 55 years or older were at a significantly increased risk of mortality, with a hazard ratio (HR) of 3.68, underscoring the necessity for more frequent monitoring and more aggressive treatment strategies.Similarly, Ma et al. developed and validated a survival nomogram for DM-DTC patients, incorporating six independent prognostic factors, including age, marital status, surgery type, lymphadenectomy, radiotherapy, and T-stage8. However, their model did not account for the substantial prognostic differences between different DTC histological types, nor did it incorporate CS analysis. Liu et al. pioneered the development of a CS-nomogram for poorly differentiated thyroid carcinoma (PDTC), demonstrating enhanced long-term survival predictions26. Unfortunately, no similar studies have yet explored CS patterns or models specifically for postoperative DM-PTC.In this study, we successfully established and validated a novel CS-nomogram for personalized, dynamic, and real-time survival prediction, providing valuable support for clinicians in optimizing treatment strategies based on the evolving risk profiles of patients.
Considering the dynamic improvement in CS observed in our cohort, this may have significant implications for postoperative management and follow-up strategies. The CS-based nomogram not only improves prognostic accuracy but also guides individualized follow-up scheduling and survivorship management. In this study, the total survival score based on the CS nomogram was used to stratify patients into low-risk (total score < 83.48) and high-risk (total score > 83.48) groups. With this risk stratification, low-risk patients can safely extend follow-up intervals after an initial period of intensive surveillance, thus reducing the burden on both patients and healthcare resources; whereas high-risk patients may require more frequent monitoring, earlier imaging or biochemical testing, and multidisciplinary reassessment. Additionally, regularly recalculating individualized survival probabilities during follow-up can support survivorship management and shared decision-making, providing objective prognostic information to both patients and clinicians with each follow-up update. Finally, the nomogram can be integrated into the electronic medical record system, so that when a patient’s conditional risk exceeds a predefined threshold, an automatic alert will be triggered, prompting timely treatment planning discussions or referral to supportive care services.
We acknowledge several limitations in this study. First, as a retrospective analysis, it is difficult to completely eliminate potential biases. Second, approximately half of the patients had a follow-up period of less than 5 years, and longer-term studies are needed to more comprehensively assess the long-term prognosis of DM-DTC patients. Third, the model lacks external validation regarding its applicability to other populations. Fourth, the SEER database contains limited variables that may influence the survival of DM-DTC patients, such as access to healthcare, imaging features, molecular markers (e.g., BRAF and/or TERT mutations), and TSH suppression therapy. Finally, this study primarily focused on the impact of tumor biology on patient survival, but did not fully consider other factors that may influence long-term health outcomes, such as advances in healthcare services and improvements in the management of comorbidities. Future studies should integrate these factors, incorporate more relevant variables and diverse populations, in order to provide a more comprehensive prognostic assessment and further enhance the applicability of the model.
Conclusions
This study is the first to explore the CS patterns in DM-DTC patients following total thyroidectomy, introducing a survival prediction model that is more dynamic and individualized compared to traditional methods. We successfully developed and validated a CS-nomogram capable of real-time survival forecasting, providing clinicians with a tool to optimize treatment strategies based on evolving patient risks. While the model demonstrated exceptional performance in predicting survival outcomes, further external validation in diverse populations is necessary to enhance its broad applicability and clinical utility. Moreover, long-term follow-up studies are warranted to refine the model’s predictive accuracy and investigate potential improvements in patient prognosis with extended survival.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author and the SEER database (https://seer.cancer.gov/).
References
Boucai, L., Zafereo, M. & Cabanillas, M. E. Rev. Jama 331(5), 425–435. https://doi.org/10.1001/jama.2023.26348 (2024) (PubMed PMID: 38319329).
Lim, H., Devesa, S. S., Sosa, J. A., Check, D. & Kitahara, C. M. Trends in thyroid cancer incidence and mortality in the united States, 1974–2013. Jama 317(13), 1338–1348. https://doi.org/10.1001/jama.2017.2719 (2017).
Zhang, L. et al. Molecular basis and targeted therapy in thyroid cancer: progress and opportunities. Biochim. Biophys. Acta Rev. Cancer. 1878 (4), 188928. https://doi.org/10.1016/j.bbcan.2023.188928 (2023). Epub 20230529.
