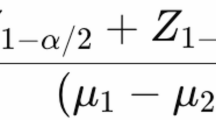Abstract
To compare 24-hour ambulatory blood pressure (ABP) between women with polycystic ovary syndrome (PCOS) and ovulatory controls, and to explore potential anthropometric, hormonal, metabolic, and inflammatory correlates of ABP in women with PCOS. In this cross-sectional study, 50 women with PCOS (diagnosed by Rotterdam criteria) and 50 ovulatory controls underwent office and 24-hour ABP monitoring. Clinical, anthropometric, hormonal, metabolic, and inflammatory parameters were assessed. Between-group comparisons were adjusted for body mass index (BMI). LASSO regression was used to identify variables independently associated with ABP in the PCOS group. Women with PCOS showed significantly higher 24-hour and daytime mean arterial pressure and heart rate compared to controls, even after adjustment for BMI (p < 0.05). No differences were observed in nighttime ABP or office blood pressure (p > 0.05). PCOS participants exhibited a more adverse cardiometabolic profile, including higher BMI, waist circumference, insulin, HbA1c, triglycerides, creatinine, and TNF-alfa, along with lower estradiol and progesterone levels. In LASSO models, BMI emerged as the only consistent independent predictor of ABP across all periods. Additional predictors, such as HbA1c (nighttime mean BP), creatinine (daytime diastolic BP), and waist circumference (daytime systolic BP), were retained in specific models, while most hormonal, metabolic, and inflammatory markers were not associated with ABP in the PCOS group. In summary, women with PCOS exhibit higher 24-hour and daytime ABP compared to ovulatory controls, independently of BMI. Adiposity, as assessed by BMI, appears to be a key factor associated with ABP in this population. These findings highlight the importance of 24-hour ABP monitoring and weight management in the cardiovascular risk assessment and care of women with PCOS.
Similar content being viewed by others
Introduction
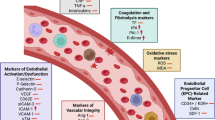
Polycystic ovary syndrome (PCOS) is a complex and the most common endocrine disorder among women of reproductive age, characterized by hyperandrogenism, ovulatory dysfunction, and polycystic ovarian morphology1,2. In addition to its well-established reproductive consequences, PCOS is strongly associated with multiple metabolic and cardiovascular disturbances, including obesity, chronic low-grade inflammation, insulin resistance, impaired glucose metabolism, dyslipidemia, metabolic syndrome, and type 2 diabetes, endothelial dysfunction, and hypertension3,4,5,6. These factors collectively contribute to an increased risk of major adverse cardiovascular events (MACE) in this population7,8.
Growing evidence indicates that women with PCOS have a higher prevalence of hypertension compared to their healthy ovulatory peers6,9,10,11,12. However, most studies have relied on office blood pressure measurements, which may not accurately reflect daily blood pressure behavior. In this context, 24-hour ambulatory blood pressure monitoring provides a more comprehensive assessment by capturing circadian patterns of blood pressure behavior during regular daily activities. Moreover, ambulatory blood pressure has been shown to be a stronger predictor of MACE than office blood pressure13,14. To date, few and conflicting studies have compared ambulatory blood pressure monitoring data between women with PCOS and ovulatory controls15,16,17,18. Additionally, little is known about factors associated with ambulatory blood pressure in women with PCOS. Therefore, this study aimed to (1) compare 24-hour, daytime, and nighttime ambulatory blood pressure between women with PCOS and ovulatory controls, and (2) explore potential anthropometric, hormonal, metabolic, and inflammatory correlates of ambulatory blood pressure in women with PCOS. Findings of this study may help identify relevant clinical, anthropometric, hormonal, metabolic, and inflammatory correlates of ambulatory blood pressure in women with PCOS, contributing to a better understanding of blood pressure regulation in this population.
Results
A total of 100 women were included in the final analysis, comprising 50 women with PCOS and 50 ovulatory controls (Table 1). There was no significant difference in age between groups (p = 0.860). However, women with PCOS had significantly higher BMI, waist circumference, total and free testosterone, HbA1c, fasting insulin, creatinine, triglycerides, and TNF-alfa (p < 0.05). They also showed a higher prevalence of acanthosis nigricans (p < 0.001). In contrast, estradiol and progesterone levels were significantly lower in the PCOS group (p < 0.05).
