Abstract
Lanatoside C (LanaC), a cardiac glycoside, has been reported to possess therapeutic potential in acute intestinal inflammation; however, its in vivo effects on ulcerative colitis (UC) remain incompletely understood. In this study, we demonstrated that LanaC effectively attenuates DSS-induced colitis in mice by reducing inflammation, mitigating epithelial damage, and preserving barrier integrity. Mechanistically, LanaC treatment was associated with reduced macrophage infiltration in the colon and spleen, suppression of pro-inflammatory M1 macrophage markers, and enhancement of M2-associated markers. In vitro, LanaC inhibited LPS-induced M1 polarization and pro-inflammatory cytokine production in BMDMs, while promoting IL-4-driven M2 polarization and anti-inflammatory cytokine expression. These effects were accompanied by attenuation of STAT1/STAT3 signaling and enhancement of STAT6 activation, suggesting a selective reprogramming of macrophage responses. Collectively, these findings reveal that LanaC alleviates DSS-induced colitis, at least in part, through regulating macrophage infiltration and repolarization supporting its potential as a macrophage-targeted therapeutic candidate in UC.
Similar content being viewed by others
Introduction
Inflammatory bowel disease (IBD) encompasses a group of chronic inflammatory disorders of the gastrointestinal tract with ulcerative colitis (UC) and Crohn’s disease being the most prevalent forms1. UC is characterized by persistent inflammation and ulceration of the colonic mucosa, manifesting clinically as abdominal pain, diarrhea, and bloody stools2. The chronic course and frequent relapses substantially impair patients’ quality of life. The global prevalence of UC is steadily increasing, particularly in industrialized countries3. Current therapeutic strategies—including mesalazine, corticosteroids, immunosuppressants, and biologics—provide symptomatic relief but are constrained by limited efficacy and adverse effects associated with long-term use4,5,6,7. These challenges highlight the urgent need for safer and more effective therapeutic agents for UC management.
Macrophages play a central role in innate immunity, acting as key regulators of inflammation, tissue repair, and immune homeostasis8,9. Their remarkable functional diversity is largely governed by polarization, a dynamic process in which environmental cues, such as cytokines and microbial components, direct macrophages toward distinct activation states10,11. Classically, macrophages are categorized into pro-inflammatory M1 or anti-inflammatory M2 subtypes. M1 macrophages, typically induced by lipopolysaccharide (LPS) or interferon-γ (IFNγ), produce cytokines including interleukin-1β (IL-1β), IL-6, and tumor necrosis factor-α (TNFα), and express inducible nitric oxide synthase (iNOS). In contrast, M2 macrophages arise in response to IL-4, IL-10, IL-13, or transforming growth factor-β (TGFβ), secrete anti-inflammatory mediators including IL-10, and express markers such as Arginase-1 (Arg1) and CD20612,13. Beyond immune regulation, M2 macrophages also promote mucosal repair by driving extracellular matrix remodeling, angiogenesis, and epithelial regeneration14. Although the M1/M2 dichotomy is widely applied, macrophage polarization is now understood as a flexible spectrum shaped by the tissue microenvironment15.
In the intestinal mucosa, macrophages are indispensable for maintaining immune tolerance by discriminating between commensal antigens and pathogens. In UC, this balance is disrupted, resulting in excessive macrophage activation that is often skewed toward the M1 phenotype9,16,17. Patient-derived macrophages exhibit aberrant signaling and dysregulated effector functions, which perpetuate chronic inflammation and mucosal damage18. These include excessive production of reactive oxygen species (ROS), recruitment of neutrophils that release ROS and proteases, and infiltration of T lymphocytes into the lamina propria10,19,20. Together, these processes drive epithelial barrier disruption and characteristic histopathological features such as mucosal ulceration, crypt abscesses, and massive immune cell infiltration9. Given their pivotal role in both disease progression and tissue repair, therapeutic strategies aimed at modulating macrophage polarization—particularly restoring the M1/M2 balance—hold substantial promise in IBD treatment21.
Cardiac glycosides represent a family of naturally derived compounds defined by a conserved steroid nucleus that serves as the pharmacophore responsible for their biological activity22. These compounds primarily act through inhibition of Na⁺/K⁺-ATPase, thereby regulating cellular cation homeostasis23. Lanatoside C (LanaC), an FDA-approved cardiac glycoside, is traditionally used in the treatment of heart failure and arrhythmias24,25. Recent studies, however, have revealed that LanaC also possesses anti-inflammatory properties. For example, it has been shown to modulate neuroinflammation and to exert protective effects against pulmonary fibrosis by suppressing fibroblast proliferation and differentiation26. More recently, a research applied network pharmacology to explore the potential of LanaC in UC, revealing strong associations between LanaC target proteins and UC-related proteins, and this study further demonstrated that LanaC reduces the secretion of pro-inflammatory cytokines in the murine macrophage cell line RAW264.727. Notably, however, the in vivo effects of LanaC on UC have not been validated, leaving a critical gap in knowledge.
In the present study, we investigated the therapeutic efficacy of LanaC in a dextran sulfate sodium (DSS)-induced mouse model of colitis and explored the underlying mechanisms. Our results show that LanaC significantly alleviates mucosal inflammation and epithelial barrier injury. Moreover, we identify macrophages as a primary cellular target of LanaC and demonstrate its dual capacity to attenuate inflammatory responses by inhibiting M1 polarization while promoting M2 polarization.
Methods
Animals and DSS-induced colitis model conduction
Six-week-old male BALB/c mice were obtained from the Charles River Laboratories (Beijing, China). Animals were housed under specific pathogen-free conditions in a 12-h light/dark cycle with unrestricted access to standard food and sterile water for 7 days before experiment. Animals were then randomly assigned into three groups (n = 6 per group): a normal control (NC) group, a DSS group, and a DSS + LanaC group. Colitis was induced by administration of 3% (w/v) dextran sulfate sodium (DSS; 42867, Sigma-Aldrich) in drinking water for 7 consecutive days, followed by normal water for an additional 3 days21. Fresh DSS solution was prepared every two days. LanaC (HY-B1030, MedChemExpress) was administered intragastrically (i.g.) at a dose of 100 mg/kg once daily. Mice in the DSS groups received an equivalent volume of corn oil as vehicle control. Food intake and water consumption were monitored daily at fixed time points. At the end of the experiment, mice were anesthetized with isoflurane in an induction chamber containing 3% isoflurane in oxygen until complete loss of consciousness and cessation of respiratory movement were observed. Cervical dislocation was subsequently performed as a secondary method to ensure death prior to tissue collection. The entire colon was then excised, measured, and photographed, while spleens were harvested and weighed to record their wet mass. All animal procedures were approved by the Institutional Animal Ethics Committee of the Beijing University of Chinese Medicine in China (Ethics approval No. BUCM-1-2020102701-0011) and conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals issued by the Ministry of Science and Technology of China. This study adheres to the ARRIVE guidelines for the reporting of animal research.
