Abstract
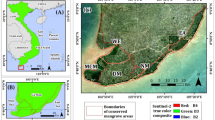
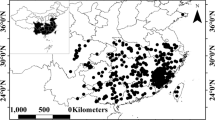
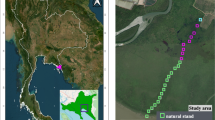
Mangrove ecosystems in Guangdong Province, China, are vital for their ecological services and biodiversity, yet comprehensive assessments of their species diversity remain limited. A biodiversity survey conducted during the months of July to August 2024 by random sampling using quadrates to assess biodiversity, species richness, and abundance as well as to evaluate species relationships with reference to the usage of mangroves area and mangrove species in the eight stations Shantou, Huiyang, Zhuhai, Huidong (Planted mangroves at higher latitudes 22–23N of non-pristine areas) Yangjiang, Lianjiang, Leizhou, and Xuwen (natural mangrove forests at lower latitudes 20–21N of pristine areas) of Guangdong’s mangrove forests. A total of 87 species (overall diversity) with eight phyla were identified, with Mollusca being the most abundant (41 species, including 28 gastropods), followed by Arthropoda (32 species) and Chordata (4 species). Seven species—Bakawan rotundata, Bakawan puti, Haloa wallisii, Platytrombidium sp., Heterocerus sp., Trichoniscus pygmaeus and SHSY140711—were recorded for the first time in China. Penthalodidae sp., key characters do not fit the diagnosis of any of its genera, represent a potential new genus in the family Penthalodidae. The brief taxonomic description for Haloa wallisii and Bakawan puti is recorded here to avoid misidentification with related species. Spatial analysis revealed Yangjiang with the highest number of specimens (N = 318), while Shantou had the lowest number of specimens (N = 25). A clear difference in biodiversity gradient was observed between natural (high biodiversity) and planted (low biodiversity) mangrove forests at pristine versus non-pristine areas, respectively. A same trend has been observed for latitudinal gradient from lower 20–21N (high biodiversity) to higher 22–23N (low biodiversity). Cluster and beta-diversity analyses highlighted distinct ecological zones, with Yangjiang, Leizhou, Xuwen, and Lianjiang exhibiting high species turnover. Analysis of species collection relative to mangrove plant species revealed that the highest proportion of organisms (30%) were found in association with Avicennia marina, while the lowest (2%) were associated with Bruguiera gymnorhiza. Over 60% of species were free-living, underscoring the ecosystem’s ecological integrity, while invasive species accounted for only 1%. This research directly aligns with the Sustainable Development Goal (SDG) 14, “Life below water” (Conserve and sustainably use the oceans, seas, and marine resources for sustainable development) and 15 “Life on land”. However, this research is also strongly supportive in achieving other SDGs such as (12. Sustainable consumption and production, 13. Climate action, 17. Partnerships for the goal). These findings emphasize the need for targeted conservation strategies, particularly in high-diversity areas, to mitigate anthropogenic threats and sustain mangrove biodiversity. The results of this study help China’s target of protecting 30 percent of the planet’s land and oceans by 2030 and provided a critical baseline for future monitoring and management efforts for biodiversity in Guangdong’s mangrove ecosystems.
Similar content being viewed by others
Data availability
“Sequence data that support the findings of this study have been deposited at GenBank under Accession No. PV700507 and PV700508 and raw sequence data is provided in the supplementary files”.
References
Alongi, D. M. Mangrove forests: Resilience, protection from tsunamis, and responses to global climate change. Estuarine Coast. Shelf Sci. 76(1), 1–13. https://doi.org/10.1016/j.ecss.2007.08.024 (2008).
Friess, D. A. et al. The state of the world’s mangrove forests: Past, present, and future. Annu. Rev. Environ. Resour. 44, 89–115. https://doi.org/10.1146/annurev-environ-101718-033302 (2019).
Wang, Y. S. & Gu, J. D. Ecological responses, adaptation and mechanisms of mangrove wetland ecosystem to global climate change and anthropogenic activities. Int. Biodeterior. Biodegrad. 162, 105248. https://doi.org/10.1016/j.ibiod.2021.105248 (2021).
Duke, N. C. et al. A world without mangroves?. Science 317(5834), 41–42. https://doi.org/10.1126/science.317.5834.41b (2007).
Aslam, S. & Wang, Y. S. Determination of nutrients, biomass, and bacterial quantification in different mangroves sites: A comparative study on nutrients dependent biomass production. Ecol. Evol. 15(7), e71697. https://doi.org/10.1002/ece3.71697 (2025).
