Abstract
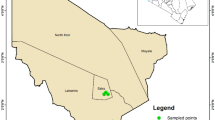
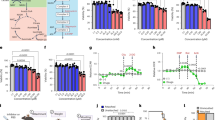
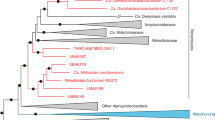
Amblyomma sculptum is a major tick species in southeastern Brazil and an important vector in the epidemiology of Brazilian Spotted Fever (BSF). This study characterized the bacterial microbiome of free-living A. sculptum ticks in a BSF-endemic area, focusing on differences among developmental stages and sexes, and investigated rickettsial agents using 16S rRNA gene (V3–V4) sequencing. A total of 154 ticks were collected and analysed as 13 pooled samples grouped by stage and sex. Sequencing identified a diverse bacterial community of 180 genera, dominated by Sphingomonas, Nocardioides, Actinomycetospora and Methylobacterium, alongside genera of potential zoonotic relevance such as Rickettsia, Anaplasma, Ehrlichia and Coxiella, mainly in nymph pools. Alpha and beta diversity analysis showed that microbial community composition differed among stages and sexes, with adult males exhibiting higher richness and compositional dispersion, whereas nymphs showed reduced diversity and tighter clustering. Because 16S sequencing does not allow species-level identification of rickettsiae, positive samples were further analysed by PCR amplification and sequencing of the gltA gene, which identified Rickettsia bellii. Together, these results highlight stage- and sex-associated patterns in the microbiome of free-living A. sculptum and support the use of integrated microbiome profiling and targeted rickettsial detection in ecological and epidemiological studies of tick-borne diseases.
Similar content being viewed by others
Data availability
Raw sequencing datasets generated in this study have been deposited in the NCBI Sequence Read Archive (SRA) under the BioProject accession number PRJNA1347853 (https://www.ncbi.nlm.nih.gov/bioproject/PRJNA1347853). The dataset includes paired-end FASTQ files corresponding to all 13 samples analyzed. In addition, the gltA amplicon sequence of Rickettsia bellii obtained in this study has been submitted to GenBank under accession number PX727355 and is also provided as Supplementary File 9 (FASTA format).
References
Narasimhan, S. & Fikrig, E. Tick microbiome: The force within. Trends Parasitol. (2015). https://doi.org/10.1016/j.pt.2015.03.010
Pfäffle, M., Littwin, N., Muders, S. V. & Petney, T. N. The ecology of tick-borne diseases. Int. J. Parasitol. 43, 1059–1077 (2013).
Barros Battesti, D. et al. Acarofauna de Importância Veterinária Parasitiformes - Ixodida (2024).
Nava, S. et al. Reassessment of the taxonomic status of amblyomma Cajennense (Fabricius, 1787) with the description of three new species, amblyomma tonelliae n. sp., amblyomma interandinum n. sp. and amblyomma patinoi n. sp., and reinstatement of amblyomma mixtum Koch, 1844, and amblyomma sculptum Berlese, 1888 (Ixodida: Ixodidae). Ticks Tick. Borne Dis 5 (2014).
Braig, H. R., Turner, B. D. & Perotti, M. A. Symbiotic rickettsia. in Insect Symbiosis vol. 3 (2008).
Guedes, E. et al. Detection of rickettsia rickettsii in the tick amblyomma Cajennense in a new Brazilian spotted fever-endemic area in the state of Minas Gerais. Mem Inst. Oswaldo Cruz 100 (2005).
Sangioni, L. A. et al. Rickettsial infection in animals and Brazilian spotted fever endemicity. Emerg Infect. Dis 11 (2005).
Gerardi, M. et al. Comparative susceptibility of different populations of amblyomma sculptum to rickettsia rickettsii. Front. Physiol. 10 (2019).
Brasil. Óbitos Por Febre Maculosa. Brasil, Regiões e Unidades Federadas (Infecção) 2007–2022 (2023).
Nakao, R. et al. A novel approach, based on BLSOMs (Batch learning Self-Organizing Maps), to the Microbiome analysis of ticks. ISME J. 7 (2013).
Budachetri, K. et al. An insight into the Microbiome of the amblyomma maculatum (Acari: Ixodidae). J Med. Entomol. 51 (2014).
Ponnusamy, L. et al. Diversity of rickettsiales in the Microbiome of the lone star tick, amblyomma Americanum. Appl. Environ. Microbiol. 80 (2014).
Thapa, S., Zhang, Y. & Allen, M. S. Bacterial microbiomes of Ixodes scapularis ticks collected from Massachusetts and Texas, USA. BMC Microbiol. 19 (2019).
van Treuren, W. et al. Variation in the microbiota of Ixodes ticks with regard to geography, species, and sex. Appl. Environ. Microbiol. 81 (2015).