Evaluation of disease activity index (DAI)
Disease activity was assessed daily during DSS administration using the disease activity index (DAI) as described by Wirtz et al28. The DAI was calculated as the mean of three parameters: body weight loss, stool consistency, and fecal blood. Scoring criteria were as follows: body weight loss (0, ≤ 1%; 1, 1–5%; 2, 5–10%; 3, 10–15%; 4, > 15%), stool consistency (0, normal; 2, loose stool; 4, watery diarrhea), and hematochezia (0, none; 2, moderate; 4, severe). The DAI was determined as the mean of the three individual scores.
Histopathological analysis
Colon tissues were fixed in 4% formaldehyde, embedded in paraffin, and sectioned at 5 μm thickness. Sections were stained with hematoxylin and eosin (H&E) (H-3502, Vector laboratory) and examined under a light microscope to evaluate histopathological changes.
Immunohistochemistry
Paraffin-embedded colon sections were deparaffinized, rehydrated, and treated with hydrogen peroxide to block endogenous peroxidase activity. Antigen retrieval was performed by microwave heating, followed by blocking with 5% bovine serum albumin (BSA) for 30 min. Sections were incubated overnight at 4 °C with primary antibodies against Claudin4 (1:200) (ab217685, abcam), Occludin (1:200) (ab242203, abcam), and ZO1 (1:300) (ab221547, abcam). After PBS washes, sections were incubated with polyperoxidase-conjugated anti-rabbit IgG (1:1000) (a9169, Sigma-Aldrich) for 1 h at room temperature. Nuclei were counterstained with hematoxylin, and images were captured by light microscopy. Images were quantified using ImageJ. Positive staining was identified using a uniform threshold applied to all images, and the positive area and integrated optical density (IOD) were quantified.
Periodic acid Schiff and Alcian blue staining
For immunohistochemistry, paraffin-embedded colon sections were stained with Periodic acid-Schiff and Alcian blue (AB-PAS) (BP-DL037, Sbjbio) and examined by light microscopy. Goblet cell numbers were quantified and normalized to crypt units29.
Real-time quantitative PCR (RT-PCR)
Total RNA was extracted from colon tissues and cultured cells using the Total RNA Isolation Kit (74104, QIAGEN). Reverse transcription was performed using the PrimeScript™ RT-PCR Kit (RR014B, Takara). RT-PCR was carried out on a StepOnePlus or QuantStudio 3 detection system (Applied Biosystems). Data were analyzed using the ΔΔCt method and normalized to Hypoxanthine Phosphoribosyltransferase (Hprt) genes. The primers sequences were used to amplify the indicated genes in this study are shown in Table 1.
Measurement of cytokines production
The cytokines production was assessed by using enzyme-linked immune sorbent assay (ELISA). Two different experiments in this study were detected: Firstly, mice serum samples were collected via the orbital venous plexus under isoflurane anesthesia. Additionally, BMDMs were pre-treated with the indicated concentration of LanaC for 2 h and then stimulated with 1 µg/ml LPS (S1735, Beyotime) or 10 ng/ml mouse IL-4 (404-ML, R&D) for 24 h. Conditional medium was collected for further analysis by ELISA. This study employed Interleukin-1β (IL-1β) (MLB00C, R&D), Interleukin-6 (IL-6) (M6000B, R&D), Tumor necrosis factor-α (TNFα) (MTA00B, R&D), Interleukin-12 (IL-12) (M1270, R&D), Interleukin-10 (IL-10) (M1000B, R&D), Interferon-γ (IFNγ) (MIF00, R&D), and Transforming growth factor-β (TGFβ) (DB100C, R&D) kit to measure the levels of cytokines in colon tissues and cell culture medium.
Splenocytes isolation
Spleens were harvested and mechanically dissociated using the plunger of a syringe. Tissues were digested in RPMI 1640 medium containing 100 U/mL collagenase D and 5 U/mL DNase I at 37 °C for 20 min. The suspension was filtered through 70–100 μm strainers and centrifuged. Red blood cells were lysed with RBC lysis buffer for 5 min at room temperature. Cells were washed, centrifuged at 1500 rpm for 5 min, resuspended in medium, and counted using Trypan blue exclusion.
Lamina propria lymphocytes isolation
Colon tissues were flushed with PBS, cut longitudinally into 1 cm pieces, washed in PBS containing 2% FBS, and vortexed to remove mucus. Samples were incubated at 37 °C with PBS containing 5% FBS and 1 μM DTT for 20 min, followed by incubation in 1.5 mM EDTA/PBS for 20–30 min to remove epithelial cells. After repeated washes, tissues were minced and digested with RPMI containing 100 U/mL collagenase D, 5 U/mL DNase I, and 5% FBS for 20 min at 37 °C (repeated twice). Digests were passed through a syringe and cell strainer, washed, and subjected to Percoll density gradient centrifugation (40%/60%). Cells at the interface were collected, washed, and counted by Trypan blue exclusion.
Flow cytometry
Single-cell suspensions were washed twice in FACS buffer. Fcγ receptors were blocked with anti-mouse CD16/CD32 antibody (553141, BD Biosciences, 2.4G2), followed by staining with fluorochrome-conjugated antibodies for 30 min. After washing, samples were analyzed on a Cytek Aurora spectral cytometer (Cytek Biosciences) and processed using FlowJo software. The following antibodies were used in this study: CD45-PE (157603, BioLegend, QA17A26), CD19-FITC (152403, BioLegend, 1D3/CD19), CD3-BV650 (100229, BioLegend, 17A2), CD4-APC (100515, BioLegend, RM4-5), CD8-BV786 (100749, BioLegend, 53-6.7), F4/80-PE-CY7 (123113, BioLegend, BM8), CD11b-BV421 (101235, BioLegend, M1/70).
Generation of bone marrow-derived macrophages (BMDM)
For isolation of BMDMs, tibias and femurs were removed from mice by sterile techniques and bone marrow was flushed with fresh RPMI medium using a 271/4 gage needle. Cells were plated in medium supplemented with 10% (v/v) fetal bovine serum and 10 ng/mL of M-CSF was added to induce differentiation of myeloid progenitors into mature macrophages. Medium was replaced every 2 days. Cells were cultured at 37 °C in a humidified atmosphere of 5% CO2 for 7 days and then harvested for subsequent experiments.
Screening of cellular drug delivery concentrations
Mature BMDMs were seeded at 5 × 103 cells per well in 96-well plates and allowed to adhere overnight. Cells were then treated with varying concentrations of LanaC for 24 h. DMSO was used as the vehicle control. CCK-8 solution (MedChemExpress) was added, and plates were incubated for 1 h at 37 °C in a humidified 5% CO₂ atmosphere. Absorbance at 450 nm was measured using a microplate reader, and the cell viability was computed. Cell viability (%) = (A sample − A blank) / (A control − A blank) * 100%. The half-maximal inhibitory concentration (IC₅₀) of LanaC was determined from dose–response curves.