Chen, Y. et al. Environmental capacity and fluxes of land-sourced pollutants around the Leizhou Peninsula in the summer. Front. Mar. Sci. 10, 1280753. https://doi.org/10.3389/fmars.2023.1280753 (2023).
Goldberg, L., Lagomasino, D., Thomas, N. & Fatoyinbo, T. Global declines in human-driven mangrove loss. Glob. Change Biol. 26(10), 5844–5855. https://doi.org/10.1111/gcb.15275 (2020).
Zhao, B. et al. Genetic diversity of two globally invasive snails in Asia and Americas in relation with agricultural habitats and climate factors. Diversity 14(12), 1069. https://doi.org/10.3390/d14121069 (2022).
Liang, C. Y., Zhang, H. H., Xie, X. Y. & Zou, F. S. Study on biodiversity of mangrove benthos in Leizhou Peninsula. Mar. Sci. 2, 18–25 (2005).
Gao, X. M., Han, W. D. & Liu, S. Q. The mangrove and its conservation in Leizhou Peninsula, China. J. For. Res. 20, 174–178. https://doi.org/10.1007/s11676-009-0032-0 (2009).
Lu, C. et al. The national nature reserves in China: Are they effective in conserving mangroves?. Ecol. Indic. 142, 109265. https://doi.org/10.1016/j.ecolind.2022.109265 (2022).
Yu, K. F., Zhao, J. X., Wei, G. J., Cheng, X. R. & Wang, P. X. Mid–late Holocene monsoon climate retrieved from seasonal Sr/Ca and δ18O records of Porites lutea corals at Leizhou Peninsula, northern coast of South China Sea. Glob. Planet. Change 47(2–4), 301–316. https://doi.org/10.1016/j.gloplacha.2004.10.018 (2005).
Brey, T. A multi-parameter artificial neural network model to estimate macrobenthic invertebrate productivity and production. Limnol. Oceanogr. Methods 10(8), 581–589. https://doi.org/10.4319/lom.2012.10.581 (2012).
Christianen, M. J. et al. Benthic primary producers are key to sustain the Wadden Sea food web: Stable carbon isotope analysis at landscape scale. Ecology 98(6), 1498–1512. https://doi.org/10.1002/ecy.1837 (2017).
Mathot, K. J., Piersma, T., & Elner, R. W. Shorebirds as integrators and indicators of mudflat ecology. in Mudflat ecology 309–338. https://doi.org/10.1007/978-3-319-99194-8_12 (2018).
Meijer, K. J. et al. Mangrove-mudflat connectivity shapes benthic communities in a tropical intertidal system. Ecol. Indic. 130, 108030. https://doi.org/10.1016/j.ecolind.2021.108030 (2021).
Chen, L., Wang, W., Zhang, Y. & Lin, G. Recent progresses in mangrove conservation, restoration and research in China. J. Plant Ecol. 2(2), 45–54. https://doi.org/10.1093/jpe/rtp009 (2009).
Zeng, J., Ai, B., Jian, Z., Zhao, J. & Sun, S. Simulation of mangrove suitable habitat in the Guangdong-Hong Kong-Macao Area under the background of climate change. J. Environ. Manag. 351, 119678. https://doi.org/10.1016/j.jenvman.2023.119678 (2024).
Li, H. Y., Peng, Y. S., Liu, J. J., Wang, S. G. & Chen, G. Z. Current state of mangrove floristic composition and characteristics of communities on the eastern coast of Guangdong Province. Acta Ecol. Sin. 36(1), 252–260. https://doi.org/10.5846/stxb201408031548 (2016).
MacKinnon, J., Verkuil, Y. I., & Murray, N. IUCN situation analysis on East and Southeast Asian intertidal habitats, with particular reference to the Yellow Sea (including the Bohai Sea). Occasional paper of the IUCN species survival commission, 47 (2012).
Yang, X. et al. Exploring the driving factors of bird diversity in mangrove natural protected areas in Guangdong Province, China. Front. Ecol. Evol. 12, 1421189. https://doi.org/10.3389/fevo.2024.1421189 (2024).
Blaber, S. J. M. Mangroves and fishes: Issues of diversity, dependence, and dogma. Bull. Mar. Sci. 80(3), 457–472 (2007).
Yates, K. K. et al. Mangrove habitats provide refuge from climate change for reef-building corals. Biogeosci. Discuss. 3(11), 5053–5088. https://doi.org/10.5194/bgd-11-5053-2014 (2014).
Kesavan, S. et al. Anthropogenic pressure on mangrove ecosystems: Quantification and source identification of surficial and trapped debris. Sci. Total Environ. 794, 148677. https://doi.org/10.1016/j.scitotenv.2021.148677 (2021).
