Abstract
Cuffed endotracheal tubes (ETTs) are widely used in general anesthesia, with recommended cuff pressure (CP) between 20 and 30 cmH2O in adults. This multi-center study investigated actual CP management practices across 19 Grade A tertiary hospitals in China, involving 2058 anesthetized patients. The median initial CP measured within 30 min post-intubation was 48 cmH2O (IQR 32–70), with only 19% within the recommended range (75.4% > 30 cmH2O; 5.6% < 20 cmH2O). All institutions demonstrated similarly suboptimal CP management. Four distinct methods were used for cuff pressure estimation: pilot balloon palpation (96.2%), fixed-volume inflation (2.8%), minimal occlusive volume technique (0.7%), and direct manometry (0.2%). Among them, only direct manometry demonstrated reliable accuracy in achieving appropriate pressures. The remaining three methods showed no clear superiority over one another in terms of pressure assessment accuracy. In orotracheally intubated patients with palpation-estimated CP, independent risk factors for elevated pressure (> 30 cmH2O) included: age < 60 years, pressure-controlled ventilation, absence of PEEP, ETT internal diameter < 7.0 mm, aminosteroid neuromuscular blocker use, and trainee-performed inflation. These findings highlight widespread challenges in optimal CP management across Chinese tertiary care centers.
Similar content being viewed by others
Introduction
Endotracheal intubation is routinely performed for patients undergoing general anesthesia. Following intubation, the endotracheal tube (ETT) cuff is immediately inflated to create an effective seal, preventing both air leakage and aspiration of oropharyngeal secretions or gastric contents. Inadequate cuff inflation increases the risk of both overt and silent aspiration, potentially leading to postoperative pulmonary complications1. Conversely, excessive cuff inflation compromises tracheal mucosal perfusion, resulting in ischemic injury2,3. Prolonged exposure to elevated cuff pressures may result in serious complications including mucosal ulceration, necrosis, tracheoesophageal fistula formation, and even tracheal rupture4,5. Even short-term exposure to excessive cuff pressures can induce postoperative respiratory sequelae, including cough, sore throat, hoarseness, and hemoptysis6. Therefore, it is widely recommended to maintain ETT cuff pressure between 20 and 30 cmH2O in adults with regular monitoring6,7. Despite these recommendations, clinical practice often relies on subjective estimation rather than objective measurement of cuff pressure. A few articles have documented the deviations from recommended cuff pressure ranges in operating room settings5,8,9. However, to date, no nationwide study has systematically evaluated the prevalence and potential clinical implications of this phenomenon. Multiple intraoperative factors may alter cuff pressure, including gas diffusion (particularly with N2O use), positional changes, iatrogenic pneumothorax or pneumoperitoneum, transesophageal echocardiography probe insertion, and direct surgical manipulation10,11,12,13,14. Nevertheless, the initial inflation pressure remains the primary determinant of cuff pressure maintenance throughout the surgical procedure.
This multicenter study was designed to: (1) quantify initial ETT cuff pressures in anesthetized patients across tertiary care centers in China; (2) evaluate current practice patterns for cuff inflation and pressure assessment; and (3) identify risk factors associated with non-compliant cuff pressures.
Methods
This prospective, multicenter observational study was conducted between April 2019 and February 2021 across 19 tertiary care centers in major Chinese cities. All participating institutions were classified as grade A tertiary hospitals, representing the highest level of medical care in China. The study protocol received ethical approval from the Institutional Review Board of the leading center (Approval No. KE 2019010027) and was registered with the Chinese Clinical Trial Registry on March 21, 2019 (registration number: ChiCTR1900022038). In accordance with the approved protocol, written informed consent was obtained from all individual participants at each participating center. The study was conducted in full compliance with the ethical principles of the Declaration of Helsinki.
Study population
This multicenter, cross-sectional study employed a stratified sampling approach to ensure geographic representation across Eastern, Central, and Western China. Nineteen Grade A tertiary hospitals were selected based on three key criteria: (1) geographic distribution, (2) volume of general anesthesia procedures, and (3) standardized anesthesia training programs.
