Abstract
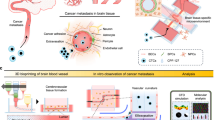
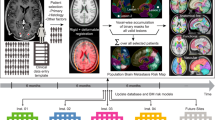
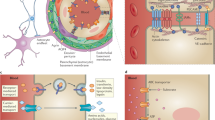
The dissemination route of brain metastases dictates their spatiotemporal distribution, but whether it alters the ultrastructural state of the blood-tumor barrier remains unresolved. Here, we compared a modified hematogenous dissemination model that minimizes extracranial signal confounds with a direct intracranial inoculation model. The hematogenous model produced multifocal brain lesions, whereas the intracranial model formed unifocal masses. Longitudinal bioluminescence imaging revealed significantly different growth rates between models, yet no statistically significant difference in overall survival was detected under the current cohort sizes and humane endpoints. Critically, exploratory transmission electron microscopy at the tumor-brain interface revealed a conserved pathological phenotype of the blood-tumor barrier in established lesions, irrespective of the seeding route. This pattern was characterized by endothelial cell swelling, a discontinuous basement membrane, and retraction of astrocytic end-feet. These findings suggest that while the seeding route determines the macroscopic pattern of disease, the established brain microenvironment imposes a stereotyped mode of neurovascular unit failure. This work provides a methodologically refined platform for studying brain metastasis and presents direct ultrastructural evidence consistent with a conserved blood-tumor barrier pathology, outlining a testable conceptual framework. These hypothesis-generating ultrastructural observations, derived from representative specimens, warrant quantitative validation.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its Supplementary Information files. The source data for tumor burden and body weights are provided in Supplementary Data 1.
References
Schaeffer, S. & Iadecola, C. Revisiting the neurovascular unit. Nat. Neurosci. 24, 1198–1209 (2021).
Lacoste, B., Prat, A., Freitas-Andrade, M. & Gu, C. The blood-brain barrier: Composition, properties, and roles in brain health. Cold Spring Harb Perspect Biol. 17, a041422 (2025).
Palomero-Gallagher, N. & Zilles, K. Cyto- and receptor architectonic mapping of the human brain. Handb Clin. Neurol 150, 355–387 (2018).
Wang, L., Xiong, X., Zhang, L. & Shen, J. Neurovascular unit: A critical role in ischemic stroke. CNS Neurosci. Ther. 27, 7–16 (2021).
Valiente, M. et al. The evolving landscape of brain metastasis. Trends Cancer 4, 176–196 (2018).
Fidler, I. J. The pathogenesis of cancer metastasis: The ‘seed and soil’ hypothesis revisited. Nat. Rev. Cancer 3, 453–458 (2003).
Kugler, E. C., Greenwood, J. & MacDonald, R. B. The ‘Neuro-Glial-Vascular’ unit: The role of glia in neurovascular unit formation and dysfunction. Front. Cell Dev. Biol. 9, 732820 (2021).
Malanchi, I. et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481, 85–89 (2012).
Calabrese, C. et al. A perivascular niche for brain tumor stem cells. Cancer Cell 11, 69–82 (2007).
Miarka, L. & Valiente, M. Animal models of brain metastasis. Neurooncol. Adv. 3, v144–v156 (2021).
Mann, B. et al. Opportunities and challenges for patient-derived models of brain tumors in functional precision medicine. npj Precis. Onc. 9, 47 (2025).
Zhang, C., Lowery, F. J. & Yu, D. Intracarotid cancer cell injection to produce mouse models of brain metastasis. JoVE 55085 (2017) https://doi.org/10.3791/55085.
Ushio, Y., Chernik, N. L., Shapiro, W. R. & Posner, J. B. Metastic tumor of the brain: Development of an experimental model. Ann. Neurol. 2, 20–29 (1977).
Kienast, Y. et al. Real-time imaging reveals the single steps of brain metastasis formation. Nat. Med. 16, 116–122 (2010).