Wang, Y. S. Molecular ecology of mangroves (The Science Press, 2019) (in Chinese).
Aslam, S., Mustaquim, J. & Siddiqui, G. First record of the polychaete worm Ceratonereis (Composetia) burmensis (Phyllodocida: Nereididae) from Pakistan. Pak. J. Sci. Ind. Res. Ser. B Biol. Sci. 63B(2), 132–134 (2020).
Aslam, S., Siddiqui, G., Dekker, H., Mustaquim, J. & Kazmi, S. J. H. Biodiversity on intertidal oyster reefs in the Hab River mouth: 35 new records from Pakistan. Reg. Stud. Mar. Sci. 39, 101415. https://doi.org/10.1016/j.rsma.2020.101415 (2020).
Guiry, M.D. & Guiry, G.M. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. Searched on YYYY-MM-DD., http://www.algaebase.org (2024).
Schram, F. R. Checklist of marine biota of China Seas. J. Crustac. Biol. 30(2), 339–339. https://doi.org/10.1651/09-3228.1 (2010).
Zenetos, A. et al. Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part I. Spatial distribution. Mediterr. Mar. Sci. 11(2), 381–493. https://doi.org/10.12681/mms.87 (2010).
Katsanevakis, S. et al. Building the European Alien Species Information Network (EASIN): A novel approach for the exploration of distributed alien species data. BioInvasions Rec. 1, 235–245 (2012).
Gabry, G., Wohltmann, A., & Mąkol, J. A redescription of Platytrombidium fasciatum (c. L. Koch, 1836) and Atractothrombium sylvaticum (c. L. Koch, 1835) (acari: Parasitengona: Microtrombidiidae) with notes on synonymy, biology and life cycle. In Annales zoologici (Vol. 55, No. 3) 477–496. (Museum and Institute of Zoology, Polish Academy of Sciences, 2005).
Khaustov, A. A. A new species of the genus Protopenthalodes (Acari: Penthalodidae) from Crimea. Acarina 23(2), 139 (2015).
Li, S. L., Liu, P. & Peng, X. J. Three new species of the genus Tetragnatha Latreille, 1804 (Araneae, Tetragnathidae) from China. ZooKeys 1125, 87–101. https://doi.org/10.3897/zookeys.1125.86905 (2022).
Liang, Y., Liu, J., Yin, H. & Xu, X. On new spider species of the genus Episinus (Araneae, Theridiidae) from China and proposal of five species groups. Biodivers. Data J. 13, e144222. https://doi.org/10.3897/BDJ.13.e144222 (2025).
Deng, Z., Agnarsson, I., Chen, Z. & Liu, J. Meotipa species (Araneae, Theridiidae) from China. ZooKeys 1082, 153–178. https://doi.org/10.3897/zookeys.1082.75400 (2022).
King, J. G., & Lago, P. K. 0275. The variegated mud-loving beetles (Coleoptera: Heteroceridae) of Mississippi and Alabama, with discussion and keys to the species occurring in the southeastern United States. Insecta Mundi, 1–53. http://digitalcommons.unl.edu/insectamundi/788 (2012).
Poon, D. Y., Chan, K. K. & Williams, G. A. Spatial and temporal variation in diets of the crabs Metopograpsus frontalis (Grapsidae) and Perisesarma bidens (Sesarmidae): Implications for mangrove food webs. Hydrobiologia 638, 29–40. https://doi.org/10.1007/s10750-009-0005-5 (2010).
Lee, S. Y. The brachyuran fauna of the Mai Po Marshes Nature Reserve and Deep Bay, Hong Kong. In: Proceedings of the International workshop on the mangrove ecosystem of Deep Bay and the Mai Po Marshes, Hong Kong (ed Lee, S. Y.) 57–82. (Hong Kong University Press, 1999).
Shih, H.-T. et al. Systematics of the family Ocypodidae Rafinesque, 1815 (Crustacea: Brachyura), based on phylogenetic relationships, with a reorganization of subfamily rankings and a review of the taxonomic status of Uca Leach, 1814, sensu lato and its subgenera. Raffles Bull. Zool. 64, 139–175 (2016).
Proud, A. J. An ecological survey of the D’ Aguilar Peninsula, Hong Kong, with recommendations for its future management. Mphil thesis. The University of Hong Kong (1977).
De Man, J. G. Uebersicht der indo-pacifischen Arten der Gattung Sesarma Say, nebst einer Kritik der von W. Hess und E. Nauck in den Jahren 1865 und 1880 beschriebenen Decapoden. Zool. Jahrb. Syst. Geogr. Biol. Thiere 2(3–4), 639–722 (1887).