Immunofluorescence
Mature BMDMs were seeded at 1 × 106 cells per well in 12-well plates and allowed to adhere overnight. Cells were then pre-treated with indicated concentration of LanaC for 2 h, with DMSO used as the vehicle control, and subsequently stimulated with 1 µg/ml LPS or 10 ng/ml mouse IL-4 for 24 h. After three washes with PBS, cells were fixed in 4% paraformaldehyde at room temperature for 30 min, and permeabilized with 0.25% Triton X-100 for 5 min when staining for iNOS. Nonspecific binding was blocked with 5% BSA for 1 h at room temperature. Cells were incubated overnight at 4 °C with rabbit polyclonal primary antibodies against iNOS (1:500), or CD206 (1:500) in PBS. After washing with PBS, cells were incubated with Alexa Fluor 488-conjugated anti-rabbit IgG secondary antibody (1:1000) at room temperature for 2 h in the dark at room temperature. Nuclei were counterstained with DAPI (1:10,000) for 10 min. Images were captured with an inverted fluorescence microscope and analyzed using the MShot Image analysis system.
Western blot
Mature BMDMs were seeded at 1 × 106 cells per well in 12-well plates and allowed to adhere overnight. Cells were then pre-treated with 20 µM of LanaC for 2 h, with DMSO used as the vehicle control, and subsequently stimulated with 1 µg/ml LPS or 10 ng/ml mouse IL-4 for indicated time. Cells were washed in PBS after the stimulation and lysed in RIPA buffer (89900, Thermo fisher) on ice for 30 min. Protein concentration was quantified using BCA kit. Equal amounts of protein were denatured by boiling, separated on Precast PAGE gels, and transferred onto PVDF membrane. Membranes were blocked with 5% skim milk in Tris-buffered saline containing 0.1% Tween-20 (TBST) for 1 h at room temperature and then incubated overnight at 4 °C with primary antibodies (1:1000) against p-P38 (58425, Cell signaling), P38 (9212, Cell signaling), p-ERK (9101, Cell signaling), ERK (9102, Cell signaling), p-P65 (3033, Cell signaling), P65 (8242, Cell signaling), p-STAT1 (9167, Cell signaling), STAT1 (9172, Cell signaling), p-STAT3 (9145, Cell signaling), STAT3 (9132, Cell signaling), p-STAT6 (9361, Cell signaling), STAT6 (9362, Cell signaling), IRF5 (20261, Cell signaling), PPARγ (2430, Cell signaling), and β-actin (4967, Cell signaling). On the second day, after three washes in TBST (15 min each), membranes were incubated with HRP-conjugated secondary antibodies (1:4000) for 1 h at room temperature. Following three additional washes in TBST, protein bands were visualized using an ECL detection kit and imaged with Image Pro Plus 6.0 (Media Cybernetics). Band intensities were quantified using ImageJ software, and the corresponding quantitative results are shown in Supplementary Fig. 4.
Statistical analysis
Data are presented as mean ± standard deviation (SD) or mean ± standard error of the mean (SEM) from at least three independent experiments. Statistical comparisons among multiple groups were performed using one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test in IBM SPSS Statistics 25.0. Graphs were generated with GraphPad Prism 10.2. Differences were considered statistically significant at p-value < 0.05.
Results
LanaC treatment protects mice from DSS-induced colitis
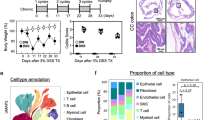
To evaluate the therapeutic potential of LanaC in UC, mice were administered 3% DSS in drinking water for 7 days, followed by normal water for 3 days to induce experimental colitis (Fig. 1A). DSS exposure resulted in a progressive reduction in body weight (Fig. 1B), reaching the lowest point on day 7, with an average loss of 21.1% ± 1.5% relative to baseline (day 0). In addition, DSS treatment induced typical manifestations of colitis, including increased disease activity index (DAI) scores (Fig. 1C) and a significant shortening of colon length (Fig. 1D) relative to the NC group. Notably, administration of LanaC markedly mitigated these colitis-associated pathological changes. Meanwhile, given that cardiac glycosides (LanaC) are known to influence Na⁺/K⁺ homeostasis30, food intake and water consumption were also monitored. DSS exposure caused a slight reduction in food intake on days 4, 5, and 6, which was not altered by LanaC treatment (Supplementary Fig. 1A). In contrast, DSS significantly reduced water consumption compared with the NC group from day4, and this decrease was not significantly alleviated by LanaC treatment (Supplementary Fig. 1B). Interestingly, mice in the DSS + LanaC group showed a transient increase in water consumption on day 1 (24 h post-first LanaC administration); however, this effect was not maintained in the following days (Supplementary Fig. 1B). Histological analysis further revealed that DSS-treated mice displayed severe colonic inflammation, characterized by mononuclear cell infiltration and destruction of the crypt, whereas these alterations were markedly attenuated by LanaC treatment (Fig. 1E).
LanaC alleviates DSS-induced colitis symptoms. (A) Experimental procedure diagram; (B) Changes in body weight of mice during the DSS-induced colitis experimental period; (C) Clinical symptoms of DSS-induced colitis were determined by the disease activity index (DAI); (D) Colon length and representative images of the colon for each groups; (E) H&E staining (upper: 100 × , lower: 400 ×) reveals histological alterations in colon tissue. Scale bars represent 100 μm. All experiments were performed with 6 mice per group. Data in (B), (D), (E) are presented as means ± SD, whereas data in (C) is presented as mean ± SEM. Statistical analysis was performed using one-way ANOVA followed by Tukey’s post hoc test. *p < 0.05, **p < 0.01, ***p < 0.001.
Having established the protective effect of LanaC in the DSS-induced colitis model, we next investigated intestinal barrier integrity by assessing tight junction proteins. DSS exposure significantly downregulated Claudin4, Occludin, and ZO1, while LanaC administration restored their expression levels (Fig. 2A). In line with these results, RT-PCR analysis confirmed that LanaC treatment upregulated Occludin and ZO1 mRNA expression (Fig. 2B). Considering the importance of goblet cells in mucosal repair, we further examined their abundance in colonic tissues. DSS challenge caused a pronounced loss of goblet cells, which was substantially preserved by LanaC treatment (Fig. 2C). Collectively, these findings demonstrate that LanaC mitigates DSS-induced colitis by preserving gut barrier integrity and attenuating intestinal tissue injury.