Lim, M. et al. Modeling brain metastasis by internal carotid artery injection of cancer cells. JoVE https://doi.org/10.3791/64216 (2022)
Törteli, A., Tóth, R., Bari, F., Farkas, E. & Menyhárt, Á. Collateral is brain: Low perfusion triggers spreading depolarization and futile reperfusion after acute ischemic stroke. J. Cereb Blood Flow Metab. 44, 1881–1887 (2024).
Valiente, M. et al. Brain metastasis cell lines panel: A public resource of organotropic cell lines. Cancer Res. 80, 4314–4323 (2020).
Iadecola, C. The neurovascular unit coming of age: A journey through neurovascular coupling in health and disease. Neuron 96, 17–42 (2017).
Palomero-Gallagher, N. et al. Multireceptor analysis in human neocortex reveals complex alterations of receptor ligand binding in focal epilepsies. Epilepsia 53, 1987–1997 (2012).
Sweeney, M. D. et al. Vascular dysfunction-The disregarded partner of Alzheimer’s disease. Alzheimer’s & Dementia 15, 158–167 (2019).
Knowland, D. et al. Stepwise recruitment of transcellular and paracellular pathways underlies blood-brain barrier breakdown in stroke. Neuron 82, 603–617 (2014).
Garton, T., Gadani, S. P., Gill, A. J. & Calabresi, P. A. Neurodegeneration and demyelination in multiple sclerosis. Neuron 112, 3231–3251 (2024).
Andreone, B. J. et al. Blood-brain barrier permeability is regulated by lipid transport-dependent suppression of Caveolae-mediated transcytosis. Neuron 94, 581-594.e5 (2017).
Zhou, M. et al. Caveolae-mediated endothelial transcytosis across the blood-brain barrier in acute ischemic stroke. JCM 10, 3795 (2021).
Bojarskaite, L. et al. Role of aquaporin-4 polarization in extracellular solute clearance. Fluids Barriers CNS 21, 28 (2024).
Mehta, S. & Lo Cascio, C. Developmentally regulated signaling pathways in glioma invasion. Cell. Mol. Life Sci. 75(3), 385–402 (2018).
Stanimirovic, D. B. & Friedman, A. Pathophysiology of the neurovascular unit: disease cause or consequence?. J. Cereb. Blood Flow Metab. 32, 1207–1221 (2012).
Essig, M. et al. Perfusion MRI: The five most frequently asked clinical questions. Am. J. Roentgenol. 201, W495–W510 (2013).
Lockman, P. R. et al. Heterogeneous blood-tumor barrier permeability determines drug efficacy in experimental brain metastases of breast cancer. Clin. Cancer Res. 16, 5664–5678 (2010).
Lee, I. S. et al. Dynamic contrast-enhanced MRI in the evaluation of soft tissue Tumors and Tumor-like lesions: Technical principles and clinical applications. Korean J Radiol 26, 1054 (2025).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC) under Grant 81872489.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC) under Grant 81872489.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, animal model establishment, and data collection were performed by Jian Zhao, Yuehua Zhang, Zhigong Wei, and Kai Li. Data analysis was performed by Jian Zhao, Yuehua Zhang, and Zhigong Wei. The first draft of the manuscript was written by Jian Zhao. Dan Li and Yongsheng Wang supervised the project. All authors commented on previous versions of the manuscript and contributed to its critical revision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All animal experiments were performed in strict accordance with a protocol approved by the Animal Research Ethics Committee of West China Hospital, Sichuan University (Approval No. 20230214012). All procedures were conducted in accordance with the ARRIVE guidelines and relevant national and institutional guidelines for the care and use of laboratory animals.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, J., Zhang, Y., Wei, Z. et al. Route-dependent dissemination with conserved blood–tumor barrier ultrastructure in intracranial metastasis models. Sci Rep (2026). https://doi.org/10.1038/s41598-026-37760-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-37760-z