Abstract
Brain metastases are common in non-small cell lung cancer (NSCLC) and affect prognosis and survival. While frailty and sarcopenia are associated with the overall survival in NSCLC the impact on outcome and survival after surgery for brain metastasis is unknown. We therefore analyzed 179 patients (81 women) with NSCLC undergoing resection for brain metastasis between 2011 and 2020 retrospectively. Frailty was measured using the Clinical Frailty Scale (CFS). Temporal Muscle Volume (TMV) was assessed in preoperative T1w MRI. The median age was 63 years. Clinical frailty was present in about 20.6%. Mean follow-up was 11 months. Frailty correlated significantly with age (r = 0.36, p < 0.001) and smaller TMV (r=-0.24, p = 0.002). However, only measurement of TMV predicted impaired survival (median OS 34.5 vs. 10.3 months, p < 0.001). Physical performance after surgery was negatively affected by frailty (r=-0.72, p < 0.001) and positively by TMV (r = 0.2, p = 0.038). Major postoperative complications were more strongly associated with sarcopenia rather than frailty. Treatment response towards immunotherapy improved in the absence of sarcopenia (B = 2.48, p = 0.031). TMV is a predictor for survival after resection of brain metastasis and an indicator of treatment response to immunotherapy in patients with NSCLC. Accounting for sarcopenia in surgical decision making could improve patient selection for different treatment modalities.
Similar content being viewed by others
Introduction
Lung cancer is the second most common malignancy with an estimated yearly incidence of 2,206,771 cases worldwide in 2020 and accounts for 18% of cancer related deaths1.
In NSCLC brain metastases are frequent with 20% of all patients suffering from CNS dissemination. Brain metastases cause substantial disability, reduced quality of life and impaired prognosis2. The cumulative incidence rises throughout the course of the disease3,4. Lung cancer is largely diagnosed in older age5. Frailty, the clinical state of increased vulnerability and susceptibility to disease resulting from aging and age associated decline is known to influence therapy response and survival6,7,8. Although the prevalence of frailty increases with age, chronological age alone fails to sufficiently address these multifactorial changes9. Thus frailty has become a valuable predictor of outcome and mortality independent from biological age10,11,12,13. Frailty is prevalent in 45% of patients suffering from NSCLC and increases the mortality risk threefold13. It is associated with lower overall survival (OS) in early-stage NSCLC with patients more likely to die of causes unrelated to the primary malignancy14. Independent from NSCLC, it has been established that frailty poses an increased risk of postoperative complications, chemotherapy intolerance, disease progression, and death23. In the context of oncological surgery frailty is associated with an increased risk for major complications in most solid malignancies8,15,16,17,18,19.
Rather than age, frailty includes different characteristics, including sarcopenia. According to international recommendations, sarcopenia is defined by low muscle strength and quantity. Muscle mass can be assessed using CT or MRI20. In the context of neurological illnesses, temporal muscle assessment correlates closely with sarcopenia21,22. Advantages for the clinical application in neuro-oncologic surgery arise from the availability in this patient collective, while whole-body imaging might not be readily available in the initial setting when key therapy decisions are made.
Sarcopenia is an established predictor of impaired survival in lung cancer and other malignancies23,24,25,26,27,28. It increases the risk of all types of complications in patients undergoing oncological surgery27. However, data in the context of neuro-oncologic surgery is sparse. In the therapy of brain metastasis immunotherapy is of increasing relevance, but most data is derived from younger and fitter patients, not the older and frail29. The current data suggests that safety and tolerability of checkpoint inhibitors is similar, while the impact of adverse events in those with frailty increased greater than in younger and fit patients30,31. In general, patients affected by frailty show impeded response to immunotherapy while being at an increased risk of suffering toxicity and complications jeopardizing treatment success and adjusted therapy planning10,12,32,33. The relevance of frailty and sarcopenia for resection and survival in patients with NSCLC and brain metastasis is still unknown. Validated cut-off values defining sarcopenia as a marker of frailty in patients with NSCLC and brain metastasis are missing. It is the aim of this study to establish the impact of frailty and sarcopenia on surgical treatment and immunotherapy in patients with NSCLC and brain metastasis.
Methods
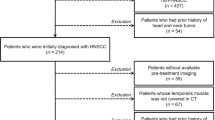
For the presented study we included patients who underwent resection for cerebral or cerebellar metastasis of non-small-cell lung cancer (NSCLC) at a single institution from 2011 to 2020. The present study follows the STROBE guidelines for cohort studies.
Patients
Patients were required to be over 18 years of age and to have undergone resection of cerebral or cerebellar NSCLC metastasis. All patients received tumor resection of at least one lesion. Progression free survival (PFS) and overall survival (OS) measured in months were defined from surgery until radiological progression or death respectively. Postoperative complications were defined as the emergence of pneumonia, pulmonary embolism (PE), deep-vein thrombosis (DVT), surgical site infection or rebleeding during the in-patient treatment period. Presence of any of these complications was termed combined as composite complications. During institutional multidisciplinary tumor board meetings, treatment decisions concerning the surgical procedure and adjuvant treatment were made prior to and after surgery.
