Abstract
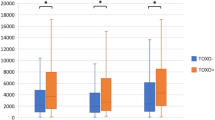
Studies on murine models show that Toxoplasma gondii infection reduces cerebral microvascular perfusion and induces neuroinflammation by activating cerebral endothelial cells, which could affect traumatic brain injury (TBI) outcomes. Whether TBI inflammatory profile and outcomes differ in persons with concurrent latent infection with Toxoplasma gondii has not yet been elucidated in TBI patients, constituting the core of this study. Understanding the impact of chronic T. gondii infection on neuroinflammation after TBI may ensure better management and prognosis of TBI. This study aimed to investigate the inflammatory profile in TBI patients and investigate the influence of concurrent T. gondii infection on inflammatory markers and TBI outcomes. The level of inflammatory markers [interleukin (IL)-1β, IL-6, IL-10, interferon-gamma (INF-ɣ), Tumour necrosis factor-alpha (TNF-α)], and Toxoplasma gondii infection were detected in serum obtained < 24 h post-injury. Glasgow Outcome Scale-Extended (GOSE) was used to evaluate the 6-month outcome post-discharge. The Wilcoxon and Kruskal-Wallis’s rank sum tests were used to compare concentrations of inflammatory markers to T. gondii infection and TBI outcome. T. gondii infection was detected in 33% (52/160) of TBI cases. There was a significant increase (p < 0.001) in the concentrations of all inflammatory markers (IM) in TBI patients compared with the healthy controls. Higher levels of IL-6, INF-ɣ, and TNF-α were associated with mortality. Findings from this study show an increase in IM levels for all the T. gondii-positive TBI cases, which were significant for IL-1β (P < 0.001) and TNF-α (P < 0.001). IL-1β and TNF-α, had significantly greater density values, above 30pg/mL and 90pg/mL, respectively, in TBI patients infected with T. gondii, compared to greater density values, below 30 pg/mL and 90 pg/mL, respectively, for TBI patients seronegative to T. gondii. Concurrent T. gondii infection in TBI significantly influenced the inflammation profile of patients. Further multicenter studies with larger sample sizes will provide more insights into T. gondii induced neuropathology and prognostication in TBI care.
Similar content being viewed by others
Data availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- TBI:
-
Traumatic Brain Injury
- SSA:
-
Sub-Saharan Africa
- LMICs:
-
Low-middle-income countries
- IL:
-
Interleukin
- INF-ɣ:
-
Interferon-gamma
- TNF-α:
-
Tumour necrosis factor-alpha
- GOSE:
-
Glasgow-coma outcome scale extended
- CT:
-
Computed tomography
- LHD:
-
Laquintinie hospital douala
- GBD:
-
Global burden of diseases
References
Wongchareon, K. et al. IMPACT and CRASH prognostic models for traumatic brain injury: external validation in a South-American cohort. Inj Prev. 26(6), 546–554. https://doi.org/10.1136/injuryprev-2019-043466 (2020).
Dewan, M. C. et al. Estimating the global incidence of traumatic brain injury. J. Neurosurg. 130, 1080–1097. https://doi.org/10.3171/2017.10.JNS17352 (2019).
GBD 2016 Traumatic Brain Injury and Spinal Cord Injury Collaborators. Global, regional, and National burden of traumatic brain injury and spinal cord injury, 1990–2016: A systematic analysis for the global burden of disease study 2016. Lancet Neurol. 18(1), P56–P87. https://doi.org/10.1016/S1474-4422(18)30415-0 (2018).
Majdam, M. et al. Epidemiology of traumatic brain injuries in europe: A cross-sectional analysis. Lancet Public. Health. 1(2), 76–83. https://doi.org/10.1016/S2468-2667(16)30017-2 (2016).
Buh, F. C. et al. Demographics, causes, and outcome of traumatic brain injury among trauma cases in Cameroon: A multi-center five year’s retrospective study. Neurotrauma Rep. 3(1), 569–583 (2022).
Gardner, A. G. & Zafonte, R. Neuroepidemiology of traumatic brain injury. Handb. Clin. Neurol. 138(3), 207–223. https://doi.org/10.1016/B978-0-12-802973-2.00012-4 (2016).
Embolo, F. N. et al. Epidemiology of traumatic brain injury based on clinical symptoms among inhabitants of the South West region of cameroon: a community-based study. Brain Inj. 35(10), 1184–1191 (2021).
Adegboyega, G. et al. The burden of traumatic brain injury in Sub-Saharan africa: A scoping review. World Neurosurg. 156, 192–205. https://doi.org/10.1016/j.wneu.2021.09.021 (2021).