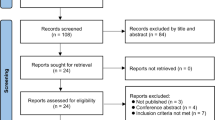
Figure 1 illustrates the patient enrollment process. Inclusion criteria comprised: (1) age ≥ 18 years, (2) scheduled for elective surgery under general anesthesia with endotracheal intubation. (3) provision of written informed consent prior to anesthesia administration. Exclusion criteria included: (1) inability to obtain cuff pressure (CP) measurements within 30 min post-intubation; (2) nitrous oxide was used before measurement; (3) non-supine positioning, significant head/neck position changes, or surgical creation of pneumothorax/pneumoperitoneum; or (4) clinical instability precluding safe CP measurement (e.g., hemodynamic or respiratory compromise requiring immediate intervention).
Flow-chart of participants’ enrollment.
Our pilot data revealed a decrease in cuff pressures for consecutive cases within the same operating room. This Hawthorne effect likely resulted from unconscious behavioral modification by anesthesia providers following initial pressure measurements. To maintain ecological validity, we implemented rigorous controls: (1) single-case enrollment per operating room per study day, (2) mandatory 5-day washout periods between study sessions, (3) complete blinding of clinical teams to measurement results, and (4) non-disclosure of study protocols until data collection concluded. These measures preserved natural clinical practice patterns while minimizing measurement bias.
Cuff pressure measurement and adjustment
The endotracheal tube (ETT) position was first verified by the operating room anesthesiologists to ensure proper placement depth. Cuff pressure (CP) was measured using a calibrated manometer (Manometer Sensitive, VBM Medizintechnik GmbH, Germany; range: 0−120 cmH2O) connected to the ETT’s pilot balloon. The manometer was directly attached to the pilot balloon valve, and baseline pressure was recorded after stabilization for at least 3 s. Pressure adjustments were made by inflating or deflating the cuff through the manometer system.
A small air leak consistently occurred during manometer disconnection. To account for this expected pressure loss, the target CP was set at 30 cmH₂O during measurement to ensure the final pressure remained within the recommended 20–30 cmH₂O range after disconnection. If air leakage was detected either by audible gas escape or discrepancy between set and delivered tidal volumes, the cuff was re-inflated through the manometer under the continuous manometric monitoring until the leak was just sealed. The pressure was then briefly increased by approximately 3 cmH₂O to compensate for the small pressure drop upon disconnecting the manometer, ensuring the final in vivo pressure remained within or near the recommended range.
Outcomes
The primary outcome was endotracheal tube cuff pressure (CP). Secondary outcomes included CP distribution and factors associated with CP > 30 cmH2O. Data were collected from three sources: patient records (age, sex, height, weight, surgical site), anesthesia parameters (Ppeak, PEEP, ventilation mode, anesthetics, ETT details), and inflating operator characteristics (age, sex, experience). All data were recorded on case report forms before entry into a structured Excel database for analysis. Variable selection was based on previous publications and clinical relevance.
Sample size estimation
The sample size calculation was based on a previously reported standard deviation of 31.8 cmH2O for endotracheal tube cuff pressures (CP), derived from prior research5. With an alpha level set at 0.05 and a margin of error of 1.17 cmH2O (representing 5% of the standard deviation), the initial sample size requirement was determined. To account for potential 20% data loss or exclusions, the final target sample size was established at 1,844 participants.
Statistical analysis
Missing data handling is detailed in Table S1. CPs were classified as: low (< 20 cmH2O), appropriate (20–30 cmH2O), or high (> 30 cmH2O). For factor analysis, participants were stratified into high CP (> 30 cmH2O) and non-high CP (≤ 30 cmH2O) groups, as high CP was the most frequently observed deviation while low CP cases were relatively rare.
Continuous variables were assessed for normality using the Kolmogorov-Smirnov test. Normally distributed data were expressed as mean ± SD and analyzed with Student’s t-test; non-normal data were reported as median (IQR) and compared using Mann-Whitney U tests. Categorical variables were presented as frequencies and analyzed with Pearson’s χ2 tests.
Multivariable logistic regression incorporated variables with clinical relevance and univariate p < 0.05. All analysis used SPSS 21.0 (IBM Corp.), with statistical significance set at p < 0.05 (two-tailed).
Conference presentation
The work was partially presented at the 28th Annual Meeting of the Chinese Society of Anesthesiology, which occurred in Changsha from September 21st to 24th, 2023.