Table 2 presents the unadjusted comparisons of office and 24-hour ambulatory blood pressure between women with PCOS and ovulatory controls. Women with PCOS had significantly higher office systolic blood pressure (120 vs. 110 mmHg; p = 0.041) and a trend toward higher diastolic blood pressure (p = 0.079). Regarding ambulatory blood pressure, women with PCOS showed significantly higher 24-hour mean blood pressure (85 vs. 80 mmHg; p = 0.007) and diastolic blood pressure (70 vs. 65 mmHg; p = 0.012), as well as a trend toward higher systolic blood pressure (p = 0.076). Additionally, the PCOS group had a significantly higher heart rate (82 vs. 78 bpm; p = 0.015) and a trend toward higher pulse pressure (p = 0.072). During the daytime period, women with PCOS exhibited significantly higher values for mean (86 vs. 82 mmHg; p = 0.010), systolic (112 vs. 108 mmHg; p = 0.036), and diastolic (73 vs. 68 mmHg; p = 0.011) blood pressures, as well as heart rate (86 vs. 83 bpm; p = 0.021), with no significant difference observed for pulse pressure (p = 0.597). During the nighttime period, the PCOS group showed a trend toward higher values in mean (p = 0.080), systolic (p = 0.088), and diastolic (p = 0.062) blood pressures, as well as heart rate (p = 0.055), while pulse pressure did not differ between groups (p = 0.458).
Table 3 presents the BMI-adjusted comparisons of office and 24-hour ambulatory blood pressure between women with PCOS and ovulatory controls. No significant differences were observed for office systolic (p = 0.221) or diastolic (p = 0.383) blood pressure. In contrast, women with PCOS had significantly higher 24-hour mean blood pressure (84 vs. 80 mmHg; p = 0.032), as well as pulse pressure (41 vs. 38 mmHg; p = 0.029) and heart rate (81 vs. 78 bpm; p = 0.034). A trend toward higher diastolic blood pressure was also observed in this group (p = 0.090). During the daytime period, the PCOS group showed significantly higher mean blood pressure (85 vs. 82 mmHg; p = 0.032) and heart rate (85 vs. 81 bpm; p = 0.034), with trends toward higher systolic (p = 0.093) and diastolic (p = 0.067) blood pressures. During the nighttime period, no significant between-group differences were found; however, a trend toward higher heart rate was observed in the PCOS group (p = 0.078).
Among women with PCOS, LASSO regression identified BMI as the only independent predictor consistently associated with all 24-hour ambulatory blood pressure outcomes (Table 4). The estimated standardized coefficients were β = 0.35 for mean blood pressure, β = 0.83 for systolic blood pressure, and β = 0.06 for diastolic blood pressure. For daytime blood pressure, BMI also emerged as a relevant predictor across all outcomes: β = 0.43 for mean blood pressure, β = 0.83 for systolic blood pressure, and β = 0.17 for diastolic blood pressure. Additionally, creatinine was retained as a predictor for daytime diastolic pressure (β = 0.56), and waist circumference showed a marginal coefficient for daytime systolic pressure (β = 0.002). During nighttime, BMI remained associated with both mean (β = 0.40) and systolic blood pressure (β = 0.80), while HbA1c was retained as a predictor of nighttime mean blood pressure (β = 0.37). No predictors were retained for nighttime diastolic pressure. All other evaluated predictors, including fasting insulin, triglycerides, estradiol, progesterone, total and free testosterone, and TNF-alfa, were excluded from the final LASSO models. RMSE values ranged from 7.6 mmHg to 11.5 mmHg across different ambulatory blood pressure outcomes, indicating moderate predictive error.
Discussion
This study aimed to compare ambulatory blood pressure between women with PCOS and ovulatory controls, and to identify predictors of ambulatory blood pressure in the PCOS group. The main findings demonstrated that women with PCOS had significantly higher 24-hour and daytime mean blood pressure compared to controls, even after adjusting for BMI. Additionally, BMI emerged as the most consistent predictor of ambulatory blood pressure across all time periods (24-hour, daytime, and nighttime) among women with PCOS.