To compare TMV values with a previously healthy cohort we included 31 patients who were treated at our department suffering from aneurysmal subarachnoid hemorrhage between January 2016 and December 2020. Patients were included if an MRI was performed within 14 days after hemorrhage to reduce the chance of bed-rest induced atrophy after critical illness.
Clinical scores
To evaluate patients according to their frailty and functional status we utilized the Karnofsky Performance Score (KPS), Clinical Frailty Scale (CFS), and Charlson Comobidity Index (CCI). The KPS quantifies the functional status of cancer patients on a scale from 0 (dead) to 100 (normal no complaints; no evidence of disease)34. The Clinical Frailty Scale is a common scoring method to evaluate frailty ranging from 1 to 7 with increasing severity35. As a cut-off for frailty a CFS of 4 or greater was used. To assess the burden of comorbidities we utilized the Charlson Comorbidity Index which is a scoring tool evaluating the 10-year mortality of patients with multiple comorbidities36.
Temporal muscle measurement
We opted for the evaluation of temporal muscle as it is readily available from pretherapeutic imaging in neuro-oncologic patients and exhibits good correlation with other imaging assessments of sarcopenia21,22. One way of assessing the temporal muscle is its thickness at an axial image but methodologies vary. Because of better reproducibility we instead opted for an assessment of the temporal muscle volume37.
Data was collected retrospectively from routine radiographic data. Data collection was planned beforehand and was not changed during this study. Volume of the temporal muscle was calculated from the routine immediate preoperative MRI using Sectra Workstation IDS7 software Version 22.2.6.4194 (Sectra AB, Sweden). For this purpose, a T1w isotropic non contrast enhanced sequence (T1 MPRAGE) was reconstructed in triplanar 1 mm slices. Afterwards the manual volume calculation tool was utilized to outline the temporal muscle with measurements being calculated automatically. Afterwards measurements were averaged between left and right sides and independently reviewed by a second researcher. In the case of non-identical measurements cases were discussed to reach consensus.
Statistics
For all ordinal or nominal variables absolute and relative frequencies were calculated. In case of rational scaled variables normal distribution was tested using the Shapiro-Wilk Test. The values of the normal-distributed variables were presented as the mean and standard deviation, while the values of the non-normal distributed variables were shown as the median and interquartile range. In both cases minima and maxima were calculated. Correlations were calculated using the spearman correlation coefficient. Survival was analyzed using Kaplan-Meier Curves and Cox Regression. For group comparisons we used the χ2-tests. Time-dependent receiver operating characteristic (ROC) analyses with calculation of the area under the curve (AUC) were performed in order to identify an optimal cutoff for OS and PFS. For the cut-off the maximal Youden Index (value for which sensitivity + specificity − 1) was chosen. Missing data was excluded from analysis.
Statistical analysis was performed using R: A language and environment for statistical computing R Core Team (2021) and IBM SPSS Version 26.0.0.0.
Ethics
According to the local laws of Rhineland Palatinate, Germany (Landeskrankenhausgesetz § 37) no formal approval and informed consent was necessary for this retrospective analysis. The patient data was de-identified before analysis.
Limitations
Our study has several inherent limitations. The retrospective design introduces a risk of bias. As assessment of frailty is a topic of ongoing debate with a multitude of tests used for classification, we decided on using broadly applied scoring systems for frailty, physical capacity and comorbidities in conjunction with temporal muscle volume.
For dichotomization we performed ROC analysis resulting in a cut-off value based on our cohort characteristics. And compared with a separate cohort suffering SAH but further external validation is needed for generalization of these results. As most other authors use their respective cohorts median TMV a universal cut-off is yet missing. The SAH cohort is of a considerably smaller size than our NSCLC cohort which is mainly due to the infrequent performance of timely MRI studies available in these patients. A short timeframe was chosen as we wanted to minimize the possibility of muscle loss after SAH to be included in our analysis.
Results
Baseline demographics
From 2011 to 2020 we included 179 patients in this study of whom 81 (45.3%) were women. The median age was 63 (56–69) years. At initial diagnosis AJCC Stage 1 was present in 3.93%, Stage 2 in 3.14, Stage 3 in 10.23% and Stage 4 in 82.67% of patients. Singular metastasis was present in 64 patients (35.8%) and 24 patients (13.4%) presented with infratentorial lesions. In 84 cases (46.9%) the central nervous system (CNS) was the first site of metastasis. Average time from disease diagnosis to first emergence of brain metastasis was 1 (0–13) months. PD-L1 was expressed on a median of 20% (2.25–70.25%) of the analyzed cells in histopathology. Of all patients 156 (87.2%) received radiotherapy with 85 (47.5%) receiving immunotherapy. Baseline variables are presented in Table 1.
The median Clinical Frailty Score (CFS) was 3 (3–4), classifying 37 patients (20.6%) as frail. Median Charlson Comorbidity Index (CCI) was 9 (8–10). Median Karnofsky Performance Score (KPS) before surgery was 80 (70–90), with the median post-surgery being 80 (80–90). Median TMV was 20.8 (14.2–26.7) cm3 with a mean of 21.1 ± 9.5 cm3.