Results
The ETT cuff pressures varied widely and frequently exceeded the recommended range
From April 2019 to February 2021, 2058 adult participants were enrolled across 19 hospitals (939 male and 1119 female, age 53 year [42–63]; height 162.6 cm [158.0–170.0], weight 62 kg [55.00–70]; BMI 23.43 kg/m2 [21.22–25.71]). Body mass index was categorized for analysis according to Chinese-specific criteria: normal weight (< 24 kg/m²), overweight (24–27.9 kg/m²), and obesity (≥ 28 kg/m²)15. All participants were intubated with high-volume, low-pressure (HVLP) cuffed endotracheal tubes (internal diameters: 6.0–8.0 mm), which were either reinforced or non-reinforced. The measured cuff pressures showed remarkable variation, ranging from 6 to 120 cmH2O (with 77 measurements reaching the barometer’s upper limit recorded as 120 cmH2O) (Fig. 2). The median CP was 48 cmH2O (IQR 32–70 cmH2O), significantly higher than recommended levels. Strikingly, only 19% of measurements fell within the optimal 20–30 cmH2O range, while 75.5% exceeded 30 cmH2O and just 5.5% were below 20 cmH2O. This pattern of excessive cuff inflation was observed consistently across all participating hospitals, though with some variation between institutions (Table 1).
The cuff pressures distribution according to the pressure gradient.
Suboptimal cuff pressure management practices in clinical settings
All illustrated in Table 2, the assessment of cuff inflation methods revealed that pilot balloon palpation was used in 96.2% of cases (1980/2058), while fixed-volume inflation (2–10 mL) was employed in 2.8% (58/2058), minimal occlusive volume technique in 0.7% (15/2058), and direct manometric measurement in only 0.2% of cases (5/2058). Among all methods evaluated, only direct pressure measurement consistently achieved proper cuff inflation pressures within the recommended 20–30 cmH₂O range. The remaining techniques failed to ensure appropriate pressure maintenance or demonstrate superior performance relative to other approaches, as evidenced by the widespread overinflation documented in our findings. This pattern highlights a critical reliance on subjective assessment methods in clinical practice, with minimal adoption of evidence-based pressure measurement techniques, underscoring the need for standardized cuff management protocols.
Factors associated with high ETT cuff pressure
Among the 2058 participants, 1961 cases underwent orotracheal intubation with cuff pressure estimated by pilot balloon palpation and had classifiable surgical sites, forming the primary analysis cohort. Potential risk factors for cuff pressures exceeding 30 cmH2O were examined in this group. Initial comparisons revealed significant differences between patients with cuff pressures ≤ 30 cmH2O and > 30 cmH2O across several variables, including patient age, ventilation mode, PEEP application, endotracheal tube internal diameter, muscle relaxant use, inflating operator experience level, and surgical site location, as detailed in Table 3.
Multivariate analysis incorporated these significant factors along with additional covariates: patient sex, body mass index, peak airway pressure, endotracheal tube type (reinforced versus unreinforced), and inflating operator sex. This comprehensive adjustment identified six independent risk factors of elevated cuff pressure: patient age below 60 years, pressure-controlled ventilation mode, absence of PEEP during ventilation, administration of aminosteroid neuromuscular blocking agents, use of endotracheal tubes with internal diameter less than 7.0 mm, and cuff inflation performed by trainee anesthetists. These final adjusted associations are presented in Table 4, demonstrating the multifactorial nature of cuff overinflation in clinical practice. The findings highlight how patient characteristics, ventilation strategies, equipment selection, and inflating operator factors collectively influence cuff pressure management outcomes.
Discussion
The cuffed ETT was first described by Eisenmenger in 1893 and has since evolve to meet the needs of surgery, anesthesia, and, more recently, critical care medicine16. The primary function of the cuff is to form a seal against the tracheal or bronchial walls, enabling positive pressure ventilation, delivery of inhalational drugs without atmospheric pollution, and prevention of secretions, fluids, and blood from the mouth or stomach from leaking into the trachea and lungs.
When the high-pressure low-volume (HPLV) cuffed ETT was widely used, excessive CP was already recognized as a significant issue. The introduction of high-volume, low-pressure (HVLP) cuffed ETTs in the 1970 s marked a significant advancement, and these have since become the standard in clinical practice16. However, the low pressure in the pilot balloon can lead to over-inflation17. In 1984, R D Seegobin directly observed mucosal blood flow in intubated patients using fiberoptic bronchoscopes and reported that the mucosal blood flow was impaired at CPs above 30 cmH2O and completely obstructed at 50 cm H2O or higher3. This finding established 30 cmH2O as the widely accepted upper limit for appropriate CP. However, a recent study had reported airway mucosal damage in patients undergoing long-term ventilation even when CPs were maintained at 30 cmH2O, suggesting that a lower safety threshold for CP may be necessary for such patients.