Regarding blood pressure comparisons, our findings align with previous studies showing elevated ambulatory blood pressure in women with PCOS compared to ovulatory controls15,18. However, other studies have reported conflicting results16,17. BMI-adjusted analysis revealed no significant differences in office blood pressure values between women with PCOS and their healthy ovulatory peers. The higher 24-hour blood pressure levels observed reinforce the importance of ambulatory monitoring, as traditional office measurements may mask impairments on blood pressure regulation under resting conditions. Interestingly, significantly higher ambulatory blood pressure values in women with PCOS were found during the daytime but not nighttime period, suggesting potential differences in circadian regulation of blood pressure in this population. In addition, in 24-hour and daytime periods, but not during nighttime, women with PCOS showed higher heart rate values compared to the ovulatory group. This finding may reflect altered cardiac autonomic regulation, potentially involving increased sympathetic activity, which has been reported in women with PCOS19,20 and could contribute to higher ambulatory blood pressure values during the daytime period. Nevertheless, as no direct measures of autonomic function were available, this interpretation remains speculative and should be considered hypothesis-generating. Our findings align with evidence suggesting that while obesity is associated with sustained 24-hour elevations17, the PCOS phenotype itself often maintains a preserved nocturnal “dipping” pattern18, indicating that circadian blood pressure regulation remains functional in these patients despite the increased daytime workload.
The PCOS group also presented a distinct anthropometric, hormonal, metabolic, and inflammatory profile, with significantly higher BMI, waist circumference, fasting insulin, HbA1c, triglycerides, creatinine, total and free testosterone, and TNF-alfa, along with lower estradiol and progesterone levels. Together, these factors contribute to an increased cardiometabolic risk profile, with a potential to impair the blood pressure regulation. In the exploratory LASSO regression models restricted to the PCOS group, BMI emerged as the only consistent independent predictor of ambulatory blood pressure across all outcomes. A small number of additional variables were retained in specific models, including HbA1c for nighttime mean blood pressure, creatinine for daytime diastolic blood pressure, and waist circumference marginally for daytime systolic blood pressure, whereas most metabolic, hormonal, and inflammatory markers were excluded through penalization. The preselection of candidate variables based on between-group differences may have excluded biologically relevant predictors that do not differ at baseline but could still contribute to within-group blood pressure variability. This pattern may reflect the dominant role of adiposity as an integrative determinant of blood pressure in women with PCOS, potentially capturing the shared variance of multiple correlated cardiometabolic factors. The exclusion of other markers in penalized models should not be interpreted as evidence of biological irrelevance, but rather as a consequence of regularization in the presence of correlated predictors and a modest sample size. Moreover, given the limited predictive performance and the restricted set of candidate variables, these findings should be viewed as hypothesis-generating and warrant confirmation in larger, independent cohorts.
From a clinical perspective, our findings demonstrated that ambulatory blood pressure monitoring detected differences between women with PCOS and controls that were not captured by resting office blood pressure measurements. This highlights the value of ambulatory blood pressure monitoring in the clinical evaluation of women with PCOS, particularly those with overweight or obesity. The approximately 3 mmHg higher 24-hour and daytime mean blood pressure observed in women with PCOS may be clinically relevant at population level. Large meta-analyses indicate that ambulatory blood pressure has a continuous association with all-cause and cardiovascular mortality and provides superior prognostic information compared with office blood pressure13,14,21. Thus, small but sustained increases in ambulatory mean blood pressure, such as those observed in our study, may be relevant for cardiovascular risk stratification, while individual-level interpretation should remain cautious. The consistent association between BMI and ambulatory blood pressure across 24-hour periods suggests that lifestyle interventions targeting weight management may help reduce the risk of blood pressure alterations and future hypertension in this population. Furthermore, our study reinforces the well-established adverse cardiometabolic profile of women with PCOS, including a wide range of anthropometric, hormonal, metabolic, and inflammatory markers that collectively increase the risk of cardiovascular disease. In this context, a large retrospective cohort study8 with an average follow-up of four years reported a higher incidence of MACE, including myocardial infarction, stroke, angina, revascularization procedures, and cardiovascular mortality, among young women with PCOS (median age ~ 29 years) compared to age-, BMI-, and primary care practiced-matched controls. Taken together, these findings underscore the importance of incorporating cardiovascular risk assessment and management into clinical practice for women with PCOS.