To compare the TMV in our NSCLC cohort with previously healthy patients, we chose a cohort suffering from aneurysmal subarachnoid hemorrhage (SAH) as this disease has a characteristic sudden onset. Median TMV in patients suffering SAH was 16.6 cm3, with a mean of 18 ± 7.9 cm3. Functional Scores and temporal muscle volumes are presented in Table 2. Characteristics of the SAH cohort are presented in Table 3.
Median CRP was 3.9 (1.32–11.32) mg/L, median TSH was 0.78 (0.4–1.5) µIU/L, median Creatinine was 0.81 (0.73–0.99) mg/dL and median Albumin was 65 (35–74) g/L. Laboratory Values are presented in Table 4.
Frailty, sarcopenia and age
Frailty as assessed by CFS correlated significantly with age (r = 0.36, p < 0.001) and preexisting comorbidities (r = 0.23 p = 0.014).
Volume of the temporal muscle correlated with sex, being higher in male patients (r = 0.46, p < 0.001). Additionally, TMV correlated with CFS (r=−0.24, p = 0.002) and TSH showed a significant correlation with TMV (r = 0.25, p = 0.021).
There were no correlations between CFS, TMV and age, stage, localization or number of metastases, preoperative KPS and CCI. Association of frailty and sarcopenia is displayed in Fig. 1.
Distribution of average temporal muscle volume and clinical frailty scale. Individuals classified as sarcopenic are shown in red. (right) Exemplary axial T1 MRI images of sarcopenic and non-sarcopenic individuals. (left).
In order to classify patients by TMV ROC analysis was performed for 1-year PFS and OS. Optimal cut-off in both tests was found to be 15.5 cm3. For further analysis patients below this cut-off were considered sarcopenic, which applied to 53 patients (29.6%). ROC Curves and AUC statistics are supplied in Supplement 1.
Survival according to sarcopenia and frailty
Median follow-up after surgery was 11 (0–120) months with a median cerebral progression free survival (cPFS) of 8 (3 - 23) months, median systemic progression free survival (sPFS) of 13 (11 - 25) months and overall survival of 11 (8 - 34) months.
In order to assess possible influences of frailty on progression free- and overall survival (OS) patients were dichotomized according to CFS or TMV. As a cut-off to consider patients frail a CFS score of 4 was used, the cut-off in TMV to consider patients sarcopenic was below or equal to 15.5 cm3. Classifying patients by CFS did not show a significant difference in PFS or OS (cPFS: 24 vs. 20 months | sPFS: 13 vs. 7 months | OS: 34 vs. 29 months; p = 0.455 | p = 0.35 | p = 0.278).
Comparing patients based on sarcopenia according to the above-mentioned classification revealed a highly significant difference in median OS of 34.5 vs. 10.3 months (p < 0.001) but no significant difference according to cPFS (27.7 vs. 16.3 months, p = 0.354) or sPFS (17 vs. 8 months, p = 0.35). Cerebral PFS did exhibit a trend towards shorter survival when sarcopenia was present but failed to achieve statistical significance. While there was no significant difference between groups, patients that were not classified as frail or sarcopenic had a markedly longer follow-up available, suggesting possible further differences omitted from our analysis. Kaplan-Meier Curves are presented in Fig. 2.
Kaplan Meier curves and risk tables for systemic/cerebral progression-free survival and overall-survival according to sarcopenia (A-C) and frailty (D-F).
Response to immunotherapy
In patients receiving immunotherapy exhibited a higher therapy response as measured by PFS in patients with TMV > 15,5cm3 but statistical significance was not reached. (cPFS 38.3 vs. 13.5 months, p = 0.071 | sPFS 14 vs. 11 months, p = 0.94) In patients receiving radiotherapy there was no difference in cPFS (34.8 vs. 21 months, p = 0.476) or sPFS (16 vs. 8 months, p = 0.072). In both groups a higher OS was observed in patients with TMV above the cut-off. (Immunotherapy: PFS 45.9 vs. 11.6 months; radiotherapy: PFS 37.9 vs. 10.2 months both, p < 0.001)
Surgical treatment
Postoperative physical performance as measured by KPS was significantly influenced by frailty according to CFS. (preoperative r=−0.81, postoperative r=−0.72, difference in r = 0.27, all p < 0.001). Sarcopenia as assessed by TMV correlated significantly with postoperative KPS (r = 0.2, p = 0.038).
The postoperative complications that were assessed included pneumonia, pulmonary embolism, surgical infection, and rebleeding. The rate of any complication was defined as the composite complication rate. Pneumonia was significantly more common in frail patients (p = 0.037), but rates of composite complications (p = 0.136), surgical infection (p = 0.55), pulmonary embolism (p = 0.84), DVT (p = 0.59), and rebleeding (p = 0.44) were not different.
Postoperative complications were more frequent in patients classified as sarcopenic (pneumonia p = 0.021, surgical infection p = 0.072, pulmonary embolism p = 0.035, DVT p = 0.025). However, there was no difference in composite complications (p = 0.38) or rebleeding (p = 0.47). Frequencies of Complications are displayed in Table 5.