In contrast, the lower limit of CP for HVLP ETTs is much more controversial. Park et al. suggested that a CP below 20 cmH2O was sufficient for tracheal occlusion based on auscultation or spirometry18. However, recent studies have challenged this view, indicating that a significant number of patients require CPs outside the recommended range (< 20 or > 30 mmH2O) to have adequate airway sealing, the required CP is influenced by the difference between the ETT cuff area and airway area18. Other studies have focused on the prevention of microaspiration. Rello et al. identified a CP < 20 cmH2O during the first eight days of intubation as an independent risk factor for ventilator-associated pneumonia (VAP)19. Additionally, research has shown that conventional HVLP cuffs may not prevent microaspiration even at CPs up to 60 cmH2O20. Seegobin’ s direct observations revealed that cuff folds, which may allow leakage of oral or gastric contents, persist even at CPs as high as 100 cm H2O3.
Recent advancements in ETT design have also introduced variability in the lowest CP required to prevent microaspiration, depending on cuff shape, material, and thickness20,21. Despite these controversies, many institutions use 20 cmH2O as the lower limit for acceptable CP, with the range of 20–30 cmH2O widely recommended for adult patients. This range balances the prevention of mucosal injury and aspiration risk, though further research is needed to optimize cuff pressure management in clinical practice7.
Liu et al. previously measured the CP of 236 patients immediately after intubation in four tertiary care university hospitals in Shanghai in 20105, reporting a mean ETT CP of 43 ± 23.3 mmHg following estimation by pilot balloon palpation. A decade later, we conducted this study across 19 large, urban, comprehensive Grade A tertiary hospitals in China, where general anesthesia with intubation is routinely performed. Despite the anesthetic staff being well-trained and regularly updated on best practices, the findings were concerning: only a small proportion of CP measurements fell within the recommended range. Over-inflation was both prevalent and severe, with proper inflation rates varying widely among hospitals, ranging from as low as 0.9% to as high as 39%.
In our study, cuff inflation was performed using one of the following methods: pilot balloon palpation (96.2% of cases), fixed volume inflation (2–10 mL) (2.8%), minimal occlusive volume (MOV) (0.7%), Although measurement using instrumentation (e.g., a manometer) has been recommended for decades22, its routine use has been limited by additional costs, increased workload, and concerns regarding calibration and sanitization. Despite accumulating evidence, including our findings, highlighting its unreliability, pilot balloon palpation remains the most widely used method for cuff inflation in daily anesthetic practice worldwide. Some studies have suggested that minimal occlusive volume (MOV) is the most appropriate technique to prevent both over- and under-inflation. However, our investigation did not demonstrate its superiority over pilot balloon palpation or fixed volume inflation. Additionally, a study reported that over-inflation is common with MOV due to the difficulty in determining when to stop inflating the cuff, often resulting in excessive pressure23.
We also identified several factors associated with Cuff Pressure Exceeding 30 cmH2O. Both in vitro and in vivo studies in pediatric patients have shown that higher CPs are associated with smaller tube sizes, likely due to the inability to achieve an effective tracheal seal with a small air volume in the cuff, necessitating additional inflation24. Recent research in adult patients has further indicated that smaller tube sizes (e.g., ID 6.0 vs. 8.0) may increase the risk of inaccurate CP estimation by pilot balloon palpation25.These findings may explain why participants using endotracheal tubes (ETTs) with smaller internal diameters in our study were more likely to exhibit excessive CP. Other factors, such as differences in cuff volume between ETT sizes, were not examined in this study and cannot be ruled out as potential contributors to the observed results.
We also found that elderly patients had lower CPs, a finding consistent with a prior study in critically ill adults. The researchers attributed this to glottic muscle atrophy with age. Studies on tracheal changes in adults have demonstrated that aging is associated with increases in the tracheal anterior-posterior diameter, area, roundness distortion, and calcification scores26,27. These factors may change the shape of cuff in trachea or reduce the palpable softness during inflation, thus contributing to the lower cuff pressures in elderly participants comprehensively.