Yang, Y. et al. Guiding the postoperative radioactive iodine-131 therapy for patients with papillary thyroid carcinoma according to the prognostic risk groups: a SEER-based study. J. Cancer Res. Clin. Oncol. 149 (19), 17147–17157. https://doi.org/10.1007/s00432-023-05299-5 (2023). Epub 20231002.
Baudin, E. & Schlumberger, M. New therapeutic approaches for metastatic thyroid carcinoma. Lancet Oncol. 8 (2), 148–156 (2007). doi: 10.1016/s1470-2045(07)70034-7. PubMed PMID: 17267329.
Osorio, M., Moubayed, S. P., Su, H. & Urken, M. L. Systematic review of site distribution of bone metastases in differentiated thyroid cancer. Head Neck. 39 (4), 812–818. https://doi.org/10.1002/hed.24655 (2017). Epub 20170111.
Akatani, N. et al. Long-term outcomes and prognostic factors of patients with lung metastases from differentiated thyroid cancer after radioiodine therapy in Japan. Endocr. J. 70 (3), 315–322. https://doi.org/10.1507/endocrj.EJ22-0463 (2023). Epub 20221224.
Ma, Q. et al. Development and validation of survival nomograms for patients with differentiated thyroid cancer with distant metastases: a SEER Program-based study. J Endocrinol Invest. ;47(1):115 – 29. Epub 20230609. (2024). https://doi.org/10.1007/s40618-023-02129-w. PubMed PMID: 37294407.
Hirsch, D. et al. Endocr. Pract. ;23(10):1193–1200. (2017). Epub 20170713. doi: 10.4158/ep171924.Or. PubMed PMID: 28704099.
Jaffar, S. et al. Predicting distant metastatic disease in differentiated thyroid cancer: a matched case-control study. ANZ J. Surg. 91 (4), 716–723 (2021). Epub 20210216. doi: 10.1111/ans.16652. PubMed PMID: 33590959.
Chen, Q. Y. et al. Conditional survival and recurrence of remnant gastric cancer after surgical resection: A multi-institutional study. Cancer Sci. 111 (2), 502–512. https://doi.org/10.1111/cas.14231 (2020). Epub 20191226.
Qaderi, S. M., Dickman, P. W., de Wilt, J. H. W. & Verhoeven, R. H. A. Conditional survival and cure of patients with colon or rectal cancer: A Population-Based study. J. Natl. Compr. Canc Netw. 18 (9), 1230–1237. https://doi.org/10.6004/jnccn.2020.7568 (2020). PubMed PMID: 32886900.
Abdallah, N. H. et al. Conditional survival in multiple myeloma and impact of prognostic factors over time. Blood Cancer J. 13 (1), 78. https://doi.org/10.1038/s41408-023-00852-4 (2023). Epub 20230515.
Hagens, E. R. C. et al. Conditional survival after neoadjuvant chemoradiotherapy and surgery for oesophageal cancer. Br. J. Surg. 107 (8), 1053–1061. https://doi.org/10.1002/bjs.11476 (2020). Epub 20200203.
Song, D., Yang, Z., Cai, L., Huang, H. & Gu, Z. Conditional survival analysis and dynamic survival prediction for intracranial solitary-fibrous tumor/hemangiopericytoma. J. Cancer Res. Clin. Oncol. 150 (2), 107. https://doi.org/10.1007/s00432-024-05629-1 (2024). Epub 20240228.
Shi, Y., Zheng, Y., Zhang, H., Dong, W. & Zhang, P. Dynamic estimates of survival in oncocytic cell carcinoma of the thyroid. Discov Oncol. 14 (1), 217. https://doi.org/10.1007/s12672-023-00839-4 (2023). Epub 20231130.
Liu, X. et al. Risk-Dependent Conditional Survival and Failure Hazard After Radiotherapy for Early-Stage Extranodal Natural Killer/T-Cell Lymphoma. JAMA Netw. Open. ;2(3):e190194. (2019). Epub 20190301. doi: https://doi.org/10.1001/jamanetworkopen.2019.0194. PubMed PMID: 30821826; PubMed Central PMCID: PMCPMC6484659.
von Elm, E. et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. ;370(9596):1453-7. (2007). https://doi.org/10.1016/s0140-6736(07)61602-x. PubMed PMID: 18064739.
Zabor, E. C., Gonen, M., Chapman, P. B. & Panageas, K. S. Dynamic prognostication using conditional survival estimates. Cancer 119 (20), 3589–3592. https://doi.org/10.1002/cncr.28273 (2013). Epub 20130801.