Moscoso, V. Catálogo de crustáceos decápodos y estomatópodos del Perú. Boletín Instituto del Mar del Perú. 27(1–2), 8–207 (2012).
Tsuge, M. A new species of the genus Ligia (Crustacea: Isopoda: Ligiidae) from the Lake Shinji (Shimane Prefecture), western Japan. Bull. Toyama Sci. Mus. 31, 51–57 (2008).
Gregory, S. Woodlice and Waterlice (Isopoda: Oniscidea & Asellota) in Britain and Ireland. Field Studies Council/Centre for Ecology & Hydrology. https://bmig.org.uk/species/trichoniscus-pygmaeus (2009).
Palacios-Vargas, J. G. & Bu, Y. New records of Collembola from marine littoral sand of Hainan Island, China, with description of a new Oudemansia species (Neanuridae: Pseudachorutinae). Zootaxa 4810(1), 117–130. https://doi.org/10.11646/zootaxa.4810.1.6 (2020).
Zhou, R. & Ma, Y. New records and two new species of Entomobryinae (Collembola: Entomobryidae) from China. Orient. Insects 57(3), 756–784. https://doi.org/10.1080/00305316.2022.2123056 (2023).
Kaleka, A. S., Jallundhara, S. & Kapoor, Y. Scanning electron microscope studies on ornamentation of egg chorion of Capissa vagesa (Moore, 1859) (Erebidae) and Trabala vishnou (Lefèbvre, 1827) (Lasiocampidae) (Ditrysia: Lepidoptera) from India. J. Entomol. Res. Soc. 25(2), 337–349. https://doi.org/10.51963/jers.2023.92 (2023).
Munn, L. Y. Using daisy (Digital automated identification system) for automated identification of moths of the superfamily Bombycoidea of Borneo. University of Malaya (Malaysia) 1–73 (2013).
Medler, J. T. The types of Flatidae (Homoptera) in the Stockholm Museum described by Stat, Melichar, Jacobi and Walker. Insect Syst. Evol. 17(3), 323–337 (1986).
Wang, H. S., Wang, M. & Fan, X. L. Notes on the tribe Nygmiini (Lepidoptera: Erebidae: Lymantriinae) from nanling national nature reserve, with description of a new species. Zootaxa 2887(1), 57–68. https://doi.org/10.11646/zootaxa.2887.1.3 (2011).
Duan, Y. & Hu, X. The complete mitochondrial genome of the cocoa tussock moth Orgyia postica Walker, 1855 (Lepidoptera: Lymantriidae). Mitochondr. DNA B 6(12), 3498–3500. https://doi.org/10.1080/23802359.2021.2005474 (2021).
Schoch, C. L. et al. NCBI Taxonomy: a comprehensive update on curation, resources and tools. Database (Oxford). baaa062 (2020).
Dayrat, B. et al. Integrative taxonomy of the genus Onchidium Buchannan, 1800 (Mollusca, Gastropoda, Pulmonata, Onchidiidae). ZooKeys 636, 1–40. https://doi.org/10.3897/zookeys.636.8879 (2016).
Swennen, C. Large mangrove-dwelling Elysia species in Asia, with descriptions of two new species (Gastropoda: Opisthobranchia: Sacoglossa). Raffles Bull. Zool. 59(1), 29–37 (2011).
Adams, A. Monograph of the family Bullidae. In Thesaurus conchyliorum, or monographs of genera of shells (ed Sowerby II, G. B.) Vol. 2 (11): 553–608, pls 119–125. London. https://www.biodiversitylibrary.org/page/43936916 (1850).
Aslam, S., Oskars, T. R., Siddiqui, G. & Malaquias, M. A. E. Beyond shells: First detailed morphological description of the mangrove-associated gastropod Haminoea cf. fusca (A. Adams, 1850) (Cephalaspidea, Haminoeidae), with a COI phylogenetic analysis. Zoosystema 41(1), 313–326. https://doi.org/10.5252/zoosystema2019v41a16 (2019).
Oskars, T. R. & Malaquias, M. A. E. Systematic revision of the Indo-West Pacific mangrove-associated snails of the genus Bakawan (Cephalaspidea: Haminoeidae). J. Molluscan Stud. 86, 323–341. https://doi.org/10.1093/mollus/eyaa012 (2020).
Oskars, T. R. & Malaquias, M. A. E. Systematic revision of the Indo-West Pacific bubble-snails of the genus Haloa (Pilsbry, 1921) (Cephalaspidea: Haminoeidae). Invertebr. Syst. 36(5), 436–492. https://doi.org/10.1071/IS21011 (2022).
