Abstract
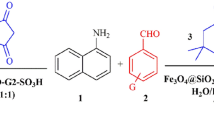
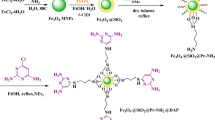
Fe₃O₄@SiO₂-NH-TCT–(trans-4-hydroxy-L-proline)₂ nanoparticles were synthesized as a chiral, magnetically recoverable nanocatalyst. The material was thoroughly characterized by FT-IR, VSM, XRD, TEM, FE-SEM, BET, CHNS elemental analysis, mass spectrometry, and ¹H/¹³C-NMR. The catalyst enabled a one-pot, multicomponent diastereoselective synthesis of indoline spirobicyclic derivatives from anilines, carbon acids, Fischer’s base, and aldehydes under solvent-free conditions at ambient temperature. The method afforded high to excellent yields with excellent diastereoselectivity. Product structures and anti/syn diastereomeric ratios were established by ¹H-, ¹³C-NMR, 2D-ROESY, mass spectrometry, and single-crystal X-ray diffraction.
Similar content being viewed by others
Data availability
All data are available in the supporting information file.
References
Thorat, B. R. et al. L-Proline: A versatile organo-catalyst in organic chemistry. Comb. Chem. High. Throughput Screen. 26 (6), 1108–1140. https://doi.org/10.2174/1386207325666220720105845 (2022).
Groot, I. M. N. Investigation of active catalysts at work. Acc. Chem. Res. 54(23), 4334–4341. https://doi.org/10.1021/acs.accounts.1c00429 (2021).
Rigby, S. P. The anatomy of amorphous, heterogeneous catalyst pellets. Materials 16(8), 3205. https://doi.org/10.3390/ma16083205 (2023).
Guo, Y., Wang, M., Zhu, Q., Xiao, D. & Ma, D. Ensemble effect for single-atom, small Cluster and nanoparticle catalysts. Nat. Catal. 5(9), 766–776. https://doi.org/10.1038/s41929-022-00839-7 (2022).
Vinita Nanomaterials as intrinsic enzyme mimetic catalysts. In: versatile solicitations of materials science in diverse science fields, 99–114. Nova Science (2021).
Saim, A. K. Review of catalytic activities of biosynthesized metallic nanoparticles in wastewater treatment. Environ. Technol. Rev. 10(1), 111–130. https://doi.org/10.1080/21622515.2021.1893831 (2021).
Yazdian-Robati, R. Application of the catalytic activity of gold nanoparticles for development of optical aptasensors. Anal. Biochem. 629, 114307. https://doi.org/10.1016/j.ab.2021.114307 (2021).
Praharaj, S. & Rout, D. Magnetic nanoparticles in catalysis industry. In: fundamentals and industrial applications of magnetic nanoparticles, pp. 477–495. Elsevier (2022). https://doi.org/10.1016/B978-0-12-822819-7.00022-3
Ghaznavi, H. et al. Effects of folate-Conjugated Fe2O3@Au core–shell nanoparticles on oxidative stress markers, DNA damage, and histopathological characteristics: Evidence from in vitro and in vivo studies. Med. Oncol. 39(9), 122. https://doi.org/10.1007/s12032-022-01713-z (2022).
Zare Fekri, L., Varma, R. S., Hassanzadia, L., Sedighi Pashakia, F. & Nikpassand, M. Green synthesis of novel 5-amino-bispyrazole-4-carbonitriles using a recyclable Fe 3 O 4 @SiO 2 @vanillin@thioglycolic acid nano-catalyst. RSC Adv. 12(2), 834. https://doi.org/10.1039/D1RA08001F (2022).
Torabi, M., Zare Fekri, L. & Nikpassand, M. Synthesis, characterization and application of Fe3O4@Silicapropyl@vaniline-covalented isoniazid-copper(I) nanocomposite as a new, mild, effective and magnetically recoverable lewis acid catalyst for the synthesis of acridines and novel azoacridines. J. Mol. Strut. 1250, 31761. https://doi.org/10.1016/ j. molstruc.2021.131761. (2022).
Wang, S., Zhou, Y. & Li, N. Superparamagnetic iron oxide nanoparticles for theranostics. In Inorganic Nanosystems, pp. 201–228. Elsevier (2023). https://doi.org/10.1016/B978-0-323-85784-0.00001-7
Inoue, M., Tsurugi, H. & Mashima, K. Early transition metal complexes for direct C H bond functionalization of heteroatom-containing organic compounds. Coord. Chem. Rev. 473, 214810. https://doi.org/10.1016/j.ccr.2022.214810 (2022).