Gall, C. A. et al. The bacterial Microbiome of dermacentor andersoni ticks influences pathogen susceptibility. ISME Journal 10 (2016).
Antão, S. C. et al. Interactions between microbiota and immunity shape pathogen acquisition and fitness in amblyomma spp. Ticks. Dev. Comp. Immunol. 172, 105493 (2025).
Egan, S. L. et al. Bacterial community profiling highlights complex diversity and novel organisms in wildlife ticks. Ticks Tick. Borne Dis. 11 (2020).
Greay, T. L. et al. Recent insights into the tick Microbiome gained through next-generation sequencing. Parasites Vectors 11 (2018).
Labruna, M. B. et al. Rickettsia species infecting amblyomma cooperi ticks from an area in the state of São Paulo, Brazil, where Brazilian spotted fever is endemic. J. Clin. Microbiol 42 (2004).
Prakash, J. A. J. et al. Molecular detection and analysis of spotted fever group rickettsia in patients with fever and rash at a tertiary care centre in Tamil Nadu, India. Pathog. Glob. Health 106 (2012).
de Paula, L. G. F. et al. Seasonal dynamics of Amblyomma sculptum: A review. Parasites Vectors (2022). https://doi.org/10.1186/s13071-022-05311-w
Sperling, J. L. et al. Comparison of bacterial 16S rRNA variable regions for Microbiome surveys of ticks. Ticks Tick. Borne Dis. 8, 453–461 (2017).
Pavanelo, D. B. et al. Comparative analysis of the midgut microbiota of two natural tick vectors of rickettsia rickettsii. Dev. Comp. Immunol. 106 (2020).
Binetruy, F. et al. Microbial community structure reveals instability of nutritional symbiosis during the evolutionary radiation of amblyomma ticks. Mol. Ecol. 29 (2020).
de Romano, D. M. M. et al. First molecular evidence of wolbachia occurrence in amblyomma sculptum (Acari: Ixodidae). Vet. Parasitol. 317 (2023).
Portillo, A. et al. Exploring the bacteriome in anthropophilic ticks: to investigate the vectors for diagnosis. PLoS ONE 14 (2019).
Dall’Agnol, B. et al. Molecular characterization of bacterial communities of two Neotropical tick species (Amblyomma aureolatum and Ornithodoros brasiliensis) using rDNA 16S sequencing. Ticks Tick. Borne Dis. 12, 101746 (2021).
Zhang, Y. K. et al. The bacterial Microbiome of field-collected dermacentor marginatus and dermacentor reticulatus from Slovakia. Parasit. Vectors 12 (2019).
Williams-Newkirk, A. J., Rowe, L. A., Mixson-Hayden, T. R. & Dasch, G. A. Characterization of the bacterial communities of life stages of free living lone star ticks (Amblyomma americanum). PLoS One 9 (2014).
Thinnabut, K., Rodpai, R., Sanpool, O., Maleewong, W. & Tangkawanit, U. Genetic diversity of tick (Acari: Ixodidae) populations and molecular detection of Anaplasma and Ehrlichia infesting beef cattle from upper-northeastern Thailand. Infect. Genet. Evol. 107 (2023).
Stanton, H. Prevalence of Ehrlichia and Rickettsia Within Ticks in Arkansas State Parks (University of Arkansas, 2023).
Garcia Ribeiro, M. et al. Pleural effusion-related nocardia otitidiscaviarum, Anaplasma platys and Ehrlichia canis coinfection in a dog. Braz. J. Microbiol. 54 (2023).
Shivaprasad, H. L. et al. Coxiella-like infection in psittacines and a Toucan. Avian Dis. 52 (2008).
Ullah, Q., Jamil, T., Saqib, M., Iqbal, M. & Neubauer, H. Q Fever—A Neglected Zoonosis. Microorganisms (2022). https://doi.org/10.3390/microorganisms10081530
Sahu, R. et al. Elsevier,. Coxiella and Q fever. in Molecular Medical Microbiology 1811–1847 (2024). https://doi.org/10.1016/B978-0-12-818619-0.00079-4
Körner, S., Makert, G. R., Ulbert, S., Pfeffer, M. & Mertens-Scholz, K. The prevalence of Coxiella burnetii in hard ticks in Europe and their role in Q fever transmission revisited—a systematic review. Front. Vet. Sci. (2021). https://doi.org/10.3389/fvets.2021.655715
Almeida, A. P. et al. Coxiella symbiont in the tick Ornithodoros rostratus (Acari: Argasidae). Ticks Tick. Borne Dis 3 (2012).