LanaC reduces DSS-induced intestinal barrier damage. (A) Analysis of Claudin4, Occludin, and ZO1 protein expression level in colon tissue by immunohistochemistry. Scale bars represent 100 μm; (B) RT-PCR determination of Claudin4, Occludin, and Zo1 mRNA levels in colon tissue; (C) Alcian Blue–PAS staining (upper: 200 × , lower: 400 ×) and goblet cell counting in colon tissue. Scale bars represent 100 μm; All experiments were performed with 6 mice per group and presented as means ± SD. Statistical analysis was performed using one-way ANOVA followed by Tukey’s post hoc test. **p < 0.01, ***p < 0.001.
LanaC inhibits local and system inflammation in DSS-induced colitis
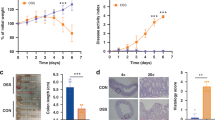
Dysregulated immune response constitutes a hallmark of UC pathogenesis. Our findings reveal that DSS-treated mice developed splenomegaly, evidenced by increased spleen weight for 50%, which was significantly reduced following LanaC treatment (Fig. 3A). Given the spleen’s critical role in immune regulation, we hypothesized that LanaC could modulate the inflammatory response induced by DSS. To test the anti-inflammatory effect of LanaC, RT-PCR was performed to determine the mRNA levels of cytokines and chemokines in colon tissue. Results revealed that the mRNA expression levels of the pro-inflammatory cytokines IL-1β, IL-6, IFNγ, and TNFα were markedly elevated in DSS-treated mice compared with the control group (Fig. 3B). Notably, administration of LanaC significantly reduced the mRNA expression of these cytokines. DSS exposure also induced an increase in IL-12 mRNA expression. However, LanaC treatment exerted no significant effect on this cytokine (Fig. 3B). In addition, we observed that LanaC treatment markedly enhanced the mRNA levels of the anti-inflammatory cytokines IL-10 and TGFβ, as well as the chemokine CCL1 (Fig. 3B). We next examined the DSS-induced cytokines and chemokines protein expression level in mice serum by ELISA. Consistent with the findings in colon tissue, ELISA results showed that LanaC treatment suppressed the protein levels of pro-inflammatory cytokines IL-1β, IL-6, IL-12, and TNFα, while showing no significant effect on IFNγ expression. Moreover, LanaC promoted the protein expression level of the anti-inflammatory cytokine IL-10 (Fig. 3C). All these results suggested that LanaC may promote intestinal tissue repair while simultaneously suppressing inflammation.
LanaC inhibits both local and systemic inflammation. (A) Spleen weight and images of spleens for each groups; (B) RT-PCR determination of IL-1β, IL-6, IFNγ, TNFα, IL-12, IL-10, TGFβ, and CCL1 mRNA levels in colon tissue; (C) ELISA analysis of IL-1β, IL-6, TNFα, IL-12, INFγ, and IL-10 in mice ocular blood serum; All experiments were performed with 6 mice per group and presented as means ± SD. Statistical analysis was performed using one-way ANOVA followed by Tukey’s post hoc test. *p < 0.05, **p < 0.01, ***p < 0.001.
LanaC reduces macrophage infiltration during DSS colitis
A large infiltration of immune cells in intestinal tissues is another characteristic of colitis. To further confirm the anti-inflammatory effect of LanaC, we used flow cytometry to detect the immunophenotype of infiltrated immune cells in colon tissues. The gating strategy is detailed in Supplementary Fig. 2A. Flow cytometric analysis revealed that DSS administration led to a marked increase in CD45⁺ immune cells infiltration in colon tissue relative to the NC group. Treatment with LanaC was associated with a slight reduction in CD45⁺ cell infiltration, although this change did not reach statistical significance (Supplementary Fig. 2B). Further characterization of the CD45⁺ immune cells indicated that the proportions of CD4+ T cells, CD8+ T cells, and CD19+ B cells were comparable between the DSS and DSS + LanaC group (Supplementary Fig. 2C). Macrophages are key regulators of intestinal homeostasis as well as the initiation and progression of gut inflammation. We subsequently assessed macrophage infiltration in colon tissue and found that DSS administration significantly increased the proportion of F4/80⁺CD11b⁺ macrophages in colon tissue, whereas LanaC treatment was associated with an attenuation of this increase (Fig. 4A). A similar pattern was observed in the spleen (the gating strategy is detail in Supplementary Fig. 3), where DSS treatment increased the proportion of macrophages in the spleen compared with the NC group, and this increase was attenuated by LanaC treatment (Fig. 4B). These findings suggest that LanaC treatment is associated with modulation of DSS-induced macrophage infiltration in both colon and spleen tissues.
LanaC reduces DSS-induced macrophage infiltration and modulates macrophage polarization. (A) The proportion of macrophages (CD11b⁺F4/80⁺) in the colon tissues were assessed by flow cytometry; (B) The proportion of macrophages (CD11b⁺F4/80⁺) in the spleen were assessed by flow cytometry; (C) The percentage of iNOS+ or CD206+ macrophages in the spleen and colon tissues were determined by flow cytometry; (D) RT-PCR determination of iNOS, CD80, Arg1, and CD206 mRNA levels in colon tissue; All experiments were performed with 6 mice per group and presented as means ± SD. Statistical analysis was performed using one-way ANOVA followed by Tukey’s post hoc test. *p < 0.05, **p < 0.01, ***p < 0.001.
Dysregulation of macrophage polarization plays a critical role in the development of UC. To assess this, we examined colonic infiltrating macrophages, using iNOS as M1-polarization marker and CD206 as M2-polarization marker. Our results demonstrated that DSS-induced macrophage polarization, with more than 30% of macrophages identified as iNOS+, indicative of M1 polarization. Treatment with LanaC significantly suppressed this M1 polarization (Fig. 4C). In contrast, approximately 20% of macrophages were CD206+ under DSS treatment, reflecting M2 polarization, and this proportion was markedly increased to 40% following LanaC treatment (Fig. 4C). RT-PCR analysis further corroborated these findings, showing reduced mRNA expression of M1 markers (iNOS and CD80) alongside increased expression of M2 markers (Arg1 and CD206) (Fig. 4D). Collectively, these results indicate that LanaC treatment is associated with a reduction in macrophage infiltration and a shift in macrophage polarization toward a more M2-like phenotype in both colon and spleen tissues.
LanaC suppresses M1 and promotes M2 polarization in BMDMs
Previous studies have shown that LanaC suppresses LPS-induced cytokine expression in RaW264.7 cells, suggesting a potential role in modulating macrophage M1 polarization27. To further verify this hypothesis, we employed mouse bone marrow–derived macrophages (BMDMs) and induced them toward either M1 or M2 polarization. Cell viability assays first established that LanaC at concentrations below 50 μM did not exert cytotoxic effects on BMDMs (Fig. 5A). We then established macrophage polarization models by stimulating cells with LPS or IL-4 to induce M1 or M2 polarization, respectively. Immunofluorescence staining revealed that LanaC markedly suppressed LPS-induced iNOS expression while enhancing IL-4-induced CD206 expression (Fig. 5B). Consistent results were obtained from flow cytometry analysis, which showed that LanaC inhibited the expression of M1 markers CD80 and MHCII, while significantly increased the expression of M2 markers CD163 and Arg1(Fig. 5C). Moreover, LanaC treatment reduced the production of LPS-induced pro-inflammatory cytokines (IL-1β, IL-6, and TNFα) (Fig. 5D) and concomitantly promoted the expression of IL-4-induced anti-inflammatory cytokines (IL-10 and TGFβ) (Fig. 5D). Collectively, these results indicate that LanaC inhibits LPS-induced M1 polarization and inflammatory cytokine production while promoting IL-4-induced M2 polarization in macrophages.