Prior studies indicated no correlation between inflating operator and cuff pressures8,28. This study, with a much larger sample size, revealed a correlation of professional experience of the inflating operator with the cuff pressures. Compared to trainees—including anesthetic residents, clinical interns, rotating doctors from other departments, nurse anesthetists, and doctors in advanced study—cuffs inflated by trainers, such as attending doctors, associate professors, and professors, were less prone to over-inflation. This better performance of trainers might be attributed to the training, clinical experience, and ongoing knowledge update8,29,30.
The effect of muscle relaxants on ETT CP are complex. Compared to the adductor pollicis, commonly used for neuromuscular blockade monitoring, neuromuscular blockade at the laryngeal muscles exhibits a faster onset, less intense, and more rapid recovery. Paralysis of the laryngeal muscle reduces the resting pressure of cuffs placed between the vocal cords31. It also diminishes or weakens swallowing and coughing, which induce CP fluctuation. In addition to blocking N2-acetylcholine receptors in skeletal muscles, muscle relaxants also bind to M2 and M3 muscarinic receptors in the airway. Their overall effect on airway constriction depends on the relative influence of antagonism of the prejunctional neural M2 muscarinic receptors, the postjunctional muscle M3 receptors, or allosteric potentiation of M3 receptors (e.g., rapacuronium)32,33,34. This could explain the discrepancies between the receptor affinities of various muscle relaxants tested in cells expressing either M2 or M3 receptors individually, and their effects on airway muscle tone in animal models and clinical settings32,33,34,35,36. Finally, histamine release by some muscle relaxants could also induce tracheobronchial spasms. The activity of smooth muscles in the proximally innervated airway may be reflected in ETT CP measurements37,38. Although this study observed that patients administered amino steroids muscle relaxants tended to have higher CPs than those given benzylisoquinolinium muscle relaxants, the lack of recorded dosages and precise time intervals between intubation and CP measurement complicates providing a clear explanation.
It is noteworthy that participants ventilated in Pressure-Controlled Ventilation (PCV) mode or without Positive End-Expiratory Pressure (PEEP) tended to exhibit higher CPs. Typically, both PCV mode and ventilation without PEEP are associated with lower peak airway pressures (Ppeak). An elevation in Ppeak, usually observed during laparoscopic surgeries, may induce increased CPs11,39. However, this was not the situation in our study, as the differences in Ppeak were minimal (14.0 cmH2O in PCV vs. 15.2 cmH2O in VCV mode; 15.5 cmH2O with PEEP vs. 15.0 cmH2O without PEEP), and no correlation between Ppeak and high CP was found (data not shown). Consequently, further studies were warranted to interpret these findings. Notably, for most ETTs, the incidence of micro-aspiration is inversely related to the PEEP level40,41. Therefore, ventilating with PEEP might be beneficial to reduce the risk of micro-aspiration and excessive CP.
Taken together, our findings highlight several key contributions. First, this multi-center study objectively quantifies a significant and persistent gap between evidence-based recommendation and clinical reality. While manometry remains the unequivocal gold standard for cuff pressure measurement, our findings reveal that manual palpation—a method shown here to be highly unreliable—is the dominant practice across diverse clinical settings. This discrepancy likely stems from non-mandatory guidelines, perceived practical barriers, and an underestimation of the impact of excessive pressure.
Crucially, this gap has demonstrable consequences. We observed a consistently high prevalence of excessive cuff pressure across all 19 participating sites, despite expected heterogeneity in patient demographics. This uniformity underscores that the issue is systemic and practice-driven. Furthermore, the risk factors we identified—such as cuff inflation by trainees and the use of specific ventilator modes—remained significant after adjusting for patient-level variables, confirming their independence from underlying case-mix variation and highlighting specific, modifiable targets for intervention.
This study has several limitations. Firstly, this study was conducted solely in high-level public hospitals in China, many of which are affiliated with medical schools or provincial medical centers. The situation in a large number of primary and secondary hospitals in China remains unknown. Secondly, we did not track postoperative complications, such as sore throat or hoarseness. This was a deliberate methodological choice: ethically, we were obliged to correct any unsafe pressure immediately, precluding a controlled comparison; scientifically, a single initial pressure is a poor proxy for the dynamic intraoperative exposure relevant to such outcomes. While establishing a direct causal link would strengthen the argument, our study provides the essential baseline data on the prevalence of the exposure (excessive pressure) that is known to cause harm. Thirdly, we did not examine the association between CPs and the material, shape, and thickness of ETT cuffs due to the wide variety of ETT types, there were 22 types of ETTs used across these institutions. Certain data were unavailable due to confidentiality agreements or because some products were discontinued.