Sonenshine, D. E. & Stewart, P. E. Microbiomes of blood-feeding arthropods: Genes coding for essential nutrients and relation to vector fitness and pathogenic infections. A review. Microorganisms (2021). https://doi.org/10.3390/microorganisms9122433
Noda, H., Munderloh, U. G. & Kurtti, T. J. Endosymbionts of ticks and their relationship to wolbachia spp. And tick-borne pathogens of humans And animals. Appl. Environ. Microbiol. 63 (1997).
Tirloni, L. et al. A physiologic overview of the organ-specific transcriptome of the cattle tick rhipicephalus Microplus. Sci. Rep. 10 (2020).
Budachetri, K., Gaillard, D., Williams, J., Mukherjee, N. & Karim, S. A snapshot of the Microbiome of amblyomma tuberculatum ticks infesting the gopher tortoise, an endangered species. Ticks Tick. Borne Dis. 7 (2016).
Song, K. et al. Bacterial microbiota in unfed ticks (Dermacentor nuttalli) from Xinjiang detected through 16S rDNA amplicon sequencing and culturomics. Zoonoses (Ireland) 1 (2021).
Imirzalioglu, C., Hain, T., Hossain, H., Chakraborty, T. & Domann, E. Erythema caused by a localised skin infection with Arthrobacter Mysorens. BMC Infect. Dis 10 (2010).
Patil, V. S. et al. Enterobacillus tribolii gen. nov., sp. nov., a novel member of the family Enterobacteriaceae, isolated from the gut of a red flour beetle, tribolium castaneum. Antonie Van Leeuwenhoek Int. J. Gen. Mol. Microbiology 107 (2015).
Thapa, S., Zhang, Y. & Allen, M. S. Effects of temperature on bacterial Microbiome composition in Ixodes scapularis ticks. Microbiologyopen 8 (2019).
Zolnik, C. P., Prill, R. J., Falco, R. C., Daniels, T. J. & Kolokotronis, S. O. Microbiome changes through ontogeny of a tick pathogen vector. Mol. Ecol. 25 (2016).
McCoy, K. D., Léger, E. & Dietrich, M. Host specialization in ticks and transmission of tick-borne diseases: A review. Front. Cell. Infect. Microbiol. 4, (2013).
Bonnet, S. I., Binetruy, F., Hernández-Jarguín, A. M. & Duron, O. The tick microbiome: Why non-pathogenic microorganisms matter in tick biology and pathogen transmission. Front. Cell. Infect. Microbiol. (2017). https://doi.org/10.3389/fcimb.2017.00236
Lau, A. C. C. et al. The dynamics of the Microbiome in Ixodidae are shaped by tick ontogeny and pathogens in Sarawak, Malaysian Borneo. Microb. Genom. 9 (2023).
Maitre, A. et al. Rickettsia helvetica infection is associated with Microbiome modulation in Ixodes ricinus collected from humans in Serbia. Sci. Rep. 12 (2022).
Krawczak, F. S., Labruna, M. B., Hecht, J. A., Paddock, C. D. & Karpathy, S. E. Genotypic Characterization of Rickettsia bellii Reveals Distinct Lineages in the United States and South America. Biomed. Res. Int. (2018).
Sakai, R. K. et al. Experimental infection with rickettsia rickettsii in an amblyomma dubitatum tick colony, naturally infected by rickettsia bellii. Ticks Tick. Borne Dis. 5 (2014).
Szabó, M. P. J. et al. Density and behavior of Capybara (Hydrochoerus hydrochaeris) ticks (Acari: Ixodidae) amblyomma sculptum and amblyomma dubitatum with notes on rickettsia bellii infection: assessing human exposure risk. Ticks Tick. Borne Dis. 15, 102330 (2024).
Vianna, M. C. B. et al. Rickettsial spotted fever in Capoeirão village, Itabira, Minas Gerais, Brazil. Rev Inst. Med. Trop. Sao Paulo 50 (2008).
de Araújo, F. E. S. et al. Seasonal dynamics of amblyomma Cajennense (Fabricius, 1787) sensu stricto in a degraded area of the Amazon biome, with notes on rickettsia amblyommatis infection. Parasit. Vectors. 16, 391 (2023).
Prado, R. F. S. et al. Molecular detection of rickettsia spp. In free living ticks In military instruction areas In Southeast Brazil. Braz. Arch. Biol. Technol. 66 (2023).
Neves, L. C. et al. Detection of Rickettsia spp. in animals and ticks in Midwestern Brazil, where human cases of rickettsiosis were reported. Animals (2023).