LanaC exerts a direct effect on BMDMs polarization: (A) The cytotoxic effect of LanaC on BMDMs was evaluated using the CCK-8 assay; (B) Immunohistochemical analysis of iNOS and CD206 expression on BMDMs stimulated with LPS or IL-4; (C) Flow cytometric analysis of CD80, MHCII, CD163, and Arg1 expression on BMDMs stimulated with LPS or IL-4; (D) ELISA analysis of IL-1β, IL-6, TNFα, IL-10, and TGFβ in cell culture medium. Results are expressed as mean ± SD from a minimum of three independent experiments. Statistical analysis was performed using one-way ANOVA followed by Tukey’s post hoc test. *p < 0.05, **p < 0.01, ***p < 0.001.
LanaC regulates activation of STAT signaling pathways in BMDMs
To further clarify the underlying mechanisms by which LanaC regulates macrophage polarization following LPS- or IL-4-induced stimulation, we first examined the activation of the MAPK31 and NF-κB32 signaling pathways, which are hallmarks of M1 macrophage polarization. The results showed that LanaC treatment did not affect LPS-induced phosphorylation of p38 or ERK (Fig. 6A). Consistently, LanaC caused a modest reduction in LPS-induced NF-κB phosphorylation; however, this effect did not reach statistical significance (Fig. 6B). Similarly, the expression of the downstream transcription factor IRF5 at both the protein (Fig. 6C) and mRNA levels (Fig. 6D) was not significantly altered. In addition, IL-4 stimulation induced a slight phosphorylation phosphorylation of p38; however, this effect was not altered by LanaC treatment neither (Fig. 6E).
LanaC selectively influences STAT signaling without affecting MAPK or NF-κB activation in BMDMs: (A) The protein levels of p-P38, P38, p-ERK and ERK in LPS stimulated BMDMs were detected by western blot; (B) The protein levels of p-P65 and P65 in LPS stimulated BMDMs were detected by western blot; (C) The protein levels of IRF5 in LPS stimulated BMDMs were detected by western blot; (D) RT-PCR determination of IRF5 mRNA levels in LPS stimulated BMDMs; (E) The protein levels of p-P38 and P38 in IL-4 stimulated BMDMs were detected by western blot; (F) The protein levels of p-STAT1, STAT1, p-STAT3 and STAT3 in LPS stimulated BMDMs were detected by western blot; (G) The protein levels of p-STAT6 and STAT6 in IL-4 stimulated BMDMs were detected by western blot; (H) The protein levels of PPARγ in IL-4 stimulated BMDMs were detected by western blot; Original blots for panels (A–C) are presented in Supplementary Fig. 5, and those for panels (E–H) are presented in Supplementary Fig. 6.
Given that STAT signaling pathways play critical roles in regulating macrophage polarization, we next assessed the activation of STAT family members33,34,35. As expected, LPS stimulation robustly induced phosphorylation of STAT1 and STAT3, whereas co-treatment with LanaC markedly suppressed this activation (Fig. 6F). In contrast, LanaC enhanced IL-4-induced phosphorylation of STAT6 (Fig. 6G) and concomitantly increased the expression of its downstream target PPARγ (Fig. 6H). Collectively, these results indicate that LanaC does not affect the activation of MAPK or NF-κB signaling pathways, but selectively inhibits LPS-induced STAT1/STAT3 activation while promoting IL-4-induced STAT6 signaling in macrophages, which may underlie the modulatory effects of LanaC on macrophage polarization.
Discussion
Ulcerative colitis (UC) continues to pose a significant global health challenge, leading to chronic morbidity and considerable socioeconomic burden36. Despite advances in therapy, the etiology of UC has not been fully elucidated, and current treatments are often limited by incomplete efficacy and adverse effects36,37. Thus, there is a pressing need to identify new therapeutic targets and explore alternative treatment options. In this context, our study provides experimental evidence that LanaC confers significant protection against colitis in a DSS-induced mouse model. LanaC treatment alleviated both systemic and local inflammatory manifestations, mitigated disease symptoms, and directly modulated macrophage responses by reducing infiltration into the spleen and colonic tissues, suppressing pro-inflammatory M1 polarization, and promoting anti-inflammatory M2 polarization.
The DSS model recapitulates key pathological and clinical features of human UC, such as mucosal inflammation, bloody diarrhea, and epithelial barrier disruption, making it a widely used platform for studying pathogenesis and testing therapies36. Although LanaC is clinically approved as a cardiac glycoside for heart failure and arrhythmias, recent network pharmacology analyses have suggested its potential in intestinal inflammation27. Consistent with these predictions, in vitro studies have shown that LanaC suppresses LPS-induced pro-inflammatory cytokine production in RAW264.7 macrophages. However, prior to our work, direct evidence for its protective role in intestinal inflammation from animal models had not been established. In the present study, LanaC administration significantly ameliorated DSS-induced colitis. Treated mice showed reduced weight loss, decreased diarrhea and hematochezia, and improved histological outcomes. Given that epithelial barrier dysfunction and persistent inflammation are central drivers of UC pathogenesis, the protective effects of LanaC on barrier integrity are noteworthy. Histological analyses revealed that LanaC preserved goblet cell populations and enhanced the expression of tight junction proteins, including Claudin4, Occludin, and ZO1, underscoring its role in maintaining epithelial barrier function. Barrier preservation is closely intertwined with immune regulation38,39. Once barrier integrity is compromised, luminal antigens can trigger excessive immune responses, leading to the release of cytokines such as IL-1β, IL-6, IL-12, IFNγ, and TNFα, which amplify tissue injury40,41,42. Our results demonstrated that LanaC markedly suppressed these pro-inflammatory cytokines in DSS-treated mice, while simultaneously elevating IL-10 and TGFβ, two key anti-inflammatory mediators. This dual action suggests that LanaC not only dampens inflammation but also actively supports resolution and mucosal healing.