This study presents strengths and limitations. Among the strengths, we highlight the use of 24-hour ambulatory blood pressure monitoring in addition to office measurements, the adoption of strict inclusion criteria for both PCOS and control groups, and the application of LASSO regression to identify independent predictors within a well-characterized cohort that included anthropometric, hormonal, metabolic, and inflammatory markers. However, some limitations should be acknowledged. The cross-sectional design limits causal inference. Although the sample size was adequate for between-group comparisons, it may have limited the performance of the exploratory LASSO regression models. Hormonal measurements were not standardized according to menstrual cycle phase, which may have influenced comparisons of reproductive hormones. In addition, relevant behavioral factors such as physical activity, dietary intake, and sleep quality were not assessed, raising the possibility of residual confounding; however, all participants were non-smoker, minimizing confounding by smoking. The modest predictive performance of the LASSO models suggests that additional unmeasured factors may contribute to ambulatory blood pressure regulation in women with PCOS. Finally, the single-center nature of the sample may limit generalizability.
Conclusion
Women with PCOS exhibited higher 24-hour ambulatory blood pressure than ovulatory controls, largely driven by daytime elevations, even after BMI adjustment. These findings support the use of ambulatory blood pressure for cardiovascular risk stratification in this population. In exploratory analyses, BMI was the most consistent correlate of ambulatory blood pressure, underscoring the potential role of excess adiposity. This association is hypothesis-generating and warrants confirmation in larger studies.
Methods
Study design
This was a cross-sectional study designed to compare 24-hour ambulatory blood pressure between women with syndrome PCOS and ovulatory controls. The study was conducted at gynecology outpatient clinics of Januário Cicco Maternity School (MEJC) and the endocrinology outpatient clinics of Onofre Lopes University Hospital (HUOL), both affiliated with the Federal University of Rio Grande do Norte (UFRN), Natal-RN, Brazil. Both outpatient clinics serve as regional referral centers for the diagnosis and management of PCOS within the Brazilian Unified Health System (SUS) in Natal, Brazil.
Participants
Participants were women aged 18 to 44 years who had been diagnosed with PCOS from the gynecology outpatient clinics of MEJC or the endocrinology outpatient clinics of HUOL. Inclusion criteria: PCOS was diagnosed according to the Rotterdam criteria22, which require the presence of at least two out of three features after the exclusion of other etiologies, such as congenital adrenal hyperplasia, androgen-secreting tumors, and Cushing’s syndrome. The three diagnostic features include: (1) oligo-ovulation or anovulation (menstrual cycles longer than 35 days or fewer than 8 cycles per year); (2) clinical and/or biochemical signs of hyperandrogenism, including hirsutism and/or elevated serum androgen levels (e.g., total or free testosterone, androstenedione, or DHEAS); and (3) polycystic ovarian morphology on ultrasound, defined as the presence of ≥ 20 follicles measuring 2–9 mm in diameter and/or an ovarian volume greater than 10 cm3 in at least one ovary. Transvaginal ultrasound was preferred when feasible, with transabdominal ultrasound used for participants who were not sexually active. The control group consisted of ovulatory women with regular menstrual cycles (21–35 days) and no clinical or biochemical signs of hyperandrogenism or polycystic ovarian morphology on ultrasound. Both groups included non-smoking women without use of hormonal contraceptives at the time of the study and without diagnosis of hypertension. Eligible participants underwent a structured evaluation following the initial screening. This evaluation included medical history, blood sampling, physical examination, anthropometric measurements (weight, height, waist circumference), and 24-hour ambulatory blood pressure monitoring. The study protocol was approved by the Research Ethics Committee of HUOL/UFRN (approval number 400/2009). All participants provided written informed consent prior to enrollment. All methods were performed in accordance with relevant guidelines and regulations, including the Declaration of Helsinki, the Brazilian National Health Council Resolution, and institutional requirements.