Naser, M. D., Yasser, A. G., Ajeel, S. G. & Auda, N. M. New record of the bubble snail Haloa wallisii (Gray, 1825) (Haminoeidae Pilsbry, 1895) from the breakwaters of the Iraqi coast, Persian Gulf. Munis Entomol. Zool. 19(suppl), 2699–2703 (2024).
Hasegawa K. Upper bathyal gastropods of the Pacific coast of northern Honshu, Japan, chiefly collected by R/V Wakataka-maru. In Deep-sea fauna and pollutants off Pacific coast of northern Japan. National Museum of Nature and Science Monographs (ed Fujita, T.) vol 39. 225–383 (2009).
Gassies, J.-B. Description de Mollusques terrestres et fluviatiles provenant de la Nouvelle-Calédonie. J. Conchyliol. 23, 227–232 (1875).
Zhang, S.-P. & Yang, J.-W. Revision of species names of several Nassarius in China Seas. Oceanol. Limnol. Sin. 41(5), 791–795 (2010).
Inoue, K., Yahagi, T., Kimura, T. & Kano, Y. Population differentiation of the endangered salt-marsh snail Ellobium chinense in Japan (Gastropoda: Ellobiidae). Plankton Benthos Res. 17(1), 66–75. https://doi.org/10.3800/pbr.17.66 (2022).
Ramírez, R., Paredes, C. & Arenas, J. Moluscos del Perú. Rev. Biol. Trop. 51(supplement 3), 225–284 (2003).
Raven, H. & Vermeulen, J. J. Notes on molluscs from NW Borneo and Singapore. 2. A synopsis of the Ellobiidae (Gastropoda, Pulmonata). Vita Malacol. 4, 29–62 (2007).
Brandt, R. A. M. The non-marine aquatic Mollusca of Thailand. Arch. Molluskenkund. 105, 1–423 (1974).
Glaubrecht, M., Brinkmann, N. & Pöppe, J. Diversity and disparity ‘down under’: Systematics, biogeography and reproductive modes of the ‘marsupial’ freshwater Thiaridae (Caenogastropoda, Cerithioidea) in Australia. Zoosyst. Evol. 85(2), 199–275. https://doi.org/10.1002/zoos.200900004 (2009).
Reid, D. G., Dyal, P. & Williams, S. T. Global diversification of mangrove fauna: A molecular phylogeny of Littoraria (Gastropoda: Littorinidae). Mol. Phylogenet. Evol. 55, 185–201. https://doi.org/10.1016/j.ympev.2009.09.036 (2010).
Reid, D. G. The comparative morphology, phylogeny and evolution of the gastropod family Littorinidae. Philos. Trans. R. Soc. B 324, 1–110. https://doi.org/10.1098/rstb.1989.0040 (1989).
Bartsch, P. Report on the Turton Collection of South African marine mollusks, with additional notes on other South African shells contained in the United States National Museum. Bullet. U. S. Natl. Museum 91, 305 (1915).
Fukuda H. & Hasegawa K. Family Assimineidae. pp. 813–816, in: T. Okutani (ed.), Marine Mollusks in Japan, ed. 2. 2 vols. 1375 pp (Tokai University Press, 2017).
Fukuda, H. & Ponder, W. F. Australian freshwater assimineids, with a synopsis of the recent genus-group taxa of the Assimineidae (Mollusca: Caenogastropoda: Rissooidea). J. Nat. Hist. 37, 1977–2032. https://doi.org/10.1080/00222930210125380 (2003).
Reid, D. G. & Ozawa, T. The genus Pirenella Gray, 1847 (= Cerithideopsilla Thiele, 1929) (Gastropoda: Potamididae) in the Indo-West Pacific region and Mediterranean Sea. Zootaxa 4076(1), 1–91. https://doi.org/10.11646/zootaxa.4076.1.1 (2016).
Reid, D. G. The genus Cerithidea Swainson, 1840 (Gastropoda: Potamididae) in the Indo-West Pacific region. Zootaxa 3775(1), 1–65. https://doi.org/10.11646/zootaxa.3775.1.1 (2014).
Huber M. Compendium of Bivalves 2. 1–907, Excel listing of species (ConchBooks, 2015).
Huber, M. Compendium of bivalves. A full-color guide to 3300 of the world’s marine bivalves. A status on Bivalvia after 250 years of research 901, 1 CD-ROM (ConchBooks, 2010).
Tan, K. S., Tan, S. H. M., Sanpanich, K., Duangdee, T. & Ambarwati, R. Xenostrobus or Vignadula (Bivalvia: Mytilidae)? A taxonomic re-evaluation of small black mussels inhabiting the upper intertidal zone of the estuaries of Southeast Asia. Zool. J. Linn. Soc. 20(1), 1–30. https://doi.org/10.1093/zoolinnean/zlac031 (2022).