Macrophages play a pivotal role in UC progression, functioning at the intersection of immune regulation and tissue repair43. The polarization state of macrophages critically influences disease outcomes: M1 macrophages exacerbate inflammation and barrier disruption, whereas M2 macrophages promote repair and resolution10,44,45. Consistent with this paradigm, our findings revealed that LanaC treatment was associated with reduced macrophage infiltration in both spleen and colon and a shift in polarization toward the M2 phenotype, suggesting a more anti-inflammatory environment. These effects were further reflected in vitro. In bone marrow–derived macrophages (BMDMs), LPS stimulation induced strong M1 polarization characterized by elevated expression of iNOS, CD80, and MHCII, whereas LanaC treatment significantly reduced these markers. Conversely, IL-4 stimulation promoted M2 polarization, marked by increased CD206, CD163, and Arg1 expression, and LanaC further amplified this effect. Moreover, LanaC inhibited LPS-induced pro-inflammatory cytokine production while enhancing IL-4–induced anti-inflammatory cytokine expression, confirming its direct role in shaping macrophage responses.
To elucidate the signaling mechanisms underlying this macrophage reprogramming, we further examined the activation of key inflammatory pathways. Notably, LanaC did not substantially alter LPS-induced activation of MAPK or NF-κB pathways. These observations suggest that the effects of LanaC on macrophage polarization are unlikely to result from a broad suppression of inflammatory signaling. Instead, LanaC selectively modulated STAT-dependent pathways that are central to macrophage fate decisions. While LPS-induced phosphorylation of STAT1 and STAT3—key drivers of M1 polarization—was markedly attenuated by LanaC, IL-4–induced activation of STAT6, a master regulator of M2 polarization, was further enhanced. This biased regulation of STAT signaling provides a mechanistic explanation for the observed suppression of pro-inflammatory macrophage responses and the concomitant promotion of an anti-inflammatory, tissue-repairing phenotype.
Taken together, our data indicate that LanaC treatment is associated with preservation of epithelial barrier integrity, rebalancing of inflammatory cytokine profiles, and modulation of macrophage polarization in DSS-induced colitis. In vitro studies further demonstrate that LanaC suppresses LPS-induced M1 differentiation while promoting IL-4–driven M2 polarization, an effect that is accompanied by attenuation of STAT1/STAT3 signaling and enhancement of STAT6 activation. By selectively rebalancing opposing STAT-dependent macrophage programs, LanaC supports an anti-inflammatory and tissue-repair–oriented environment, highlighting its potential as a therapeutic candidate for ulcerative colitis.
Although this study provides valuable insights into the therapeutic potential of LanaC in DSS-induced colitis models, several limitations should be acknowledged. First, our investigation primarily focused on the impact of LanaC on macrophage polarization, demonstrating its ability to inhibit M1 polarization and promote M2 polarization. However, LanaC is a cardiac glycoside and a known inhibitor of Na⁺/K⁺-ATPase24, while Na⁺/K⁺-ATPase activity has been implicated in the regulation of macrophage differentiation and inflammatory signaling46,47. A limitation of the present study is that we did not directly assess whether LanaC alters Na⁺/K⁺-ATPase activity in macrophages. Therefore, it remains unclear to what extent the observed effects of LanaC on macrophage polarization are mediated through modulation of Na⁺/K⁺-ATPase activity versus downstream signaling pathways independent of ion transport. Future studies will be required to systematically dissect the contribution of Na⁺/K⁺-ATPase-dependent and -independent mechanisms in LanaC-mediated regulation of macrophage differentiation, including direct measurements of Na⁺/K⁺-ATPase activity, as well as targeted deletion of key compenents involved in Na⁺/K⁺-ATPase-associated signaling, to determine whether disruption of these pathways abolishes the immunomodulatory effects of LanaC.
Furthermore, although our in vivo data indicate that LanaC treatment is associated with altered macrophage polarization in DSS-induced colitis, and our results further demonstrate that LanaC can directly modulate LPS/IL-4-induced macrophage polarization in vitro, these observations remain correlative in nature. Future studies employing macrophage depletion48 or adoptive transfer49 approaches will be necessary to establish a causal role for macrophages in mediating the therapeutic effects of LanaC in DSS-induced colitis.
Lastly, the use of BALB/c mice, a prototypical Th2-type strain50,51, may have influenced the outcomes of this study. DSS-induced colitis is characterized by a sustained upregulation of Th1-type cytokines (TNFα, IFNγ, IL-1β, and IL-12) during the acute phase52, whereas Th2-associated mediators (IL-4, IL-6, and IL-10) are predominantly involved in the chronic phase53,54. Given the inherent Th2 bias of BALB/c mice, the expression of Th1-type cytokines during the acute phase may have been attenuated, while Th2-associated responses were potentially favored. This strain-specific bias could have contributed to the limited observable effects of LanaC on Th1-type immune response–associated cells55,56, which may also explain why we did not detect significant DSS-induced infiltration of CD4⁺ T cells compare with NC group (Supplementary Fig. 2C). Future studies utilizing additional mouse strains, such as Th1-type stain C57BL/6, will be critical to enhance the generalizability of our findings and to achieve a more comprehensive understanding of the immunomodulatory effects of Lanas.
Conclusion
In conclusion, this study demonstrates that LanaC represents a novel therapeutic agent for experimental colitis, acting primarily by alleviating intestinal inflammation and restoring epithelial barrier integrity. Importantly, LanaC treatment was associated with modulation of innate immune responses, including reduced macrophage infiltration, a shift from M1 to M2 polarization, and altered cytokine profiles. Mechanistically, these immunomodulatory effects are accompanied by attenuation of STAT1/STAT3 signaling and enhancement of STAT6 activation in macrophages, suggesting a STAT-biased reprogramming of macrophage responses. These findings provide new insights into the immunopathogenesis of ulcerative colitis and highlight LanaC as a promising candidate for the development of macrophage-targeted therapies in inflammatory bowel diseases.
Data availability
No datasets were generated or analysed during the current study. The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Gros, B. & Kaplan, G. G. Ulcerative colitis in adults: A review. JAMA 330, 951–965. https://doi.org/10.1001/jama.2023.15389 (2023).
Du, L. & Ha, C. Epidemiology and pathogenesis of ulcerative colitis. Gastroenterol. Clin. North. Am. 49, 643–654. https://doi.org/10.1016/j.gtc.2020.07.005 (2020).
M’Koma, A. E. Inflammatory bowel disease: An expanding global health problem. Clin. Med. Insights Gastroenterol. 6, 33–47. https://doi.org/10.4137/CGast.S12731 (2013).
Awan, H., Fatima, U., Eaw, R., Knox, N. & Alrubaiy, L. The Efficacy of currently licensed biologics for treatment of ulcerative colitis: A literature review. Cureus 15, e37609. https://doi.org/10.7759/cureus.37609 (2023).
Bruscoli, S., Febo, M., Riccardi, C. & Migliorati, G. Glucocorticoid therapy in inflammatory bowel disease: Mechanisms and clinical practice. Front. Immunol. 12, 691480. https://doi.org/10.3389/fimmu.2021.691480 (2021).