Multivariate analysis
In multivariate analysis shorter overall survival was significantly impacted by age (HR 1.1, p < 0.001) and sarcopenia (HR 2.0, p = 0.003) but not by frailty according to CFS (HR 0.8, p = 0.4), sex (HR 1.1, p = 0.6) or presence of complications (HR 1.6, p = 0.1). The cox regression model attained global significance with p <0.001. Cox regression for PFS failed to reach global significance. Forest Plots for both are displayed in Supplement 2.
Response to immune- or radiotherapy could independently be predicted by sarcopenia according to TMV (B 2.48, p = 0.031) but not age (B 0.08, p = 0.28) or frailty classified by CFS (B −0.05, p = 0.881).
Discussion
We aimed to assess the influence of frailty and sarcopenia on surgical treatment and immunotherapy in patients suffering brain metastasis from NSCLC. In our analysis sarcopenia independently predicted OS and response to immunotherapy. Its presence was associated with postoperative complications and reduced postoperative physical performance possibly indicating impeded recovery of affected patients. Frailty did not predict survival but correlated with impeded physical performance and the incidence of postoperative pneumonia. In recent years awareness for age-related changes and their consequences for therapeutic decisions has greatly increased. There is a mounting body of evidence pointing towards a need for more detailed assessment of these beyond numerical age38. In the context of age-related changes, a range of terms have garnered increasing attention in both clinical practice and research. Frailty and sarcopenia are of exceptional importance among these, and differences and areas of overlap are present39. Frailty is defined as a state of increased vulnerability and susceptibility to disease resulting from aging, whereas sarcopenia is characterized by the ongoing loss of muscle mass and function. Sarcopenia constitutes a significant component of frailty; however, frailty itself is a rarer condition, and it is characterized by more profound changes40. In the presence of cancer frailty and sarcopenia were both shown to influence treatment efficacy and results as well as tolerability of therapeutic measures like chemo- or immunotherapy23,28,41,42. Additionally neoplastic disease can increase muscle loss and further strain limited resources in the affected individual39.
Improvements in neuroimaging, and better prognosis through more effective systemic therapies lead to earlier and more frequent detection of CNS dissemination of systemic tumors43. Additionally, tumor incidence increases with age. Given the physical demands of multimodal therapy, prognostic markers and assessment tools for age-related changes are necessary to optimize individualized treatment. Multiple studies demonstrate the importance of assessing frailty when determining oncologic treatment44.
In the presented cohort, frailty according to CFS was present in 20.6% of all patients with NSCLC. In a meta-analysis the prevalence of frailty was reported to be as high as 45% of patients suffering from NSCLC13. The reported prevalence is higher, compared to the presented data and other literature. Frailty in a uniform cohort of patients with brain metastasis from NSCLC was found to be present in 24%.24 This proportion fits the prevalence of frailty in our presented cohort. Differences in frailty between patients with NSCLC per se and those with brain metastasis might be explained by differences in age or clinical performance status as CNS invasion occurs more often in later stages of the disease after longer previous periods of therapy3,45. Another possible explanation for the lower frailty rates in patients receiving surgical treatment for brain metastasis of NSCLC might be preselection and therapeutic bias by the treating physicians, leading to exclusion of patients that could potentially benefit from surgical treatment24. To prevent treatment disparity, more independent prognostic markers for patient stratification other than patients’ age and physician estimation, are needed.
Since frailty is a clinical state influenced by various age-related changes, it often occurs alongside other conditions. In our study frailty correlated with age, comorbidities, physical performance status as well as sarcopenia. As frailty is defined as a state of increased risk for adverse health outcomes that is determined by multiple factors and is associated with age, the observation in the presented cohort aligns with previous findings46. However, there was no independent correlation between frailty and survival in the presented cohort. This stands in contrast to previously published data where frailty is associated with all-cause mortality and specific mortality among cancer patients44. Prognostic relevance of frailty is established in different entities including glioma, colorectal carcinoma and NSCLC11,13,24,47. One potential contributing factor is the absence of a universally applicable assessment tools for frailty. Therefore, studies employ a variety of methodologies to identify patients afflicted with the condition, thereby accounting for potential discrepancies in data comparison13,24,48. Moreover, frailty evaluation frequently involves a certain degree of subjectivity, further complicating the comparison of findings49. An additional factor that may be considered is the relatively young age of the subjects in our cohort. Ilic et al. did not observe a prediction of survival by utilizing the modified Frailty Index in patients younger than 65 years of age who suffered from brain metastasis of non-small cell lung cancer (NSCLC)24. Yet in the broader oncologic literature frailty is to be considered a relevant factor for survival despite our observations13. However, differences in frailty assessment and spatial severity render frailty a particular hard to standardize and potentially bias-afflicted clinical parameter. Muscle loss is an important contributor to frailty and promises to add reproducibility in frailty assessment as it is less prone to fluctuation and interobserver variance24.
