Abstract
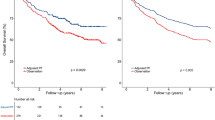
While surgical intervention administered after neoadjuvant therapy represents the standard treatment in esophageal cancer, the survival benefit conferred by adjuvant therapy administered postoperatively among this patient cohort continues to be controversial. Using real-world data, 6141 esophageal cancer patients administered neoadjuvant treatment preoperatively between 2007 and 2021 were categorized into adjuvant therapy group (n = 1116) or no adjuvant therapy group(n = 5025). No significant differences in overall survival (OS) or cancer-specific survival (CSS) were observed between groups before or after propensity score matching (PSM), and adjuvant therapy was not identified as an independent prognostic factor. However, subgroup analyses revealed important variations: patients treated in 2015–2021 derived greater survival benefit from adjuvant therapy than those in 2007–2014; female patients experienced worse CSS with adjuvant therapy, which was identified as an independent risk factor; among patients receiving preoperative systemic therapy alone, continuing systemic therapy postoperatively showed superior outcomes compared with radiotherapy or combined treatment; and among patients receiving preoperative chemoradiotherapy, no significant differences were observed across different postoperative adjuvant strategies. The findings suggest that while postoperative adjuvant therapy does not provide an overall survival advantage, significant subgroup heterogeneity exists, indicating that clinical decisions should be individualized based on diagnosis year, gender, and preoperative treatment regimen.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available in the Surveillance, Epidemiology, and End Results (SEER) repository.
References
Yang, H., Wang, F., Hallemeier, C. L., Lerut, T. & Fu, J. Oesophageal cancer. Lancet 404, 1991–2005. https://doi.org/10.1016/s0140-6736(24)02226-8 (2024).
Shah, M. A. et al. Treatment of locally advanced esophageal carcinoma: ASCO guideline. J. Clin. Oncol. 38, 2677–2694. https://doi.org/10.1200/jco.20.00866 (2020).
Wang, Y., Yang, W., Wang, Q. & Zhou, Y. Mechanisms of esophageal cancer metastasis and treatment progress. Front. Immunol. 14, 1206504. https://doi.org/10.3389/fimmu.2023.1206504 (2023).
Eyck, B. M. et al. Ten-year outcome of neoadjuvant chemoradiotherapy plus surgery for esophageal cancer: The randomized controlled CROSS trial. J. Clin. Oncol. 39, 1995–2004. https://doi.org/10.1200/jco.20.03614 (2021).
van Hagen, P. et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl. J. Med. 366, 2074–2084. https://doi.org/10.1056/NEJMoa1112088 (2012).
Swisher, S. G. et al. Improved long-term outcome with chemoradiotherapy strategies in esophageal cancer. Ann. Thorac. Surg. 90, 892–898. https://doi.org/10.1016/j.athoracsur.2010.04.061 (2010). discussion 898–899.
Lee, Y. et al. Role of adjuvant therapy in esophageal cancer patients after neoadjuvant therapy and esophagectomy: A systematic review and meta-analysis. Ann. Surg. 275, 91–98. https://doi.org/10.1097/sla.0000000000005227 (2022).
Lin, H. N., Chen, L. Q., Shang, Q. X., Yuan, Y. & Yang, Y. S. A meta-analysis on surgery with or without postoperative radiotherapy to treat squamous cell esophageal carcinoma. Int. J. Surg. 80, 184–191. https://doi.org/10.1016/j.ijsu.2020.06.046 (2020).
Mokdad, A. A. et al. Adjuvant chemotherapy vs postoperative observation following preoperative chemoradiotherapy and resection in gastroesophageal cancer: A propensity score-matched analysis. JAMA Oncol. 4, 31–38. https://doi.org/10.1001/jamaoncol.2017.2805 (2018).
Chen, H. et al. Postoperative adjuvant therapy for resectable thoracic esophageal squamous cell carcinoma: A retrospective analysis of 426 cases. Med. Oncol. 32, 417. https://doi.org/10.1007/s12032-014-0417-6 (2015).
Hsu, P. K. et al. Pre- versus postoperative chemoradiotherapy for locally advanced esophageal squamous cell carcinoma. J. Thorac. Cardiovasc. Surg. 154, 732–740. https://doi.org/10.1016/j.jtcvs.2017.03.038 (2017). .e732.
Zheng, S. Y., Qi, W. X., Zhao, S. G. & Chen, J. Y. No survival benefit could be obtained from adjuvant radiotherapy in esophageal cancer treated with neoadjuvant chemotherapy followed by surgery: A SEER-based analysis. Front. Oncol. 12, 897476. https://doi.org/10.3389/fonc.2022.897476 (2022).
Pasquali, S. et al. Survival after neoadjuvant and adjuvant treatments compared to surgery alone for resectable esophageal carcinoma: A network meta-analysis. Ann. Surg. 265, 481–491. https://doi.org/10.1097/sla.0000000000001905 (2017).
Jack, S. et al. The effect of neoadjuvant chemotherapy on physical fitness and survival in patients undergoing oesophagogastric cancer surgery. Eur. J. Surg. Oncol. 40, 1313–1320. https://doi.org/10.1016/j.ejso.2014.03.010 (2014).