Dadas, A., Washington, J., Diaz-Arrastia, R. & Janigro, D. Biomarkers in traumatic brain injury (TBI): A review. Neuropsychiatr Dis. Treat. 8, 14:2989–3000 (2018).
Kumar, A. & Loane, D. J. Neuroinflammation after traumatic brain injury: Opportunities for therapeutic intervention. Brain Behav. Immun. 26(8), 1191–1201 (2012).
Hong, H., Kim, B. S. & Im, H. I. Pathophysiological role of neuroinflammation in neurodegenerative diseases and psychiatric disorders. Int. Neurourol. J. 20(Suppl 1), S2–S7 (2016).
Baker, T. L. et al. Catastrophic consequences: can the feline parasite Toxoplasma gondii prompt the purrfect neuroinflammatory storm following traumatic brain injury? J. Neuroinflammation. 25(17(1)), 222 (2020).
Corrigan, F., Mander, K. A., Leonard, A. V. & Vink, R. Neurogenic inflammation after traumatic brain injury and its potentiation of classical inflammation. J Neuroinflammation. 11;13(1):264 (2016).
Xiong, Y., Mahmood, A. & Chopp, M. Current Understanding of neuroinflammation after traumatic brain injury and cell-based therapeutic opportunities. Chin. J. Traumatol. 21(3), 137–151 (2018).
Mareze, M. et al. Socioeconomic vulnerability associated to Toxoplasma gondii exposure in Southern Brazil. PLoS ONE. 14(2), e0212375 (2019).
Baker, T. L. et al. Pre-existing Toxoplasma gondii infection increases susceptibility to pentylenetetrazol-induced seizures independent of traumatic brain injury in mice. Front. Mol. Neurosci. 15, 1079097 (2023).
Estato, V. et al. The neurotropic parasite Toxoplasma gondii induces sustained neuroinflammation with microvascular dysfunction in infected mice. Am. J. Pathol. 2188(11), 2674–2687 (2018).
Baker, T. L. et al. A pre-existing Toxoplasma gondii infection exacerbates the pathophysiological response and extent of brain damage after traumatic brain injury in mice. J. Neuroinflammation. 21, 14 (2024).
Luo, P., Fei, F., Zhang, L., Qu, Y. & Fei, Z. The role of glutamate receptors in traumatic brain injury: Implications for postsynaptic density in pathophysiology. Brain Res. Bull. 85(6), 313–320 (2011).
Guerriero, R. M., Giza, C. C. & Rotenberg, A. Glutamate and GABA imbalance following traumatic brain injury. Curr. Neurol. Neurosci. Rep. 15(5), 27 (2015).
Schimmel, S. J., Acosta, S. & Lozano, D. Neuroinflammation in traumatic brain injury: A chronic response to an acute injury. Brain Circ. 3(3), 135–142 (2017).
Gruenbaum, B. F., Zlotnik, A., Fleidervish, I., Frenkel, A. & Boyko, M. G. Neurotoxicity and destruction of the Blood-Brain barrier: Key pathways for the development of neuropsychiatric consequences of TBI and their potential treatment strategies. Int. J. Mol. Sci. 25(17), 9628 (2022).
Carpio, A. et al. Parasitic diseases of the central nervous system: Lessons for clinicians and policy makers. Expert Rev. Neurother. 16(4), 401–414 (2016).
Sribnick., E. A., Popovich, P. G. & Hall, M. W. Central nervous system injury-induced immune suppression. Neurosurg. Focus. 52(2), E10 (2022).
Bouras, M., Asehnoune, K. & Roquilly, A. Immune modulation after traumatic brain injury. Front. Med. (Lausanne). 9, 995044 (2022).
Griffin, G. D. The injured brain: TBI, mTBI, the immune system, and infection: Connecting the Dots. Mil. Med. 176(4), 364–368 (2011).
Sharma, R. et al. Infections after a traumatic brain injury: The complex interplay between the immune and neurological systems. Brain. Behav. Immun. 79, 63–74 (2019).
Daher, D. et al. Comprehensive overview of Toxoplasma gondii-Induced and associated diseases. Pathogens 20(11), 1351 (2021).
Jafari, M. M. et al. Immune system roles in pathogenesis, prognosis, control, and treatment of Toxoplasma gondii infection. Int. Immunopharmacol. 124(Pt A), 110872 (2023).
Moghaddami, R., Mahdipour, M. & Ahmadpour, E. Inflammatory pathways of toxoplasmagondii infection in pregnancy. Travel Med. Infect. Dis. 62, 102760 (2024).