Coan, E. V. & Kabat, A. R. The malacological works and taxa of Sylvanius Hanley (1819–1899). Malacologia 55(2), 285–359. https://doi.org/10.4002/040.055.0208 (2012).
Lim, J. Y. et al. Mytella strigata (Bivalvia: Mytilidae): An alien mussel recently introduced to Singapore and spreading rapidly. Molluscan Res. 38(3), 170–186. https://doi.org/10.1080/13235818.2018.1423858 (2018).
Yahya, N., Idris, I., Rosli, N. S. & Bachok, Z. Population dynamics of mangrove clam, Geloina expansa (Mousson, 1849) (Mollusca, Bivalvia) in a Malaysian mangrove system of South China Sea. J. Sustain. Sci. Manag. 13(5), 203–216 (2018).
Xie, S. F. et al. First report of leaf spot caused by Corynespora cassiicola on Acanthus ilicifolius in China. Plant Dis. 105(2), 509–509. https://doi.org/10.1094/PDIS-05-20-1021-PDN (2021).
Froese, R. & D. Pauly (eds). FishBase. World Wide Web electronic publication. version (06/2024). https://www.fishbase.org (2024).
Sievers, F. & Higgins, D. G. Clustal omega. Curr. Protoc. Bioinform. 48(1), 3–13. https://doi.org/10.1002/0471250953.bi0313s48 (2014).
Nguyen, L. T., Schmidt, H. A., Von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32(1), 268–274. https://doi.org/10.1093/molbev/msu300 (2015).
Bleeker, P. (1849). Bijdrage tot de kennis der Blennioïden en Gobioïden van den Soenda-Molukschen Archipel, metbeschrijving van 42 nieuwe soorten. Verhandelingen van het Bataviaasch Genootschap van Kunsten enWetenschappen. v. 22 [6]: 1-40.
Pepito, L. B. & Cabili, T. M. Mollusk species diversity in mangrove ecosystems of Northern Samar: Basis in the design of mangrove rehabilitation programs. J. Coast. Life Med. 11, 750–765 (2023).
Wei, M. et al. Biodiversity of mangrove mollusks in the west coast of Hainan Island, China. Biodivers. Sci. 26(7), 707. https://doi.org/10.17520/biods.2018104 (2018).
Bharate, M. et al. Description of a new species of Haminoea (Gastropoda: Cephalaspidea) from India, with an account of the diversity of the genus in the Indo-West Pacific. J. Nat. Hist. 52(37–38), 2437–2456. https://doi.org/10.1080/00222933.2018.1533598 (2018).
Bravo, H. et al. A DNA barcode library for mangrove gastropods and crabs of Hong Kong and the Greater Bay Area reveals an unexpected faunal diversity associated with the intertidal forests of Southern China. BMC Ecol. Evol. 21, 180. https://doi.org/10.1186/s12862-021-01914-6 (2021).
Khan, W. R. et al. Assessing the productivity of the matang mangrove forest reserve: Review of one of the best-managed mangrove forests. Forests 15(5), 747. https://doi.org/10.3390/f15050747 (2024).
Macintosh, D. J., Ashton, E. C. & Havanon, S. Mangrove rehabilitation and intertidal biodiversity: A study in the Ranong mangrove ecosystem, Thailand. Estuar. Coast. Shelf Sci. 55(3), 331–345. https://doi.org/10.1006/ecss.2001.0896 (2002).
Jaafar, Z., Lim, K. K. & Chou, L. M. Taxonomical and morphological notes on two species of mudskippers, Periophthalmus walailakae and Periophthalmodon schlosseri (Teleostei: Gobiidae) from Singapore. Zool. Sci. 23(11), 1043–1047. https://doi.org/10.2108/zsj.23.1043 (2006).
Hutchings, P. Biodiversity and functioning of polychaetes in benthic sediments. Biodivers. Conserv. 7(9), 1133–1145. https://doi.org/10.1023/A:1008871430178 (1998).
Lobban, C. S. & Harrison, P. J. Seaweed ecology and physiology (Cambridge University Press, 1994).
Zhang, Y., Li, Y., Shi, F., Sun, X. & Lin, G. Seasonal and spatial variation in species diversity, abundance, and element accumulation capacities of macroalgae in mangrove forests of Zhanjiang, China. Acta Oceanol. Sin. 33, 73–82. https://doi.org/10.1007/s13131-014-0414-9 (2014).