Yamashita, Y. & Gröger, H. Organocatalyst/biocatalyst dual catalysis. In Science of Synthesis, pp. 339–364 (2019). Georg Thieme Verlag: Stuttgart,. (2019). https://doi.org/10.1055/sos-SD-232-00103
Yao, R., Li, Y., Wang, J., Chen, J. & Xu, Y. Crown ether as organocatalyst for reductive upgrading of CO2 to N-containing benzoheterocyclics and N-formamides. J. Catal. 418, 78–89. https://doi.org/10.1016/j.jcat.2023.01.005 (2023).
Xu, J. et al. Guanidinium as bifunctional organocatalyst for ring-opening polymerizations. Polymer 154, 17–26. https://doi.org/10.1016/j.polymer.2018.08.074 (2018).
Kalantari, F., Ramazani, A., Poor Heravi, M. R., Aghahosseini, H. & Ślepokura, K. Magnetic nanoparticles functionalized with copper hydroxyproline complexes as an efficient, recoverable, and recyclable nanocatalyst: Synthesis and its catalytic application in a tandem Knoevenagel–Michael cyclocondensation reaction. Inorg. Chem. 60(19), 15010–15023. https://doi.org/10.1021/acs.inorgchem.1c02470 (2021).
Ozhogin, I. V. et al. Novel indoline spiropyrans based on human hormones β-estradiol and estrone: Synthesis, structure, chromogenic and cytotoxic roperties. Molecules 28(9), 3866. https://doi.org/10.3390/molecules28093866 (2023).
Sinibaldi, M. E. & Canet, I. J. Synthetic approaches to spiroaminals. Eur. J. Org. Chem. https://doi.org/10.1002/ejoc.200800371 (2008).
Dommaraju, Y., Borthakur, S. & Prajapati, D. Rapid access to indoline spiropyran scaffolds by domino-Knoevenagel/oxa‐Diels‐Alder (DKODA) reaction at room temperature. ChemistrySelect 1, 6768–6771. https://doi.org/10.1002/slct (2016).
Gayen, P., Sar, S. & Ghorai, P. Stereodivergent synthesis of spiroaminals via chiral bifunctional hydrogen bonding organocatalysis. Angew. Chem. Int. Ed. 63, e20244106. https://doi.org/10.1002/anie.202404106 (2024).
Najafabadi, S. M. & Safaei Ghomi, J. Synthesis of COF-SO3H immobilized on manganese ferrite nanoparticles as an efficient nanocomposite in the preparation of spirooxindoles. Sci. Rep. 13, 22731 (2023).
Vaishali, Sharma, S. et al. Magnetic nanoparticle-ccatalyzed synthesis of indole derivatives: a green and sustainable method. Monatsh fur Chem. 156, 247–277. https://doi.org/10.1007/s00706-025-03295-8 (2025).
Esmaeilpour, M., Zahmatkesh, S., Fahimi, N. & Nosratabadi, M. Palladium nanoparticles immobilized on EDTA-modified Fe3O4@SiO2 nanospheres as an efficient and magnetically separable catalyst for Suzuki and Sonogashira cross-coupling reactions. Appl. Organomet. Chem. 32(4), e4302. https://doi.org/10.1002/aoc.4302 (2018).
Rasmussen, R. S. & Brattain, R. R. Infrared spectra of some carboxylic acid derivatives. J. Am. Chem. Soc. 71, 1073–1079. https://doi.org/10.1021/ja01171a088 (1949).
Sevukarajan, M., Thanuja, B., Sodanapalli, R. & Nair, R. Synthesis and characterization of a pharmaceutical co-crystal: (Aceclofenac: Nicotinamide). J. Pharm. Sci. Res. 3, 1288–1293 (2011).
Zhao, H., Sun, H. & Li, X. Synthesis and catalytic property of iron pincer complexes generated by Csp3–H activation. Organomet 33, 3535–3539. https://doi.org/10.1021/om500429r (2014).
Takei, K. I., Takahashi, R. & Noguchi, T. Correlation between the hydrogen-bond structures and the C = O stretching frequencies of carboxylic acids as studied by density functional theory calculations: Theoretical basis for interpretation of infrared bands of carboxylic groups in proteins. J. Phys. Chem. B 112(21), 6725–6731. https://doi.org/10.1021/jp801151k (2008).
Acknowledgements
The authors gratefully acknowledge the University of Shiraz for its support through the provision of facilities.
Author information
Authors and Affiliations
Contributions
Danial Rafipour : Data curation, formal analysis, investigation, resources, validation, visualization, writing original draft; Marzieh Jamali: Formal analysis, investigation, methodology, writing original draft, project administration; Ali Reza Sardarian : conceptualization, supervision, writing-review&editing, funding acquisition; Saeed Davoodi: Formal analysis, validation, investigation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rafipour, D., Sardarian, A.R., Jamali, M. et al. One-pot multicomponent diastereoselective synthesis of indoline spirobicyclics using a recyclable chiral nanomagnetic L-proline catalyst under mild conditions. Sci Rep (2026). https://doi.org/10.1038/s41598-026-40598-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-40598-0