Caixeta, B. T. et al. Survey of rickettsia spp. In ticks (Acari: Ixodidae) Infesting opossums (Didelphis albiventris) and capybaras (Hydrochoerus hydrochaeris) from an urban park In southeastern Brazil. Exp. Appl. Acarol. 92, 463–477 (2024).
Gomes, I. R. C. et al. Effect of environmental variables on the abundance of amblyomma ticks, potential vectors of rickettsia Parkeri in central Brazil. PLoS One. 19, e0301685 (2024).
Barbieri, A. R. M. et al. Rickettsia communities and their relationship with tick species within and around the National park of Iguaçu, Brazil. Exp. Appl. Acarol. 91, 339–358 (2023).
Perez, C. A. et al. Protocolo De Priorização De Áreas Para Prevenir a Ocorrência Da Febre Maculosa Brasileira (FEALQ, 2023).
Meira, A. M. D. et al. Febre Maculosa: Dinâmica Da Doença, Hospedeiros e VetoresPiracicaba (2013).
Labruna, M. B., Ogrzewalska, M., Martins, T. F., Pinter, A. & Horta, M. C. Comparative susceptibility of larval stages of amblyomma aureolatum, amblyomma cajennense, and rhipicephalus sanguineus to infection by rickettsia rickettsii. J. Med. Entomol. 45, 1156–1159 (2008).
Martins, L. et al. The distinct transcriptional response of the midgut of amblyomma sculptum and amblyomma aureolatum ticks to rickettsia rickettsii correlates to their differences in susceptibility to infection. Front. Cell. Infect. Microbiol 7 (2017).
Wilson, J. G., Kinzer, D. R., Sauer, J. R. & Hair, J. A. Chemo-attraction in the lone star tick (Acarina: Ixodidae). I. Response of different developmental stages to carbon dioxide administered via traps. J. Med. Entomol 9 (1972).
Gray, J. S. A carbon dioxide trap for prolonged sampling of Ixodes ricinus L. populations. Exp. Appl. Acarol. 1 (1985).
Barros-Battesti, D., Arzua, M. & Bechara, H. Carrapatos de importância médico-veterinária Da região neotropical: Um Guia ilustrado Para identificação de espécies (Universidad De São Paulo, 2006).
Martins, T. F., Onofrio, V. C., Barros-Battesti, D. M. & Labruna, M. B. Nymphs of the genus amblyomma (Acari: Ixodidae) of brazil: Descriptions, redescriptions, and identification key. Ticks Tick. Borne Dis 1 (2010).
Illumina Inc. 16S Metagenomic Sequencing Library Preparation - Preparing 16S Ribosomal RNA Gene Amplicons for the Illumina MiSeq System. 16S Metagenomic Sequencing Library Preparation Manual (2013).
Callahan, B. J. et al. DADA2: High-resolution sample inference from illumina amplicon data. Nat. Methods 13 (2016).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41 (2013).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215 (1990).
Drummond, A. J. et al. Geneious Version 6.0.6 (2011). http://www.geneious.comvol
Stenos, J. & Walker, D. H. The rickettsial outer-membrane protein A and B genes of rickettsia australis, the most divergent rickettsia of the spotted fever group. Int J. Syst. Evol. Microbiol 50 (2000).
Ortiz, F. T. et al. Revisiting the role of opossums (Marsupialia) as Sentinel for Brazilian spotted fever caused by rickettsia rickettsii. Am. J. Trop. Med. Hyg. tpmd250026 https://doi.org/10.4269/ajtmh.25-0026 (2025).
Edgar, R. C. Muscle5: High-accuracy alignment ensembles enable unbiased assessments of sequence homology and phylogeny. Nat. Commun. 13, 6968 (2022).
Cowling, D. W., Gardner, I. A. & Johnson, W. O. Comparison of methods for Estimation of individual-level prevalence based on pooled samples. Prev. Vet. Med. 39, 211–225 (1999).
Acknowledgements
Special thanks to technicians and postgraduate students at the Functional Genomics Center/Animal Biotechnology Laboratory at ESALQ-USP.
Funding
A.P.A. is supported by a research fellowship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil. L.L.C. is supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil.
Author information
Authors and Affiliations
Contributions
A.P.A. conceived, conducted the experiment and analyzed the results, C.T.M.G. jointly conducted the preparation of the sequencing library. L.L.C. conceived and supervised the work. C.P.G., I.S.G., T.R.S and L.G.C collaborated on interpretation and discussion of the results. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Almeida, A.P., Moncau-Gadbem, C.T., Goes, C.P. et al. Characterization of bacterial microbiome and molecular detection of rickettsiosis in free-living ticks (Amblyomma sculptum Berlese, 1888). Sci Rep (2026). https://doi.org/10.1038/s41598-026-38069-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-38069-7