Guo, R., Meng, Q., Wang, B. & Li, F. Anti-inflammatory effects of Platycodin D on dextran sulfate sodium (DSS) induced colitis and E. coli Lipopolysaccharide (LPS) induced inflammation. Int. Immunopharmacol. 94, 107474. https://doi.org/10.1016/j.intimp.2021.107474 (2021).
Ko, C. W. et al. AGA clinical practice guidelines on the management of mild-to-moderate ulcerative colitis. Gastroenterology 156, 748–764. https://doi.org/10.1053/j.gastro.2018.12.009 (2019).
Chen, S. et al. Macrophages in immunoregulation and therapeutics. Signal Transduct. Target Ther. 8, 207. https://doi.org/10.1038/s41392-023-01452-1 (2023).
Na, Y. R., Stakenborg, M., Seok, S. H. & Matteoli, G. Macrophages in intestinal inflammation and resolution: A potential therapeutic target in IBD. Nat. Rev. Gastroenterol. Hepatol. 16, 531–543. https://doi.org/10.1038/s41575-019-0172-4 (2019).
Luo, M., Zhao, F., Cheng, H., Su, M. & Wang, Y. Macrophage polarization: An important role in inflammatory diseases. Front. Immunol. 15, 1352946. https://doi.org/10.3389/fimmu.2024.1352946 (2024).
Moreira Lopes, T. C., Mosser, D. M. & Goncalves, R. Macrophage polarization in intestinal inflammation and gut homeostasis. Inflamm. Res. 69, 1163–1172. https://doi.org/10.1007/s00011-020-01398-y (2020).
Chen, Y. H. et al. Regorafenib enhances M1/M2 macrophage polarization by inhibiting the secretion of plasminogen activator inhibitor-1 in head and neck squamous cell carcinoma. Life Sci. 358, 123147. https://doi.org/10.1016/j.lfs.2024.123147 (2024).
Xiao, Q. et al. Formononetin alleviates ulcerative colitis via reshaping the balance of M1/M2 macrophage polarization in a gut microbiota-dependent manner. Phytomedicine 135, 156153. https://doi.org/10.1016/j.phymed.2024.156153 (2024).
Orecchioni, M., Ghosheh, Y., Pramod, A. B. & Ley, K. Corrigendum: Macrophage polarization: Different gene signatures in M1(LPS+) vs. classically and M2(LPS-) vs. alternatively activated macrophages. Front. Immunol. 11, 234. https://doi.org/10.3389/fimmu.2020.00234 (2020).
Ley, K., Pramod, A. B., Croft, M., Ravichandran, K. S. & Ting, J. P. How mouse macrophages sense what is going on. Front. Immunol. 7, 204. https://doi.org/10.3389/fimmu.2016.00204 (2016).
Han, X., Ding, S., Jiang, H. & Liu, G. Roles of macrophages in the development and treatment of gut inflammation. Front. Cell. Dev. Biol. 9, 625423. https://doi.org/10.3389/fcell.2021.625423 (2021).
Zhang, K., Guo, J., Yan, W. & Xu, L. Macrophage polarization in inflammatory bowel disease. Cell. Commun. Signal. 21, 367. https://doi.org/10.1186/s12964-023-01386-9 (2023).
Wu, M. M. et al. Dioscin ameliorates murine ulcerative colitis by regulating macrophage polarization. Pharmacol. Res. 172, 105796. https://doi.org/10.1016/j.phrs.2021.105796 (2021).
Canton, M. et al. Reactive oxygen species in macrophages: sources and targets. Front. Immunol. 12, 734229. https://doi.org/10.3389/fimmu.2021.734229 (2021).
Formentini, L. et al. Mitochondrial ROS production protects the intestine from inflammation through functional M2 macrophage polarization. Cell. Rep. 19, 1202–1213. https://doi.org/10.1016/j.celrep.2017.04.036 (2017).
Ito, R. et al. Involvement of IL-17A in the pathogenesis of DSS-induced colitis in mice. Biochem. Biophys. Res. Commun. 377, 12–16. https://doi.org/10.1016/j.bbrc.2008.09.019 (2008).
Botelho, A. F. M., Pierezan, F., Soto-Blanco, B. & Melo, M. M. A review of cardiac glycosides: Structure, toxicokinetics, clinical signs, diagnosis and antineoplastic potential. Toxicon 158, 63–68. https://doi.org/10.1016/j.toxicon.2018.11.429 (2019).
Ponce, A., Flores-Maldonado, C. & Contreras, R. G. Cardiac glycosides: From natural defense molecules to emerging therapeutic agents. Biomolecules https://doi.org/10.3390/biom150608854 (2025).
Chao, M. W. et al. Lanatoside C, a cardiac glycoside, acts through protein kinase Cdelta to cause apoptosis of human hepatocellular carcinoma cells. Sci. Rep. 7, 46134. https://doi.org/10.1038/srep46134 (2017).
Liu, X. & Lv, K. Cruciferous vegetables intake is inversely associated with risk of breast cancer: A meta-analysis. Breast 22, 309–313. https://doi.org/10.1016/j.breast.2012.07.013 (2013).
Jansson, D. et al. Cardiac glycosides target barrier inflammation of the vasculature, meninges and choroid plexus. Commun. Biol. 4, 260. https://doi.org/10.1038/s42003-021-01787-x (2021).
Zhu, W., Zhang, Z. & Wang, X. Network pharmacology analysis of Lanatoside C: Molecular targets and mechanisms in the treatment of ulcerative colitis. Front. Mol. Biosci. 12, 1552360. https://doi.org/10.3389/fmolb.2025.1552360 (2025).
Wirtz, S. et al. Chemically induced mouse models of acute and chronic intestinal inflammation. Nat. Protoc. 12, 1295–1309. https://doi.org/10.1038/nprot.2017.044 (2017).
Morampudi, V. et al. The goblet cell-derived mediator RELM-beta drives spontaneous colitis in Muc2-deficient mice by promoting commensal microbial dysbiosis. Mucosal. Immunol. 9, 1218–1233. https://doi.org/10.1038/mi.2015.140 (2016).
Gonlubol, F., Siegel, A. & Bing, R. J. Effect of a cardiac glycoside (cedilanid) on the sodium and potassium balance of the human heart. Circ. Res. 4, 298–301. https://doi.org/10.1161/01.res.4.3.298 (1956).
Kerneur, C., Cano, C. E. & Olive, D. Major pathways involved in macrophage polarization in cancer. Front. Immunol. 13, 1026954. https://doi.org/10.3389/fimmu.2022.1026954 (2022).
Liu, T., Zhang, L., Joo, D. & Sun, S. C. NF-kappaB signaling in inflammation. Signal Transduct. Target. Ther. 2, 17023. https://doi.org/10.1038/sigtrans.2017.23 (2017).
Ohmori, Y. & Hamilton, T. A. Requirement for STAT1 in LPS-induced gene expression in macrophages. J. Leukoc. Biol. 69, 598–604 (2001).