In conclusion, ETT cuffs were frequently over-inflated in the operating room. The assessment of CP was often inaccurate, necessitating direct measurement using instrumentation. Our findings underscore that excessive cuff pressure is a multifactorial, systemic issue. They provide a compelling evidence base for implementing targeted interventions—such as mandatory manometric monitoring and structured training for trainees—to improve patient safety.
Data availability
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request.
References
Maertens, B. et al. Effectiveness of continuous cuff pressure control in preventing Ventilator-Associated pneumonia: A systematic review and Meta-Analysis of randomized controlled Trials*. Crit. Care Med. 50, 1430–1439. https://doi.org/10.1097/ccm.0000000000005630 (2022).
Touat, L. et al. Intubation-related tracheal ischemic lesions: incidence, risk factors, and outcome. Intensive Care Med. 39, 575–582. https://doi.org/10.1007/s00134-012-2750-6 (2013).
Seegobin, R. D. & van Hasselt, G. L. Endotracheal cuff pressure and tracheal mucosal blood flow: endoscopic study of effects of four large volume cuffs. Br. Med. J. (Clin. Res. Ed.). 288, 965–968. https://doi.org/10.1136/bmj.288.6422.965 (1984).
Cooper, J. D. Tracheal injuries complicating prolonged intubation and tracheostomy. Torac. Surg. Clin. 28, 139–. https://doi.org/10.1016/j.thorsurg.2018.01.001 (2018).
Liu, J. et al. Correlations between controlled endotracheal tube cuff pressure and postprocedural complications: A multicenter study. Anesth. Analg. 111, 1133–1137. https://doi.org/10.1213/ANE.0b013e3181f2ecc7 (2010).
Klompas, M. et al. Strategies to prevent ventilator-associated pneumonia in acute care hospitals: 2014 update. Infect. Control Hosp. Epidemiol. 35 (Suppl 2), 133–154. https://doi.org/10.1017/s0899823x00193894 (2014).
Hung, W. C., Ko, J. C., Weil, A. B. & Weng, H. Y. Evaluation of Endotracheal Tube Cuff Pressure and the Use of Three Cuff Inflation Syringe Devices in Dogs. Front. Veterinary Sci. https://doi.org/10.3389/fvets.2020.00039 (2020).
Ozcan, A. T. D., Doger, C., But, A., Kutlu, I. & Aksoy, S. M. Comparison of endotracheal tube cuff pressure values before and after training seminar. J. Clin. Monit. Comput. 32, 527–531. https://doi.org/10.1007/s10877-017-0046-7 (2018).
Stevens, G. J., Warfel, J. W., Aden, J. K. & Blackwell, S. D. Intraoperative endotracheal cuff pressure study: how education and availability of manometers help guide safer pressures. Mil. Med. 183, E416–E419. https://doi.org/10.1093/milmed/usx127 (2018).
Kim, D. et al. The changes of endotracheal tube cuff pressure by the position changes from supine to prone and the flexion and extension of head. Korean J. Anesthesiology. 68, 27–31. https://doi.org/10.4097/kjae.2015.68.1.27 (2015).
Rosero, E. B., Ozayar, E., Eslava-Schmalbach, J., Minhajuddin, A. & Joshi, G. P. Effects of increasing airway pressures on the pressure of the endotracheal tube cuff during pelvic laparoscopic surgery. Anesth. Analg. 127, 120–125. https://doi.org/10.1213/ane.0000000000002657 (2018).
Kim, T. K. et al. Increased tracheal cuff pressure during insertion of a transoesophageal echocardiography probe A prospective, observational study. Eur. J. Anaesthesiol. 32, 549–554. https://doi.org/10.1097/eja.0000000000000204 (2015).
Tu, H. N. et al. Nitrous oxide increases endotracheal cuff pressure and the incidence of tracheal lesions in anesthetized. Anesth. Analg. 89, 187–190. https://doi.org/10.1097/00000539-199907000-00033 (1999).