Ambulatory blood pressure monitoring
Ambulatory blood pressure monitoring was conducted using a validated oscillometric device (Spacelabs Healthcare 90121, Software Version 03.016a, 2010), which records multiple blood pressure readings over a 24-hour period. Measurements were taken every 20 min. For device programming purposes, daytime was initially defined as 06:00 a.m. to 10:00 p.m. and nighttime as 10:01 p.m. to 05:59 a.m. However, for analytical purposes, the classification of daytime and nighttime periods was based on participants’ self-reported sleep diaries. In cases of discrepancy between predefined clock-time intervals and diary-reported sleep and wake times, the diary information was prioritized. For ambulatory blood pressure monitoring data to be considered valid, a minimum of 16 daytime and 8 nighttime readings was required. All procedures adhered to the recommendations of the Brazilian guidelines on ambulatory blood pressure monitoring23.
Office blood pressure
Office blood pressure was measured in triplicate after a 10-minute rest period, with a 1-minute interval between measurements, in a seated position. Measurements were obtained using the auscultatory method by an experienced evaluator, following the recommendations of the Brazilian guidelines on hypertension24. The average of the last two blood pressure readings was used for data analysis.
Anthropometric measures
Anthropometric measures included weight (kg), height (m), and waist circumference (cm). BMI (kg/m²) was calculated as weight divided by the square of height.
Hormonal, metabolic, and inflammatory analyses
Blood samples were collected in collaboration with an accredited clinical laboratory, following standardized procedures. All samples were obtained in the morning after a 12-hour overnight fast. A total of 20 mL of peripheral venous blood was collected. Hormonal markers included dehydroepiandrosterone sulfate (DHEAS), ultrasensitive thyroid-stimulating hormone (TSH), free thyroxine (free T4), luteinizing hormone (LH), follicle-stimulating hormone (FSH), estradiol, sex hormone-binding globulin (SHBG), total and free testosterone, progesterone, and prolactin. Blood sampling was performed without formal standardization according to menstrual cycle phase. In women with PCOS, many of whom were oligo- or anovulatory, samples were collected independent of cycle phase, reflecting routine clinical practice. Metabolic markers included glycemic and insulin responses measured at fasting and at 30, 60, 90, and 120 min using enzymatic and electrochemiluminescence methods; glycated hemoglobin (HbA1C), analyzed by high-performance liquid chromatography; lipid profile, including triglycerides, total cholesterol, HDL cholesterol, and LDL cholesterol (calculated using Martin’s formula); and renal function markers, such as urea and creatinine. Inflammatory markers included tumor necrosis factor-alpha (TNF-alfa), interleukin-6 (IL-6), and C-reactive protein (CRP). All laboratory analyses were performed using validated protocols to ensure the accuracy, reliability, and reproducibility of the results.
Sample size
The sample size was calculated based on data from Luque-Ramírez et al.18, considering a statistical power of 80% and a significance level of 5%. To detect a 24-hour systolic blood pressure difference of 5 ± 9 mmHg between women with PCOS and ovulatory controls, 52 participants are required in each group.