Kim, G. J. et al. The benefit of chemotherapy in esophageal cancer patients with residual disease after trimodality therapy. Am. J. Clin. Oncol. 39, 136–141. https://doi.org/10.1097/coc.0000000000000036 (2016).
Darby, S. C. et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl. J. Med. 368, 987–998. https://doi.org/10.1056/NEJMoa1209825 (2013).
Shapiro, J. et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): Long-term results of a randomised controlled trial. Lancet Oncol. 16, 1090–1098. https://doi.org/10.1016/s1470-2045(15)00040-6 (2015).
Kakeji, Y. et al. Multimodality approaches to control esophageal cancer: Development of chemoradiotherapy, chemotherapy, and immunotherapy. Esophagus 18, 25–32. https://doi.org/10.1007/s10388-020-00782-1 (2021).
Low, D. E. et al. Guidelines for perioperative care in esophagectomy: Enhanced recovery after surgery (ERAS(®)) society recommendations. World J. Surg. 43, 299–330. https://doi.org/10.1007/s00268-018-4786-4 (2019).
Bai, X. H., Dang, J., Chen, Z. Q., He, Z. & Li, G. Comparison between Intensity-Modulated radiotherapy and Three-Dimensional conformal radiotherapy for their effectiveness in esophageal cancer treatment: A retrospective single institution study. J. Oncol. 2020, 6582341. https://doi.org/10.1155/2020/6582341 (2020).
Xu, D., Li, G., Li, H. & Jia, F. Comparison of IMRT versus 3D-CRT in the treatment of esophagus cancer: A systematic review and meta-analysis. Med. (Baltim). 96, e7685. https://doi.org/10.1097/md.0000000000007685 (2017).
Yap, W. K. et al. Adjuvant chemoradiotherapy associated with improved overall survival in resected esophageal squamous cell carcinoma after neoadjuvant chemoradiotherapy in Intensity-Modulated radiotherapy era. Biomedicines https://doi.org/10.3390/biomedicines10112989 (2022).
Park, S. Y. et al. The role of adjuvant chemotherapy after neoadjuvant chemoradiotherapy followed by surgery in patients with esophageal squamous cell carcinoma. Cancer Res. Treat. 55, 1231–1239. https://doi.org/10.4143/crt.2022.1417 (2023).
Kelly, R. J. et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl. J. Med. 384, 1191–1203. https://doi.org/10.1056/NEJMoa2032125 (2021).
Unger, J. M. et al. Sex differences in risk of severe adverse events in patients receiving Immunotherapy, targeted therapy, or chemotherapy in cancer clinical trials. J. Clin. Oncol. 40, 1474–1486. https://doi.org/10.1200/jco.21.02377 (2022).
Kim, H. I., Lim, H. & Moon, A. Sex differences in cancer: Epidemiology, genetics and therapy. Biomol. Ther. (Seoul). 26, 335–342. https://doi.org/10.4062/biomolther.2018.103 (2018).
Pala, L. et al. Sex differences in efficacy and toxicity of systemic cancer treatments: Role of the microbiome. J. Clin. Oncol. 37, 439. https://doi.org/10.1200/jco.18.01270 (2019).
Glatz, T. et al. Perioperative platin-based chemotherapy for locally advanced esophagogastric adenocarcinoma: Postoperative chemotherapy has a substantial impact on outcome. Eur. J. Surg. Oncol. 41, 1300–1307. https://doi.org/10.1016/j.ejso.2015.07.010 (2015).
Ajani, J. A. et al. Esophageal and esophagogastric junction cancers, version 2.2019, NCCN clinical practice guidelines in oncology. J. Natl. Compr. Canc Netw. 17, 855–883. https://doi.org/10.6004/jnccn.2019.0033 (2019).
Wang, X. et al. Incidence and onset of severe cardiac events after radiotherapy for esophageal cancer. J. Thorac. Oncol. 15, 1682–1690. https://doi.org/10.1016/j.jtho.2020.06.014 (2020).
Verma, V., Moreno, A. C. & Lin, S. H. Advances in radiotherapy management of esophageal cancer. J. Clin. Med. https://doi.org/10.3390/jcm5100091 (2016).
Funding
This research was supported by the Natural Science Foundation of Fujian Province, China (Grant No. 2024J011169; Program Type: General Program).
Author information
Authors and Affiliations
Contributions
Jia-Lei Huang and Zi-Xin Lin contributed equally to this work, co-drafting the initial manuscript and participating in data curation and analysis. Ke-Yi Zeng was responsible for the collection and analysis of key experimental data. Lu Zheng, Xuan-Ping Lin, and He-Rui Wang participated in data collection, curation, and the initial preparation of tables and figures. Yong-Wei Xie and Jing-Rong Yang (corresponding authors) conceptualized and designed the study, oversaw the entire research process, and critically reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, JL., Lin, ZX., Zeng, KY. et al. Impact of adjuvant therapy on survival in esophageal cancer patients after neoadjuvant therapy investigated by a population based cohort study. Sci Rep (2026). https://doi.org/10.1038/s41598-026-39930-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-39930-5