Sperry, R. J., Johnson, J. O. & Apfelbaum, R. I. Endotracheal tube cuff pressure increases significantly during anterior cervical fusion with the Caspar instrumentation system. Anesth. Analg. 76, 1318–1321. https://doi.org/10.1213/00000539-199376060-00023 (1993).
Chen, C. & Lu, F. C. Department of disease control ministry of Health, P. R. C. The guidelines for prevention and control of overweight and obesity in Chinese adults. Biomed. Environ. Sci.: BES. 17 Suppl, 1–36 (2004).
Haas, C. F., Eakin, R. M., Konkle, M. A. & Blank, R. Endotracheal tubes: old and new. Respir. Care. 59, 933–952. https://doi.org/10.4187/respcare.02868 (2014).
Guyton, D., Banner, M. J. & Kirby, R. R. High-volume, low-pressure cuffs. Are they always low pressure? Chest 100, 1076–1081, (1991). https://doi.org/10.1378/chest.100.4.1076
Park, H. Y., Kim, M. & In, J. Does the minimal occlusive volume technique provide adequate endotracheal tube cuff pressure to prevent air leakage? A prospective, randomized, crossover clinical study. Anesth. Pain Med. 15, 365–370. https://doi.org/10.17085/apm.20020 (2020).
Rello, J. et al. Pneumonia in intubated patients: role of respiratory airway care. Am. J. Respir. Crit Care Med. 154, 111–115. https://doi.org/10.1164/ajrccm.154.1.8680665 (1996).
Dullenkopf, A., Gerber, A. & Weiss, M. Fluid leakage past tracheal tube cuffs: evaluation of the new microcuff endotracheal tube. Intensive Care Med. 29, 1849–1853. https://doi.org/10.1007/s00134-003-1933-6 (2003).
Monsel, A. et al. Tapered-cuff endotracheal tube does not prevent early postoperative pneumonia compared with Spherical-cuff endotracheal tube after major vascular surgery: A randomized controlled trial. Anesthesiology 124, 1041–1052. https://doi.org/10.1097/aln.0000000000001053 (2016).
Vu Quoc, D. et al. Effectiveness of continuous endotracheal cuff pressure control for the prevention of Ventilator-Associated respiratory infections: an Open-Label Randomized, controlled trial. Clin. Infect. Dis. 74, 1795–1803. https://doi.org/10.1093/cid/ciab724 (2022).
Bolzan, D. W. et al. Clinical use of the Volume-Time curve for endotracheal tube cuff management. Respir. Care. 59, 1628–1635. https://doi.org/10.4187/respcare.02683 (2014).
Krishna, S. G. et al. Cuffed endotracheal tubes in children: the effect of the size of the cuffed endotracheal tube on intracuff pressure. Pediatr. Anesth. 27, 494–500. https://doi.org/10.1111/pan.13099 (2017).
Ban, M. G. et al. Accuracy of pilot balloon palpation for cuff pressure assessment in small versus large sized tubes: a prospective non-randomized observational study. Sci. Rep. https://doi.org/10.1038/s41598-023-32704-3 (2023).
Li, S. et al. Factors Associated with Age-Related Changes in Non-Smoking Urban Men and Women in China Determined by Low-Dose Computed Tomography Imaging. Med. Sci. Monit. https://doi.org/10.12659/msm.931006 (2021).
Sakai, H. et al. Age-related changes in the trachea in healthy adults. Adv. Exp. Med. Biol. 662, 115–120. https://doi.org/10.1007/978-1-4419-1241-1_16 (2010).
Saracoglu, A., Dal, D., Pehlivan, G. & Gogus, F. Y. The professional experience of anaesthesiologists in proper inflation of laryngeal mask and endotracheal tube cuff. Turkish J. Anaesthesiol. Reanimation. 42, 234–238. https://doi.org/10.5152/tjar.2014.87487 (2014).
Maboudi, A., Abtahi, H., Hosseini, M., Tamadon, A. & Safavi, E. Accuracy of endotracheal tube cuff pressure adjustment by fingertip palpation after training of intensive care unit nurses. Iran. Red Crescent Med. J. 15, 381–384. https://doi.org/10.5812/ircmj.4164 (2013).
Murugiah, U. R. et al. Knowledge acquisition and retention among nurses after an educational intervention on endotracheal cuff pressure. Nurs. Crit. Care. 26, 363–371. https://doi.org/10.1111/nicc.12600 (2021).