Statistical analysis
The normality of continuous variables was assessed using the Shapiro-Wilk test and z-scores for skewness and kurtosis. Variables with normal distribution were expressed as mean ± standard deviation, and those without as median and interquartile range (25th-75th percentiles). Categorical variables were reported as absolute (n) and relative (%) frequencies. Unadjusted comparisons between women with PCOS and controls were performed using the independent t-test or Mann-Whitney U test, as appropriate. Homogeneity of variances was verified with Levene’s test. Comparisons of office and ambulatory blood pressure between women with PCOS and controls, adjusted for BMI category (≥ 25 kg/m²), were conducted using generalized linear models (GzLM). Estimated marginal means (with 95% CI) were reported for adjusted values, and parameter estimates (with 95% CI) were presented for between-group differences. A p-value < 0.05 was considered statistically significant. Finally, we used Least Absolute Shrinkage and Selection Operator (LASSO) regression models to identify variables associated with ambulatory blood pressure outcomes (24-hour, daytime, and nighttime) based on penalized regression, exclusively in the PCOS group. We selected LASSO due to its ability to handle multicollinearity and perform variable selection in models with potentially correlated predictors (e.g., BMI and waist circumference). This step was exploratory in nature and aimed to identify potential predictors for hypothesis generation in future studies. Accordingly, the LASSO analyses were intended as exploratory and hypothesis-generating rather than confirmatory. Predictor variables were selected based on those that differed significantly between women with PCOS and ovulatory controls in univariate analyses (p < 0.05). All variables were standardized prior to modeling. Model performance was evaluated using the root mean squared error (RMSE) from 10-fold cross-validation. The lambda (λ) value that minimized prediction error was selected for the final model. Variables with non-zero coefficients in the final model were considered retained predictors. The analyses were performed in JAMOVI (Version 2.3) using the JMV 2.3.1 and GAMLJ 2.6.6 modules. The LASSO regression analyses were conducted in R version 2025.05.1 + 513 (R Foundation for Statistical Computing, Vienna, Austria) using the “glmnet” package.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Teede, H. J. et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil. Steril. 110, 364–379. https://doi.org/10.1016/j.fertnstert.2018.05.004 (2018).
Azziz, R. et al. The androgen excess and PCOS society criteria for the polycystic ovary syndrome: the complete task force report. Fertil. Steril. 91, 456–488. https://doi.org/10.1016/j.fertnstert.2008.06.035 (2009).
Sprung, V. S. et al. Endothelial function measured using flow-mediated dilation in polycystic ovary syndrome: a meta‐analysis of the observational studies. Clin. Endocrinol. (Oxf). 78, 438–446. https://doi.org/10.1111/j.1365-2265.2012.04490.x (2013).
Bril, F. et al. Adipose tissue dysfunction in polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 109, 10–24. https://doi.org/10.1210/clinem/dgad356 (2023).
Lim, S. S. et al. Metabolic syndrome in polycystic ovary syndrome: a systematic review, meta-analysis and meta‐regression. Obes. Rev. 20, 339–352. https://doi.org/10.1111/obr.12762 (2019).
Wekker, V. et al. Long-term cardiometabolic disease risk in women with PCOS: a systematic review and meta-analysis. Hum. Reprod. Update. 26, 942–960. https://doi.org/10.1093/humupd/dmaa029 (2020).
Tay, C. T. et al. 2023 International evidence-based polycystic ovary syndrome guideline update: insights from a systematic review and meta‐analysis on elevated clinical cardiovascular disease in polycystic ovary syndrome. J. Am. Heart Assoc. 13 https://doi.org/10.1161/JAHA.123.033572 (2024).
Berni, T. R., Morgan, C. L. & Rees, D. A. Women with polycystic ovary syndrome have an increased risk of major cardiovascular events: a population study. J. Clin. Endocrinol. Metab. 106, e3369–e3380. https://doi.org/10.1210/clinem/dgab392 (2021).
Amiri, M., Ramezani Tehrani, F., Behboudi-Gandevani, S., Bidhendi-Yarandi, R. & Carmina, E. Risk of hypertension in women with polycystic ovary syndrome: a systematic review, meta-analysis and meta-regression. Reprod. Biol. Endocrinol. 18 (23). https://doi.org/10.1186/s12958-020-00576-1 (2020).
Wu, C. H. et al. Hypertension risk in young women with polycystic ovary syndrome: A nationwide population-based cohort study. Front. Med. 7 https://doi.org/10.3389/fmed.2020.574651 (2020).
Azevedo, M. F. & de Elevated blood pressure in women with polycystic ovary syndrome: prevalence and associated risk factors. Rev. Bras. Ginecol. E Obs. 33, 31–36. https://doi.org/10.1590/s0100-72032011000100005 (2011). CostaE.C.Oliveira, A.I.N.; Silva, I.B. de O.; Marinho, J.C.D.B.; Rodrigues, J.A.M.; Azevedo.
Macut, D. et al. Hypertension in polycystic ovary syndrome: novel insights. Curr. Hypertens. Rev. 16, 55–60. https://doi.org/10.2174/1573402115666190531071422 (2020).