Retrospective measurements of sarcopenia frequently rely on available imaging as a standard using whole body- or lumbar paravertebral musculature as surrogate parameters20. Especially in oncologic studies imaging analyses are widely accepted as measurements of muscle strength might be confounded by cachexia caused by neoplastic disease39. The most relevant measurement in oncologic studies is the paravertebral muscle area at the L3 level39. As radiographic whole-body measurements aren’t necessarily available through standard diagnostics in the setting of neurosurgical treatment, temporal muscle volume is widely used and correlates well with measurements of body composition and frailty21. While some authors recommend assessment of sarcopenia to include muscle quality in oncologic studies muscle mass analysis is commonly used alone, as strength measurements can be impeded by tumor related cachexia which seems to be less relevant for muscle mass evaluations39. For this study temporal muscle was used because of its availability from preoperative cranial MRI. When assessing the temporal muscle, measuring muscle thickness and volume is commonly used. As measurement of TMT reduces a three-dimensional structure to a two-dimensional representation, we decided on volumetric analysis to possibly achieve more accurate representation of muscle mass. Measurement of TMT is performed parallel oriented to the anterior-posterior commissure line with the sylvian fissure and the orbital roof as anatomical landmarks26. To define sarcopenia in our cohort we performed ROC analysis for 1-year PFS and OS, 15.5cm3 were identified as cut-off value. In literature, the median temporal muscle thickness is often used for differentiation, yet there is a lack of evidence and broader applicability for those classification20,50. In the presented cohort, determination of individual AUC threshold values proved statistically superior with better estimation and potentially offers insight for future, disease specific, standardization. To validate the calculated TMV cut-off value and comparison with previously healthy patients suffering from aneurysmal subarachnoid hemorrhage was performed. Due to the apoplectic onset of SAH those patients provide evidence of baseline TMV to differentiate sarcopenia from the influence of cachexia during the course of malignant disease51. When comparing these cohorts the higher percentage of female patients in the SAH cohort needs to be noted which might reduce the temporal muscle mass in this cohort due to known differences between genders52. The calculated cut-off of 15.5cm3 is reasonably close to the median TMV of 16.6cm3 within the SAH cohort and well within the standard deviation of TMV in both cohorts. A similar cohort of patients with spontaneous SAH (median age 61) had a baseline TMV of 18.5cm3, where a TMV of 15cm3 and less, was associated with impaired functional outcome53. This observation is transferable to the presented Cohort of patients with brain metastasis in NSCLC and potentially argues for a more absolute state of sarcopenia to determine functional outcome across different disease.
In the presented dataset, sarcopenia independently predicts overall survival. A recent meta-analysis including 1675 patients suffering from brain metastasis of different entities found shortened PFS by sarcopenia54. Our observation is in line with these results. However, only studies utilizing lumbar paravertebral muscle mass were included in this meta-analysis. Since the applied measurement technique differ, we think this further supports the broader significance of sarcopenia. The interrelation of sarcopenia with age currently remains less clear in the setting of brain metastasis. In our multivariate analysis we observed a greater independent impact of sarcopenia (HR 2 p = 0.003) than age (HR 1.1 p < 0.001) on OS. Yet there is conflicting data in this regard. There are studies which only observed a relationship between sarcopenia and survival only in patients older than 65 years suffering from brain metastasis of NSCLC, while others observed a similar influence irrespective of age groups but found age to be another independent predictor of survival24,55. Evaluation of sarcopenia as a predicting factor on progression free survival in patients receiving immunotherapy showed a tendency towards increased PFS in Kaplan Meier analysis. There was no such trend noted in patients receiving radiotherapy. These results are consistent with previous findings on immunotherapy in advanced NSCLC56,57. Possible explanations for these observations are decreased immunoreactivity thus decreasing antitumoral immune activity and decreased endocrine function of muscle tissue in sarcopenia promoting dysregulation of immunoresponse58,59. To our knowledge our results are the first from patients receiving surgical treatment for brain metastasis considering response to consecutive immunotherapy.
Considering our observation that sarcopenia but not frailty predicted survival in our cohort a certain conflict with established literature is to be addressed. Available data suggest that both, frailty and sarcopenia are risk factors of decreased progression-free and overall survival in patients suffering brain metastases irrespective of primary tumor type24,42,55. Possible explanations for these results include the relatively young median age in our cohort and comparatively low rates of frailty in comparison to other studies evaluating patients suffering brain metastasis24,42. This might be a reflection of potential selection by involved physicians or could potentially be based in some degree of subjectivity in the applied screening tools for frailty49. Another reason may be the prevalence of age-related treatment bias, which makes frailty analysis susceptible to misinterpretation. We believe that this issue should be emphasized in the highlighting of recent studies, as frailty is now increasingly recognized in clinical routine.
Reflecting the currently available data on frailty and sarcopenia in conjunction with the presented findings, sarcopenia is an independent predictor of survival in patients suffering from brain metastases of NSCLC. While we did not observe a significant influence of frailty, the available literature supports a relevant influence of frailty on survival in afflicted patients.