Nayak, A. et al. Macrobenthic community of an anthropogenically influenced mangrove associated estuary on the East coast of India: An approach for ecological assessment. Front. Mar. Sci. 9, 1008912. https://doi.org/10.3389/fmars.2022.1008912 (2022).
Thu, P. M. & Populus, J. Status and changes of mangrove forest in Mekong Delta: Case study in Tra Vinh, Vietnam. Estuar. Coast. Shelf Sci. 71(1–2), 98–109. https://doi.org/10.1016/j.ecss.2006.08.007 (2007).
Ghahramani, P. et al. Macrobenthos of natural vs. planted mangroves: Species compositions and functional perspective. Mar. Environ. Res. 212, 107525. https://doi.org/10.1016/j.marenvres.2025.107525 (2025).
Carugati, L. et al. Impact of mangrove forests degradation on biodiversity and ecosystem functioning. Sci. Rep. 8(1), 13298. https://doi.org/10.1038/s41598-018-31683-0 (2018).
Bracewell, S. A., Clark, G. F. & Johnston, E. L. Habitat complexity effects on diversity and abundance differ with latitude: An experimental study over 20 degrees. Ecology 99(9), 1964–1974. https://doi.org/10.1002/ecy.2408 (2018).
Witman, J. D., Etter, R. J. & Smith, F. The relationship between regional and local species diversity in marine benthic communities: A global perspective. Proc. Natl. Acad. Sci. U. S. A. 101(44), 15664–15669. https://doi.org/10.1073/pnas.0404300101 (2004).
Aji, L. P. et al. Shifts in dominance of benthic communities along a gradient of water temperature and turbidity in tropical coastal ecosystems. PeerJ 12, e17132. https://doi.org/10.7717/peerj.17132 (2024).
Engle, V. D. & Summers, J. K. Latitudinal gradients in benthic community composition in Western Atlantic estuaries. J. Biogeogr. 26(5), 1007–1023. https://doi.org/10.1046/j.1365-2699.1999.00341.x (1999).
Silberberger, M. J., Koziorowska‐Makuch, K., Reiss, H. & Kędra, M. Trophic niches of macrobenthos: Latitudinal variation indicates climate change impact on ecosystem functioning. Glob. Change Biol. 30(1), e17100. https://doi.org/10.1111/gcb.17100 (2024).
Mieszkowska, N. & Sugden, H. E. Climate-driven range shifts within benthic habitats across a marine biogeographic transition zone. In Advances in ecological research Vol. 55 325–369 (Academic Press, 2016). https://doi.org/10.1016/bs.aecr.2016.08.007.
Klunder, L. M. Marine benthic metabarcoding: anthropogenic effects on benthic diversity from shore to deep sea; assessed by metabarcoding and traditional taxonomy. Thesis fully internal (DIV), University of Groningen. https://doi.org/10.33612/diss.135301602 (2020).
Rivadeneira, M. M. & Nielsen, S. N. Deep anthropogenic impacts on benthic marine diversity of the Humboldt Current Marine Ecosystem: Insights from a Quaternary fossil baseline. Front. Mar. Sci. 9, 948580. https://doi.org/10.3389/fmars.2022.948580 (2022).
Magurran, A. E. Measuring biological diversity. Curr. Biol. 31(19), R1174–R1177 (2021).
Kathiresan, K. & Bingham, B. L. Biology of mangroves and mangrove ecosystems. Adv. Mar. Biol. 40, 81–251. https://doi.org/10.1016/S0065-2881(01)40003-4 (2001).
Satyanarayana, B. et al. Status of the undisturbed mangroves at Brunei Bay, East Malaysia: A preliminary assessment based on remote sensing and ground-truth observations. PeerJ 6, e4397. https://doi.org/10.7717/peerj.4397 (2018).
Clarke, K. R., Gorley, R. N., Somerfield, P. J. & Warwick, R. M. Change in marine communities: An approach to statistical analysis and interpretation (PRIMER-E Ltd., 2020).
Becking, L. E. et al. Beta diversity of tropical marine benthic assemblages in the Spermonde Archipelago, Indonesia. Mar. Ecol. 27(1), 76–88. https://doi.org/10.1111/j.1439-0485.2005.00051.x (2006).
Ellison, A. M. Managing mangroves with benthic biodiversity in mind: Moving beyond roving banditry. J. Sea Res. 59(1–2), 2–15. https://doi.org/10.1016/j.seares.2007.05.003 (2008).
Alongi, D. M. Present state and future of the world’s mangrove forests. Environ. Conserv. 29(3), 331–349. https://doi.org/10.1017/S0376892902000231 (2002).