Yang, W. Y. et al. Association of office and ambulatory blood pressure with mortality and cardiovascular outcomes. JAMA 322, 409. https://doi.org/10.1001/jama.2019.9811 (2019).
Zhang, D. Y. et al. Ambulatory blood pressure monitoring, European guideline targets, and cardiovascular outcomes: an individual patient data meta-analysis. Eur. Heart J. 46, 2974–2987. https://doi.org/10.1093/eurheartj/ehaf220 (2025).
Holte, J., Gennarelli, G., Berne, C., Bergh, T. & Lithell, H. Elevated ambulatory day-time blood pressure in women with polycystic ovary syndrome: a sign of a pre-hypertensive state? Hum. Reprod. 11, 23–28. https://doi.org/10.1093/oxfordjournals.humrep.a019028 (1996).
Sampson, M., Kong, C., Patel, A., Unwin, R. & Jacobs, H. S. Ambulatory blood pressure profiles and plasminogen activator inhibitor (PAI-1) activity in lean women with and without the polycystic ovary syndrome. Clin. Endocrinol. (Oxf). 45, 623–629. https://doi.org/10.1046/j.1365-2265.1996.00863.x (1996).
Luque-Ramírez, M., Álvarez-Blasco, F., Mendieta-Azcona, C., Botella-Carretero, J. I. & Escobar-Morreale, H. F. Obesity is the major determinant of the abnormalities in blood pressure found in young women with the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 92, 2141–2148 (2007). 10.1210/jc.2007 – 0190.
Luque-Ramírez, M. et al. Office blood pressure, ambulatory blood pressure Monitoring, and echocardiographic abnormalities in women with polycystic ovary syndrome. Hypertension 63, 624–629. https://doi.org/10.1161/HYPERTENSIONAHA.113.02468 (2014).
Gui, J. & Wang, R. Cardiovascular autonomic dysfunction in women with polycystic ovary syndrome: a systematic review and meta-analysis. Reprod. Biomed. Online. 35, 113–120. https://doi.org/10.1016/j.rbmo.2017.03.018 (2017).
de Sá, J. C. F. et al. Analysis of heart rate variability in polycystic ovary syndrome. Gynecol. Endocrinol. 27, 443–447. https://doi.org/10.3109/09513590.2010.501881 (2011).
Soleimani, H. et al. Predictive accuracy of 24-hour ambulatory blood pressure monitoring versus clinic blood pressure for cardiovascular and all-cause mortality: a systematic review and meta-analysis. Curr. Hypertens. Rev. 21, 102–116. https://doi.org/10.2174/0115734021337639250203175636 (2025).
Rotterdam, E. S. H. R. E. & ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 19, 41–47. https://doi.org/10.1093/humrep/deh098 (2004).
Grupo de Trabalho, M. A. P. A. & Grupo de Trabalho, M. R. P. A. V Brazilian guidelines for ambulatory monitoring of arterial pressure and III Brazilian guidelines for home monitoring of blood pressure. J. Bras. Nefrol. 33, 365–388. https://doi.org/10.1590/S0101-28002011000300013 (2011).
Barroso, W. K. S. et al. Jr. et al. Brazilian guidelines of hypertension – 2020. Arq. Bras. Cardiol. 116, 516–658. https://doi.org/10.36660/abc.20201238 (2021).
Acknowledgements
The authors would like to thank the PCOS and control women who volunteered to participate in this study and Professor PhD. George Dantas Azevedo.
Author information
Authors and Affiliations
Contributions
MFA, GDA and ECC, conceptualization of research project; MFA, AKCR, LMBM, LMBM, and RLAC investigation and data curation; MFA and ECC formal analysis and main manuscript text; GDA and ECC, writing—Review & Editing. ECC, supervision; MFA, project administration; All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
de Fátima Azevedo, M., Rocha, A.K.C., de Melo, L.M.B. et al. 24-hour ambulatory blood pressure and associated factors in women with polycystic ovary syndrome compared with ovulatory controls. Sci Rep 16, 7215 (2026). https://doi.org/10.1038/s41598-026-38731-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-38731-0