Conclusion
Sarcopenia is an independent predictor of OS and predicts response to immunotherapy better than frailty or age. Additionally, sarcopenia impedes postoperative physical performance and increases complication rates. Sarcopenia should therefore be accounted for in therapeutic and surgical decision making to improve patient selection for different treatment modalities.
Data Availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 Countries. CA: A cancer. J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
Myall, N. J., Yu, H., Soltys, S. G., Wakelee, H. A. & Pollom, E. Management of brain metastases in lung cancer: evolving roles for radiation and systemic treatment in the era of targeted and immune therapies. Neurooncol Adv. 3, v52–v62. https://doi.org/10.1093/noajnl/vdab106 (2021).
Barnholtz-Sloan, J. S. et al. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the metropolitan Detroit cancer surveillance system. J. Clin. Oncol. 22, 2865–2872. https://doi.org/10.1200/jco.2004.12.149 (2004).
Schouten, L. J., Rutten, J., Huveneers, H. A. M. & Twijnstra, A. Incidence of brain metastases in a cohort of patients with carcinoma of the breast, colon, kidney, and lung and melanoma. Cancer 94, 2698–2705. https://doi.org/10.1002/cncr.10541 (2002).
Thandra, K. C., Barsouk, A., Saginala, K., Aluru, J. S. & Barsouk, A. Epidemiology of lung cancer. Contemp. Oncol. (Pozn). 25, 45–52. https://doi.org/10.5114/wo.2021.103829 (2021).
Proietti, M., Cesari, M. & Frailty What is it? Adv. Exp. Med. Biol. 1216, 1–7. https://doi.org/10.1007/978-3-030-33330-0_1 (2020).
Sternberg, S. A., Schwartz, A. W., Karunananthan, S. & Bergman, H. Mark Clarfield, A. The identification of frailty: A systematic literature review. J. Am. Geriatr. Soc. 59, 2129–2138. https://doi.org/10.1111/j.1532-5415.2011.03597.x (2011).
Handforth, C. et al. The prevalence and outcomes of frailty in older cancer patients: a systematic review. Ann. Oncol. 26, 1091–1101. https://doi.org/10.1093/annonc/mdu540 (2015).
Kojima, G., Liljas, A. E. M. & Iliffe, S. Frailty syndrome: implications and challenges for health care policy. Risk Manag Healthc. Policy. 12, 23–30. https://doi.org/10.2147/rmhp.S168750 (2019).
Ruiz, J. et al. Frailty assessment predicts toxicity during first cycle chemotherapy for advanced lung cancer regardless of chronologic age. J. Geriatric Oncol. 10, 48–54. https://doi.org/10.1016/j.jgo.2018.06.007 (2019).
Krenzlin, H. et al. Frailty in glioblastoma is independent from chronological age. Front. Neurol. 12, 777120. https://doi.org/10.3389/fneur.2021.777120 (2021).
Olson, E. et al. Impact of age and frailty markers on overall survival among hospitalized patients with lung cancer treated with immunotherapy. J. Clin. Oncol. 39, e21151–e21151. https://doi.org/10.1200/JCO.2021.39.15_suppl.e21151 (2021).
Komici, K. et al. Frailty in patients with lung cancer: A systematic review and Meta-Analysis. Chest 162, 485–497. https://doi.org/10.1016/j.chest.2022.02.027 (2022). https://doi.org/https://doi.org/
Franco, I. et al. Use of frailty to predict survival in elderly patients with early stage non-small-cell lung cancer treated with stereotactic body radiation therapy. J. Geriatr. Oncol. 9, 130–137. https://doi.org/10.1016/j.jgo.2017.09.002 (2018).
Clough-Gorr, K. M., Thwin, S. S., Stuck, A. E. & Silliman, R. A. Examining five- and ten-year survival in older women with breast cancer using cancer-specific geriatric assessment. Eur. J. Cancer. 48, 805–812. https://doi.org/10.1016/j.ejca.2011.06.016 (2012).
Lu, J. et al. The preoperative frailty versus Inflammation-Based prognostic score: which is better as an objective predictor for gastric cancer patients 80 years and older? Ann. Surg. Oncol. 24, 754–762. https://doi.org/10.1245/s10434-016-5656-7 (2017).
Adams, P. et al. Frailty as a predictor of morbidity and mortality in inpatient head and neck surgery. JAMA Otolaryngol. Head Neck Surg. 139, 783–789. https://doi.org/10.1001/jamaoto.2013.3969 (2013).
Chappidi, M. R. et al. Frailty as a marker of adverse outcomes in patients with bladder cancer undergoing radical cystectomy. Urol. Oncol. 34, 256e251–256e256. https://doi.org/10.1016/j.urolonc.2015.12.010 (2016).
Mogal, H. et al. Modified frailty index predicts morbidity and mortality after pancreaticoduodenectomy. Ann. Surg. Oncol. 24, 1714–1721. https://doi.org/10.1245/s10434-016-5715-0 (2017).
Cruz-Jentoft, A. J. et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 48, 16–31. https://doi.org/10.1093/ageing/afy169 (2019).