Konradt, C. et al. Endothelial cells are a replicative niche for entry of Toxoplasma gondii to the central nervous system. Nat. Microbiol. 15(1), 16001 (2016).
Cabral, C. M. et al. Neurons are the primary target cell for the brain-tropic intracellular parasite Toxoplasma gondii. PLoS Pathog. 12(2), e1005447 (2016).
Zheng, R. et al. Neuroinflammation following traumatic brain injury: take it seriously or not. Front. Immunol. 13, 855701 (2022).
Eaton, J. et al. Epidemiology, management, and functional outcomes of traumatic brain injury in Sub-Saharan Africa. World Neurosurg. 108, 650–655. https://doi.org/10.1016/j.wneu.2017.09.084 (2018).
Benneh, G., Douala Cameroon & Britanica Consulted 30.03.2022. Available online: (2022). https://www.britannica.com/place/Cameroon (2022).
World Population Review. Douala Population 2023. Available online (2023). https://worldpopulationreview.com/world-cities/doualapopulation
Buh, F. C. et al. Traumatic brain injury in Cameroon: A prospective observational study in a level I trauma centre. Medicina (Kaunas). 59(9), 1558. (2023). https://doi.org/10.3390/medicina59091558
Czeiter, E. et al. Blood biomarkers on admission in acute traumatic brain injury: Relations to severity, CT findings and care path in the CENTER-TBI study. EBioMedicine 56, 102785 (2020).
Ebrahimzadeh, A., Shahraki, M. K. & Mohammadi, A. Serological and molecular diagnosis of Toxoplasma gondii in patients with schizophrenia. J. Parasit. Dis. 42(2), 177–181 (2018).
Baird, A. C. et al. Dysregulation of innate immunity in ulcerative colitis patients who fail anti-tumor necrosis factor therapy. World J. Gastroenterol. 22(41), 9104–9116 (2016).
Wu, D., Dinh, T. L., Bausk, B. P. & Walt, D. R. Long-term measurements of human inflammatory cytokines reveal complex baseline variations between individuals. Am. J. Pathol. 187(12), 2620–2626 (2017).
Wilking, H., Thamm, M., Stark, K., Aebischer, T. & Seeber, F. Prevalence, incidence estimations, and risk factors of Toxoplasma gondii infection in germany: A representative, cross-sectional, serological study. Sci. Rep. 6, 22551 (2016).
van den Berg, O. E. et al. Seroprevalence of Toxoplasma gondii and associated risk factors for infection in the netherlands: Third cross-sectional National study. Epidemiol. Infect. 151, e136 (2023).
Morais, R. D. A. P. B., Carmo, E. L. D., Costa, W. S., Marinho, R. R. & Póvoa, M. M. T. gondii infection in urban and rural areas in the amazon: Where is the risk for toxoplasmosis? Int. J. Environ. Res. Public. Health. 17(16), 8664 (2021).
Buh, F. C. et al. Serum biomarker concentrations upon admission in acute traumatic brain injury: Associations with TBI Severity, Toxoplasma gondii Infection, and outcome in a referral hospital setting in Cameroon. NeuroSci 4(3), 164–177 (2023).
Rodney, T., Osier, N. & Gill, J. Pro- and anti-inflammatory biomarkers and traumatic brain injury outcomes: A review. Cytokine 110, 248–256 (2018).
LaPar, D. J. et al. Severe traumatic head injury affects systemic cytokine expression. J. Am. Coll. Surg. 214(4), 478–486 (2012). discussion 486-8.
Tsitsipanis, C. et al. Inflammation biomarkers IL-6 and IL-10 May improve the diagnostic and prognostic accuracy of currently authorized traumatic brain injury tools. Exp. Ther. Med. 26, 364 (2023).
Aisiku, I. P. et al. Plasma cytokines IL-6, IL-8, and IL-10 are associated with the development of acute respiratory distress syndrome in patients with severe traumatic brain injury. Crit. Care. 15(20), 288 (2016).
Covington, N. V. & Duff, M. C. Heterogeneity is a hallmark of traumatic brain Injury, not a limitation: A new perspective on study design in rehabilitation research. Am. J. Speech Lang. Pathol. 30(2S), 974–985 (2021).
Edwards, K. A. et al. Inflammatory cytokines associate with neuroimaging after acute mild traumatic brain injury. Front. Neurol. 11, 348 (2020).
Longhi, L. et al. Tumor necrosis factor in traumatic brain injury: Effects of genetic deletion of p55 or p75 receptor. J. Cereb. Blood Flow. Metab. 33(8), 1182–1189 (2013).