Balic, J. J. et al. STAT3 serine phosphorylation is required for TLR4 metabolic reprogramming and IL-1beta expression. Nat. Commun. 11, 3816. https://doi.org/10.1038/s41467-020-17669-5 (2020).
Takeda, K. et al. Essential role of Stat6 in IL-4 signalling. Nature 380, 627–630. https://doi.org/10.1038/380627a0 (1996).
Kobayashi, T. et al. Ulcerative colitis. Nat. Rev. Dis. Primers 6, 74. https://doi.org/10.1038/s41572-020-0205-x (2020).
Solitano, V. et al. Reaching the therapeutic ceiling in IBD: Can advanced combination treatment (ACT) offer a solution?. Best Pract. Res. Clin. Gastroenterol. https://doi.org/10.1016/j.bpg.2025.101981 (2025).
Matar, A., Damianos, J. A., Jencks, K. J. & Camilleri, M. Intestinal barrier impairment, preservation, and repair: An update. Nutrients https://doi.org/10.3390/nu16203494 (2024).
Yang, W. H. et al. Innate mechanism of mucosal barrier erosion in the pathogenesis of acquired colitis. iScience 26, 107883. https://doi.org/10.1016/j.isci.2023.107883 (2023).
Antoni, L., Nuding, S., Wehkamp, J. & Stange, E. F. Intestinal barrier in inflammatory bowel disease. World J. Gastroenterol. 20, 1165–1179. https://doi.org/10.3748/wjg.v20.i5.1165 (2014).
Neurath, M. F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 14, 329–342. https://doi.org/10.1038/nri3661 (2014).
Neurath, M. F., Artis, D. & Becker, C. The intestinal barrier: a pivotal role in health, inflammation, and cancer. Lancet Gastroenterol. Hepatol. 10, 573–592. https://doi.org/10.1016/S2468-1253(24)00390-X (2025).
Bain, C. C. & Schridde, A. Origin, differentiation, and function of intestinal macrophages. Front. Immunol. 9, 2733. https://doi.org/10.3389/fimmu.2018.02733 (2018).
Lissner, D. et al. Monocyte and M1 macrophage-induced barrier defect contributes to chronic intestinal inflammation in IBD. Inflamm. Bowel Dis. 21, 1297–1305. https://doi.org/10.1097/MIB.0000000000000384 (2015).
Zhou, X. et al. YAP aggravates inflammatory bowel disease by regulating M1/M2 macrophage polarization and gut microbial homeostasis. Cell. Rep. 27, 1176–1189. https://doi.org/10.1016/j.celrep.2019.03.028 (2019).
Zhang, J. et al. Na/K-ATPase suppresses LPS-induced pro-inflammatory signaling through Lyn. iScience 25, 104963. https://doi.org/10.1016/j.isci.2022.104963 (2022).
Chen, Y. et al. Cardiotonic steroids stimulate macrophage inflammatory responses through a pathway involving CD36, TLR4, and Na/K-ATPase. Arterioscler. Thromb. Vasc. Biol. 37, 1462–1469. https://doi.org/10.1161/ATVBAHA.117.309444 (2017).
Li, Z. et al. Depletion of tumor associated macrophages enhances local and systemic platelet-mediated anti-PD-1 delivery for post-surgery tumor recurrence treatment. Nat. Commun. 13, 1845. https://doi.org/10.1038/s41467-022-29388-0 (2022).
Kang, D. S. et al. Macrophage transfer promotes intestinal mucosal healing by encouraging transit-amplifying cell expansion in mice. Front. Immunol. 16, 1555695. https://doi.org/10.3389/fimmu.2025.1555695 (2025).
Watanabe, H., Numata, K., Ito, T., Takagi, K. & Matsukawa, A. Innate immune response in Th1- and Th2-dominant mouse strains. Shock 22, 460–466. https://doi.org/10.1097/01.shk.0000142249.08135.e9 (2004).
Hartmann, W., Blankenhaus, B., Brunn, M. L., Meiners, J. & Breloer, M. Elucidating different pattern of immunoregulation in BALB/c and C57BL/6 mice and their F1 progeny. Sci. Rep. 11, 1536. https://doi.org/10.1038/s41598-020-79477-7 (2021).
Nakase, H., Sato, N., Mizuno, N. & Ikawa, Y. The influence of cytokines on the complex pathology of ulcerative colitis. Autoimmun. Rev. 21, 103017. https://doi.org/10.1016/j.autrev.2021.103017 (2022).
Yang, C. & Merlin, D. Unveiling colitis: A journey through the dextran sodium sulfate-induced model. Inflamm. Bowel Dis. 30, 844–853. https://doi.org/10.1093/ibd/izad312 (2024).
Bamias, G. & Cominelli, F. Role of type 2 immunity in intestinal inflammation. Curr. Opin. Gastroenterol. 31, 471–476. https://doi.org/10.1097/MOG.0000000000000212 (2015).
Yang, F. et al. Th1/Th2 balance and Th17/Treg-mediated immunity in relation to murine resistance to dextran sulfate-induced colitis. J. Immunol. Res. 2017, 7047201. https://doi.org/10.1155/2017/7047201 (2017).
Chandwaskar, R. et al. Dysregulation of T cell response in the pathogenesis of inflammatory bowel disease. Scand. J. Immunol. 100, e13412. https://doi.org/10.1111/sji.13412 (2024).
Funding
This study was supported by the National Natural Science Foundation of China (T2341019), NSFC Guangdong Joint Fund (U1132001), General Program of the National Natural Science Foundation of China (82174243 and 82204948), Natural Science Foundation of Guangdong Province, China (2023A1515110757), Guangzhou Science and Technology Plan Project (2024B03J1343), Major Sci-entific and Technological Project of Guangzhou Municipal Health Commission (20252D003), Re-search Project of Traditional Chinese Medicine Bureau of Guangdong Province (20241208), Gen-eral project of Beijing Natural Science Foundation (7242227), Fundamental Research Funds for the Central Universities (BZY-JMZY-2022-001 and 2023-JYB-JBZD-009), High-level Key Discipline of the National Administration of Traditional Chinese Medicine-Traditional Chinese Constitutional Medicine (zyyzdxk-2023251), Major Science and Technology Special Projects in Hubei Province (2023BCA005), and the Chief Scientist Research Project of Hubei Shizhen Laboratory (HSL2024SX0002).
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. LY and HZ conceived and designed the research. LY, HZ, and JL performed all experiments. LY and JL analyzed the data and prepared the figures. LY, HZ, and JW interpreted the experimental results and drafted the manuscript. XZ and JW were responsible for project administration and funding acquisition. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, L., Liu, J., Zhao, X. et al. Lanatoside C ameliorates DSS-induced colitis with improved intestinal barrier integrity and reduced M1 macrophage polarization. Sci Rep 16, 6556 (2026). https://doi.org/10.1038/s41598-026-37484-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37484-0