Ranganathan, K. et al. Temporalis muscle morphomics: the Psoas of the craniofacial skeleton. J. Surg. Res. 186, 246–252. https://doi.org/10.1016/j.jss.2013.07.059 (2014).
Leitner, J. et al. High correlation of Temporal muscle thickness with lumbar skeletal muscle cross-sectional area in patients with brain metastases. PloS One. 13, e0207849. https://doi.org/10.1371/journal.pone.0207849 (2018).
Zhang, X. M. et al. Sarcopenia as a predictor of mortality in women with breast cancer: a meta-analysis and systematic review. BMC Cancer. 20, 172. https://doi.org/10.1186/s12885-020-6645-6 (2020).
Ilic, I. et al. Combined assessment of preoperative frailty and sarcopenia allows the prediction of overall survival in patients with lung cancer (NSCLC) and surgically treated brain metastasis. Cancers 13, 3353 (2021).
Trejo-Avila, M., Bozada-Gutiérrez, K., Valenzuela-Salazar, C., Herrera-Esquivel, J. & Moreno-Portillo, M. Sarcopenia predicts worse postoperative outcomes and decreased survival rates in patients with colorectal cancer: a systematic review and meta-analysis. Int. J. Colorectal Dis. 36, 1077–1096. https://doi.org/10.1007/s00384-021-03839-4 (2021).
Furtner, J. et al. Survival prediction using Temporal muscle thickness measurements on cranial magnetic resonance images in patients with newly diagnosed brain metastases. Eur. Radiol. 27, 3167–3173. https://doi.org/10.1007/s00330-016-4707-6 (2017).
Yang, Y. W. et al. Prognostic value of Temporal muscle thickness, a novel radiographic marker of sarcopenia, in patients with brain tumor: A systematic review and meta-analysis. Nutrition 112, 112077. https://doi.org/10.1016/j.nut.2023.112077 (2023).
Shachar, S. S., Williams, G. R., Muss, H. B. & Nishijima, T. F. Prognostic value of sarcopenia in adults with solid tumours: A meta-analysis and systematic review. Eur. J. Cancer. 57, 58–67. https://doi.org/10.1016/j.ejca.2015.12.030 (2016).
Gomes, F. et al. (2020).
Sakakida, T. et al. Safety and tolerability of PD-1/PD-L1 inhibitors in elderly and frail patients with advanced malignancies. Oncol. Lett. 20, 14. https://doi.org/10.3892/ol.2020.11875 (2020).
Montrone, M. et al. Immunotherapy in elderly patients affected by Non-Small cell lung cancer: A narrative review. J. Clin. Med. 12 https://doi.org/10.3390/jcm12051833 (2023).
Dai, S., Yang, M., Song, J., Dai, S. & Wu, J. Impacts of frailty on prognosis in lung cancer patients: A systematic review and Meta-Analysis. Front. Med. 8 https://doi.org/10.3389/fmed.2021.715513 (2021).
Jiménez Galán, R., Prado-Mel, E., Alvarez de Sotomayor, M. & Martin, L. A. K. Impact of frailty on outcomes of First-Line pembrolizumab monotherapy in a Real-World population with advanced Non-Small cell lung cancer. Biology 12, 191 (2023).
Mor, V., Laliberte, L., Morris, J. N. & Wiemann, M. The Karnofsky performance status Scale. An examination of its reliability and validity in a research setting. Cancer 53, 2002–2007. https://doi.org/10.1002/1097-0142(19840501)53:9%3C2002::aid-cncr2820530933%3E3.0.co;2-w (1984).
Rockwood, K. et al. A global clinical measure of fitness and frailty in elderly people. Cmaj 173, 489–495. https://doi.org/10.1503/cmaj.050051 (2005).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J. Chronic Dis. 40, 373–383. https://doi.org/10.1016/0021-9681(87)90171-8 (1987).
Onodera, H. et al. High protein intake after subarachnoid hemorrhage improves oral intake and Temporal muscle volume. Clin. Nutr. 40, 4187–4191. https://doi.org/10.1016/j.clnu.2021.01.040 (2021).
Rostoft, S., O’Donovan, A., Soubeyran, P., Alibhai, S. M. H. & Hamaker, M. E. Geriatric assessment and management in cancer. J. Clin. Oncol. 39, 2058–2067. https://doi.org/10.1200/JCO.21.00089 (2021).
Williams, G. R., Dunne, R. F., Giri, S., Shachar, S. S. & Caan, B. J. Sarcopenia in the older adult with cancer. J. Clin. Oncol. 39, 2068–2078. https://doi.org/10.1200/jco.21.00102 (2021).
Williams, G. R. et al. Frailty and skeletal muscle in older adults with cancer. J. Geriatr. Oncol. 9, 68–73. https://doi.org/10.1016/j.jgo.2017.08.002 (2018).
Shachar, S. S. et al. The association between geriatric assessment, muscle measures, and treatment-related toxicity in older adults with cancer: an Israeli prospective study. J. Geriatric Oncol. 13, 1203–1207. https://doi.org/10.1016/j.jgo.2022.08.007 (2022).