Ozen, I. et al. Interleukin-1 beta neutralization attenuates traumatic brain injury-induced microglia activation and neuronal changes in the globus pallidus. Int J Mol Sci. 21(2), 387 (2020).
Woodcock, T. & Morganti-Kossmann, M. C. The role of markers of inflammation in traumatic brain injury. Front. Neurol. 4, 18 (2013).
French, T. et al. Neuronal impairment following chronic Toxoplasma gondii infection is aggravated by intestinal nematode challenge in an IFN-γ-dependent manner. J. Neuroinflammation. 16(1), 159. https://doi.org/10.1186/s12974-019-1539-8 (2019).
Lindblad, C., Rostami, E. & Helmy, A. Interleukin-1 receptor antagonist as therapy for traumatic brain injury. Neurotherapeutics 20(6), 1508–1528 (2023).
Olmos, G. & Lladó, J. Tumor necrosis factor alpha: A link between neuroinflammation and excitotoxicity. Mediators Inflamm. 2014, 861231 (2014).
French, T. et al. Neuronal impairment following chronic Toxoplasma gondii infection is aggravated by intestinal nematode challenge in an IFN-γ-dependent manner. J. Neuroinflammation. 16(1), 159. https://doi.org/10.1186/s12974-019-1539-8 (2019).
Postolache, T. T. et al. Inflammation in traumatic brain injury. J. Alzheimers Dis. 74(1), 1–28 (2020).
Thelin, E. P. et al. Elucidating pro-inflammatory cytokine responses after traumatic brain injury in a human stem cell model. J. Neurotrauma. 35(2), 341–352 (2018).
Ziebell, J. M., & Morganti-Kossmann, M. C. Involvement of pro-and anti-inflammatory cytokines and chemokines in the pathophysiology of traumatic brain injury. Neurotherapeutics 7(1), 22–30 (2010).
Loane, D. J., Stoica, B. A. & Faden A.I. Neuroprotection for traumatic brain injury. Handb. Clin. Neurol. 127, 343–366 (2015).
Shohami, E., Ginis, I. & Hallenbeck, J. M. Dual role of tumor necrosis factor alpha in brain injury. Cytokine Growth Factor Rev. 10(2), 119–130 (1999).
Ciryam, P., Gerzanich, V. & Simard, J. M. Interleukin-6 in traumatic brain injury: A Janus-faced player in damage and repair. J. Neurotrauma. 40(21–22), 2249–2269 (2023).
El Saftawy, E. A. et al. Can Toxoplasma gondii pave the road for dementia? J. Parasitol. Res. 30, 8859857 (2020).
Sun, M. The Effect of Toxoplasma gondii Infection on Common Acquired Brain Insults. Project, Monash University 2022. Available online (2022). https://brainfoundation.org.au/research-grants/2019/neural-infections/
Bokaba, R. P. et al. Toxoplasma gondii in African wildlife: A systematic review. Pathogens 11(8), 868 (2022).
Acknowledgements
We thank the administration and health personnel of the Laquintinie Hospital Douala for facilitating the data collection phase and to LabEx Physiology, Pharmacological Targets and Therapeutics of the University of Buea, Cameroon for their assistance laboratory analysis. The research was funded by the NIHR ref: NIHR132455 using UK aid from the UK Government to support global health research. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR or the UK Government.
Funding
The research was funded by the NIHR ref: NIHR132455 using UK aid from the UK Government to support global health research.
Author information
Authors and Affiliations
Contributions
F.C.B.: study design, data collection, Laboratory analysis, writing, G.S.T; study design, editing, A.I.R.M.: study design, editing, M.M.: study design, F.H.K.: editing, V.T.J.: Laboratory analysis, E.Y.: data analysis, B.K.M.: editing; K.W.W.: editing, P.J.A.H.: study design, editing, I.U.N.S.: study design, editing, and validation.All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Ethical clearance for the study was obtained from the Institutional Review Board of the Faculty of Health Sciences (IRB-FHS), University of Buea (Reference N◦ 1238-08), and administrative authorization was obtained from LHD. Informed consent was given by the participants or their family members, and all data collected was kept strictly confidential with physical and electronic barriers.
Consent for publication
Not Applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Buh, F.C., Taiwe, G.S., Maas, A.I.R. et al. Concurrent Toxoplasma gondii infection and neuroinflammation in traumatic brain injury patients in a referral hospital in Douala Cameroon. Sci Rep (2026). https://doi.org/10.1038/s41598-026-40284-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-40284-1