Nagelkerken, I. S. J. M. et al. The habitat function of mangroves for terrestrial and marine fauna: A review. Aquat. Bot. 89(2), 155–185. https://doi.org/10.1016/j.aquabot.2007.12.007 (2008).
Lee, S. Y., Primavera, J. H., Dahdouh‐Guebas, F., McKee, K., Bosire, J. O., Cannicci, S., Diele, K., Fromard, F., Koedam,N., Marchand, C. and Mendelssohn, I., Mukherjee, N., & Record, S. (2014). Ecological role and services of tropicalmangrove ecosystems: a reassessment. Global ecology and biogeography, 23(7), 726-743.https://doi.org/10.1111/geb.12155
Duke, N. C. A systematic revision of the mangrove genus Avicennia (Avicenniaceae) in Australasia. Aust. Syst. Bot. 4(2), 299–324. https://doi.org/10.1071/SB9910299 (1991).
Saifullah, S.M., Nizamuddin, M., 1977. Studies of the marine algae from Pakistan: Ulvales. Bot. Mar. 20, 521–536.http://dx.doi.org/10.1515/botm.1977.20.8.521.
Husain, S.A., Shameel, M., 1997. Observations on Gelidium pusillum (Stackh.) Le Jolis (Rhodophyta) from the coastof Pakistan. Pak. J. Bot. 29, 85–190.
Kuntner M. 2017. Nephilidae. In: Ubick D., Paquin P., Cushing P.E., Roth V., editors. Spiders of North America: anidentifi cation manual. (2nd ed.) American Arachnological Society, p. 191–192.
Mustaquim, J., Mufti, S.A., Wood, C.A., Hasan, S.A., 1997. Marine worms (Polychaeta) of Pakistan. In: Biodiversity ofPakistan. Florida Museum of Natural History, Gainesville, 1-221-227.
Manson, F. J., Loneragan, N. R., Harch, B. D., Skilleter, G. A. & Williams, L. A broad-scale analysis of links between coastal fisheries production and mangrove extent: A case-study for northeastern Australia. Fish. Res. 74(1–3), 69–85. https://doi.org/10.1016/j.fishres.2005.04.001 (2005).
Acknowledgements
The authors are grateful for the following experts for species identification: Dr. Zhang Jing (Testing Centre of Marine Environment, South China Sea Institute of Oceanology, Chinese academy of sciences); Prof. Xiang Wenzhou (Phycology expert, South China Sea Institute of Oceanology, Chinese academy of sciences); Prof. Enyi Xie (Seaweed expert, Guangdong Ocean University); Prof. Chun Wang (Shanghai Ocean University) identified Crabs through the introduction of Prof. Jixing Zou (South China Agricultural University); Prof. Graham Oliver (Seashells expert, National Museum Wales Cardiff, United Kingdom); Prof. Koh Siang Tan (Asian mussels’ expert, Tropical Marine Science Institute, National University of Singapore); Dr. Trond Rogger Oskars (Molluscan expert, Senior Researcher at Møreforsking, Kristiansund, Norway); Prof. Joanna Mąkol (Parasitengonina expert, Wroclaw University of Environmental and Life Sciences, Poland) and Ronald Laniecki (Penthalodidae mites expert, Adam Mickiewicz University, Poznan, Poland).
Funding
This research was supported by the Nansha District High-Level Talent Innovation Team Project in 2021 (Mangrove Wetland Blue Carbon Sequestration Technology Innovation Team); Key Tasks Guarantee Special Fund Project for Green-Beautiful Guangdong Ecological Construction in 2024 (Precise Restoration and Ecological Function Evaluation of Mangroves); Strategic Priority Research Program of the Chinese Academy of Sciences (the Beautiful China Ecological Civilization Construction Science and Technology Project) (XDA23050200); National Natural Science Foundation of China (U1901211, 41876126); National Key Research and Development Program of China (2017FY100700).
Author information
Authors and Affiliations
Contributions
**S.A.** wrote the original manuscript, undertook laboratory work, field sampling and designed the research. **M.W.H.C.** carried out the conceptualization & visualization. **T. R. O.** contributed to the taxonomic identification for bubble snails, writing and proofreading. **G.C.** and **J.D.** contributed to the molecular work, writing and review-proofreading. **H.D.** contributed to the identification of mollusks and edited-proofread. **Y-S.W.** supervised the entire project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
In this study no live animal has been used. All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities. The study is compliant with CBD and Nagoya protocols.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Aslam, S., Chan, M.W.H., Oskars, T.R. et al. Biodiversity of natural versus planted mangrove forests across latitudes in Guangdong China with seven new records and potential new species. Sci Rep (2026). https://doi.org/10.1038/s41598-026-38032-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-38032-6