Lim, M. J. R. et al. Effect of sarcopenia and frailty on outcomes among patients with brain metastases. J. Neurooncol. 167, 169–180. https://doi.org/10.1007/s11060-023-04542-w (2024).
Nayak, L., Lee, E. Q. & Wen, P. Y. Epidemiology of brain metastases. Curr. Oncol. Rep. 14, 48–54. https://doi.org/10.1007/s11912-011-0203-y (2012).
Zhai, C. et al. Association of frailty with mortality in cancer survivors: results from NHANES 1999–2018. Sci. Rep. 14, 1619. https://doi.org/10.1038/s41598-023-50019-1 (2024).
Ostrom, Q. T., Wright, C. H. & Barnholtz-Sloan, J. S. Brain metastases: epidemiology. Handb. Clin. Neurol. 149, 27–42. https://doi.org/10.1016/b978-0-12-811161-1.00002-5 (2018).
Howlett, S. E., Rutenberg, A. D. & Rockwood, K. The degree of frailty as a translational measure of health in aging. Nat. Aging. 1, 651–665. https://doi.org/10.1038/s43587-021-00099-3 (2021).
Schneider, M. et al. Newly diagnosed glioblastoma in geriatric (65 +) patients: impact of patients frailty, comorbidity burden and obesity on overall survival. J. Neurooncol. 149, 421–427. https://doi.org/10.1007/s11060-020-03625-2 (2020).
Martin, F. C. & O’Halloran, A. M. Tools for assessing frailty in older people: general concepts. Adv. Exp. Med. Biol. 1216, 9–19. https://doi.org/10.1007/978-3-030-33330-0_2 (2020).
Mizuguchi, Y. et al. Machine learning-based gait analysis to predict clinical frailty scale in elderly patients with heart failure. Eur. Heart J. Digit. Health. 5, 152–162. https://doi.org/10.1093/ehjdh/ztad082 (2024).
Beaudart, C. et al. Sarcopenia in daily practice: assessment and management. BMC Geriatr. 16, 170. https://doi.org/10.1186/s12877-016-0349-4 (2016).
Mohile, S. G. et al. Association of a cancer diagnosis with vulnerability and frailty in older medicare beneficiaries. J. Natl. Cancer Inst. 101, 1206–1215. https://doi.org/10.1093/jnci/djp239 (2009).
Pesonen, E. K. et al. Age- and sex-adjusted CT-based reference values for Temporal muscle thickness, cross-sectional area and radiodensity. Sci. Rep. 15, 2393. https://doi.org/10.1038/s41598-025-86711-7 (2025).
Kofler, M. et al. The loss of Temporal muscle volume is associated with poor outcome in patients with subarachnoid hemorrhage: an observational cohort study. Neurocrit Care. 39, 198–206. https://doi.org/10.1007/s12028-023-01751-z (2023).
Luo, L. et al. Sarcopenia as a risk factor of progression-free survival in patients with metastases: a systematic review and meta-analysis. BMC Cancer. 23, 127. https://doi.org/10.1186/s12885-023-10582-2 (2023).
Kim, Y. I. et al. Association between Temporal muscle thickness and overall survival in Non-Small cell lung cancer patients with brain metastasis. Curr. Oncol. 29, 6463–6471. https://doi.org/10.3390/curroncol29090508 (2022).
Roch, B. et al. Cachexia - sarcopenia as a determinant of disease control rate and survival in non-small lung cancer patients receiving immune-checkpoint inhibitors. Lung Cancer. 143, 19–26. https://doi.org/10.1016/j.lungcan.2020.03.003 (2020).
Wang, J., Cao, L. & Xu, S. Sarcopenia affects clinical efficacy of immune checkpoint inhibitors in non-small cell lung cancer patients: A systematic review and meta-analysis. Int. Immunopharmacol. 88, 106907. https://doi.org/10.1016/j.intimp.2020.106907 (2020).
Cortellini, A. et al. Predictive value of skeletal muscle mass for immunotherapy with nivolumab in non-small cell lung cancer patients: A hypothesis-generator preliminary report. Thorac. Cancer. 10, 347–351. https://doi.org/10.1111/1759-7714.12965 (2019).
Afzali, A. M., Müntefering, T., Wiendl, H., Meuth, S. G. & Ruck, T. Skeletal muscle cells actively shape (auto)immune responses. Autoimmun. Rev. 17, 518–529. https://doi.org/10.1016/j.autrev.2017.12.005 (2018).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
LS collected and analyzed the data and wrote the manuscript. HK designed the study, analyzed the data and wrote the manuscript. AS collected and analyzed the data. DJ and AD analyzed the data and wrote the manuscript. CS, MB and FR helped to design the study and wrote the manuscript. NK designed the study, analyzed the data and helped to write the manuscript. Sections of the presented data are part of the doctoral thesis of AS.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schmidt, L., Krenzlin, H., Schmitz, A. et al. Association of sarcopenia with survival and treatment response in brain metastasis of non-small cell lung cancer. Sci Rep 16, 5261 (2026). https://doi.org/10.1038/s41598-026-37138-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37138-1