Girling, K. J., Bedforth, N. M., Spendlove, J. L. & Mahajan, R. P. Assessing neuromuscular block at the larynx: the effect of change in resting cuff pressure and a comparison with video imaging in anesthetized humans. Anesth. Analg. 88, 426–431. https://doi.org/10.1097/00000539-199902000-00038 (1999).
Sunaga, H., Zhang, Y., Savarese, J. J. & Emala, C. W. Gantacurium and CW002 do not potentiate muscarinic Receptor-mediated airway smooth muscle constriction in Guinea pigs. Anesthesiology 112, 892–899. https://doi.org/10.1097/ALN.0b013e3181d32016 (2010).
Jooste, E., Zhang, Y. & Emala, C. W. Neuromuscular blocking agents’ differential bronchoconstrictive potential in Guinea airways. Anesthesiology 106, 763–772. https://doi.org/10.1097/01.anes.0000264763.48920.c9 (2007).
Hou, V. Y., Hirshman, C. A. & Emala, C. W. Neuromuscular relaxants as antagonists for M2 and M3 muscarinic receptors. Anesthesiology 88, 744–750. https://doi.org/10.1097/00000542-199803000-00026 (1998).
Milchert, M. et al. Skeletal muscle relaxants inhibit rat tracheal smooth muscle tone < i > In vitro. J. Physiol. Pharmacol. 60, 5–11 (2009).
Cembala, T. M., Sherwin, J. D., Tidmarsh, M. D., Appadu, B. L. & Lambert, D. G. Interaction of neuromuscular blocking drugs with Recombinant human m1-m5 muscarinic receptors expressed in Chinese hamster ovary cells. Br. J. Pharmacol. 125, 1088–1094. https://doi.org/10.1038/sj.bjp.0702166 (1998).
Nishino, T., Sugimori, K., Hiraga, K. & Honda, Y. Effects of tracheal irritation and hypercapnia on tracheal smooth muscle in humans. J. Appl. Physiol. (Bethesda Md. : 1985). 69, 419–423. https://doi.org/10.1152/jappl.1990.69.2.419 (1990).
Ishikawa, T., Sekizawa, S. & Sant’ Ambrogio, F. B. Sant’ Ambrogio, G. Endotracheal cuff pressure as an index of airway smooth muscle activity: comparison with total lung resistance. Respir. Physiol. 112, 175–184. https://doi.org/10.1016/s0034-5687(98)00029-2 (1998).
Sen, O. et al. Effects of pressure-controlled and volume-controlled ventilation on respiratory mechanics and systemic stress response during laparoscopic cholecystectomy. Springerplus 5 https://doi.org/10.1186/s40064-016-1963-5 (2016).
Pitts, R. et al. Variables affecting leakage past endotracheal tube cuffs: a bench study. Intensive Care Med. 36, 2066–2073. https://doi.org/10.1007/s00134-010-2048-5 (2010).
Zanella, A. et al. Fluid leakage across tracheal tube cuff, effect of different cuff material, shape, and positive expiratory pressure: a bench-top study. Intensive Care Med. 37, 343–347. https://doi.org/10.1007/s00134-010-2106-z (2011).
Funding
The work was supported by grants from the Natural Science Foundation of Hunan Province in China (No. 2019JJ50933) and the National Natural Science Foundation of China (No. 82101319).
Author information
Authors and Affiliations
Contributions
Study conception and design: YQW, ZHT, QLG ,Patient recruitment and Data collection: YQW, HQP, ZHT, YLL, KMD, HZ, LLB, YLZ, PW, JXT, YJH, XHZ, GC, JX, JQG, XZZ, JLS, YKS, MZ, SZ, HW, HMMData analysis: YQW, HQP, ZW, Manuscript preparation: YQW, HQP, Manuscript editing: YQW, HQP and WYZ, Study supervision: YQW, ZHT.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the medical ethics committee in every hospital and registered in the Chinese Clinical Trial Registry on March 21th, 2019 (The Investigation of tracheal tube cuff pressure in patients with tracheal intubation under general anesthesia, ChiCTR1900022038, https://www.chictr.org.cn/showproj.html?proj=36902).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Peng, H., Tang, Z., Li, Y. et al. The investigation of initial endotracheal tube cuff pressures in the operating room: a multi-center cross-sectional study in China. Sci Rep 16, 6856 (2026). https://doi.org/10.1038/s41598-026-37279-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37279-3