Perrier, N. D., Brierley, J. D. & Tuttle, R. M. Differentiated and anaplastic thyroid carcinoma: major changes in the American joint committee on cancer eighth edition cancer staging manual. CA Cancer J. Clin. 68 (1), 55–63 (2018). PubMed PMID: 29092098; PubMed Central PMCID: PMCPMC5766386.
Skuladottir, H. & Olsen, J. H. Conditional survival of patients with the four major histologic subgroups of lung cancer in Denmark. J. Clin. Oncol. 21 (16), 3035–3040. https://doi.org/10.1200/jco.2003.04 (2003). .521. PubMed PMID: 12915592.
Zamboni, B. A. et al. Conditional survival and the choice of conditioning set for patients with colon cancer: an analysis of NSABP trials C-03 through C-07. J. Clin. Oncol. 28 (15), 2544–2548 (2010). PubMed PMID: 20406942; PubMed Central PMCID: PMCPMC2881729.
Kim, Y. et al. Conditional survival after surgical resection of gastric cancer: a multi-institutional analysis of the Us gastric cancer collaborative. Ann. Surg. Oncol. 22 (2), 557–564. https://doi.org/10.1245/s10434-014-4116-5 (2015). Epub 20141007.
Kurta, M. L. et al. Prognosis and conditional disease-free survival among patients with ovarian cancer. J. Clin. Oncol. 32 (36), 4102–4112. https://doi.org/10.1200/jco.2014.55.1713 (2014). Epub 20141117.
Xu, L. et al. Conditional survival and changing risk profile in patients with gliosarcoma. Front. Med. (Lausanne). 11, 1443157. https://doi.org/10.3389/fmed.2024.1443157 (2024). Epub 20240906.
Liu, Z., Xu, Q., Xia, H. & Wang, M. Dynamic estimates of survival of patients with poorly differentiated thyroid carcinoma: a population-based study. Front. Endocrinol. (Lausanne). 15, 1375274. https://doi.org/10.3389/fendo.2024.1375274 (2024). Epub 20240913.
Wilhelm, A. et al. Disease-Specific survival trends for patients presenting with differentiated thyroid cancer and distant metastases in the united States, 1992–2018. Thyroid 33 (1), 63–73. https://doi.org/10.1089/thy.2022.0353 (2023). Epub 20221118.
Yang, Z., Flores, J., Katz, S., Nathan, C. A. & Mehta, V. Comparison of survival outcomes following postsurgical radioactive iodine versus external beam radiation in stage IV differentiated thyroid carcinoma. Thyroid 27 (7), 944–952. https://doi.org/10.1089/thy.2016.0650 (2017). Epub 20170517.
Guo, H. et al. Radioactive iodine therapy strategies for distinct types of differentiated thyroid cancer: a propensity score-matched analysis. Front. Endocrinol. (Lausanne). 14, 1158581. https://doi.org/10.3389/fendo.2023.1158581 (2023). Epub 20230817.
Orosco, R. K. et al. Analysis of age and disease status as predictors of thyroid cancer-specific mortality using the Surveillance, Epidemiology, and end results database. Thyroid 25 (1), 125–132 (2015). PubMed PMID: 25369076; PubMed Central PMCID: PMCPMC4291211.
Ganly, I. et al. Survival from differentiated thyroid cancer: what has age got to do with it? Thyroid 25 (10), 1106–1114. https://doi.org/10.1089/thy.2015.0104 (2015). Epub 20150812.
Funding
This study were supported under the Liaoning Provincial Science and Technology Plan project (Grant no.2022JH2/1013).The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Chenglin Sun, Ji Wu: proposed the research questions; was responsible for drafting and revising the manuscript; reviewed and approved the final version of the paper. Ying Qi, Junjie Zhang: was responsible for collecting and analyzing data; reviewed and approved the final version of the paper. Hongpeng Guo: was responsible for drafting and revising the manuscript; reviewed and approved the final version of the paper. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The SEER database can be accessed publicly and provides patient data without specific identification, so ethics approval and informed consent were not required.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, H., Qi, Y., Zhang, J. et al. Conditional survival and prognostic nomogram for distant metastatic differentiated thyroid cancer after total thyroidectomy. Sci Rep 16, 5897 (2026). https://doi.org/10.1038/s41598-026-37103-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37103-y