Abstract
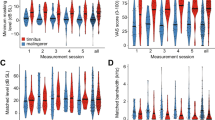
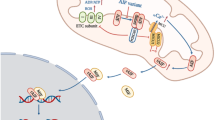
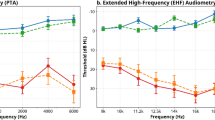
It is widely accepted that tinnitus arises from maladaptive neuroplasticity in the central auditory system, which compensates for reduced peripheral input; notably, the loss of inner hair cell (IHC) synapses is considered a key underlying factor. IHC synapses can immediately and permanently degenerate after noise exposure, independent of hair cell loss, whereas the cell bodies and central projections of cochlear neurons can survive for months to years, providing an extended therapeutic window for improving afferent input through synapse regeneration. Using NHPN-1010 (a combination of 2,4-disulfophenyl-N-tert-butylnitrone and N-acetylcysteine), we propose a delayed treatment strategy (≥ 4 weeks after trauma) for noise-induced tinnitus (NIT) in rats. NHPN-1010 enhanced gap-prepulse inhibition of acoustic startle responses—indicative of diminished tinnitus-like deficits—coupled with a concomitant upregulation of GABAergic inhibition in the dorsal cochlear nucleus, relative to vehicle alone (saline). Furthermore, NHPN-1010, compared to saline, led to increased wave-I amplitudes of auditory brainstem responses, the effect accompanied by a corresponding rise in the number of presynaptic ribbons in IHCs. Our findings suggest that NHPN-1010 plays a therapeutic role in NIT by promoting cochlear synaptogenesis and normalizing central auditory signaling.
Similar content being viewed by others
Data availability
All data are available from the corresponding author.
References
Schmuzigert, N., Fostiropoulos, K. & Probst, R. Long-term assessment of auditory changes resulting from a single noise exposure associated with non-occupational activities. Int. J. Audiol. 45, 46–54 (2006).
Kreuzer, P. M. et al. Trauma-associated tinnitus: Audiological, demographic and clinical characteristics. PLoS ONE 7, e45599 (2012).
Bhatt, J. M., Lin, H. W. & Bhattacharyya, N. Prevalence, severity, exposures, and treatment patterns of tinnitus in the United States. JAMA Otolaryngol. Head Neck Surg. 142, 959–965 (2016).
Jastreboff, P. J. Phantom auditory perception (tinnitus): Mechanisms of generation and perception. Neurosci. Res. 8, 221–254 (1990).
Penner, M. J. & Bilger, R. C. Psychophysical observations and the origin of tinnitus. In Mechanisms of Tinnitus (eds Vernon, J. A. & Møller, A. R.) 219–230 (Allyn and Bacon, 1995).
Lockwood, A. H. et al. The functional neuroanatomy of tinnitus: evidence for limbic system links and neural plasticity. Neurology 50, 114–120 (1998).
Auerbach, B. D., Rodrigues, P. V. & Salvi, R. J. Central gain control in tinnitus and hyperacusis. Front. Neurol. 5, 206 (2014).
Henry, J. A., Roberts, L. E., Caspary, D. M., Theodoroff, S. M. & Salvi, R. J. Underlying mechanisms of tinnitus: Review and clinical implications. J. Am. Acad. Audiol. 25, 5–22 (2014).
Schaette, R. & Kempter, R. Predicting tinnitus pitch from patients’ audiograms with a computational model for the development of neuronal hyperactivity. J. Neurophysiol. 101, 3042–3052 (2009).
Noreña, A. J. An integrative model of tinnitus based on a central gain controlling neural sensitivity. Neurosci. Biobehav. Rev. 35, 1089–1099 (2011).
Schaette, R. & McAlpine, D. Tinnitus with a normal audiogram: Physiological evidence for hidden hearing loss and computational model. J. Neurosci. 31, 13452–13457 (2011).
Roberts, L. E. et al. Ringing ears: The neuroscience of tinnitus. J. Neurosci. 30, 14972–14979 (2010).
Geven, L. I., de Kleine, E., Free, R. H. & van Dijk, P. Contralateral suppression of otoacoustic emissions in tinnitus patients. Otol. Neurotol. 32, 315–321 (2011).
Xiong, H. et al. Hidden hearing loss in tinnitus patients with normal audiograms: Implications for the origin of tinnitus. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 27, 362–365 (2013) (in Chinese).
Paul, B. T., Bruce, I. C. & Roberts, L. E. Evidence that hidden hearing loss underlies amplitude modulation encoding deficits in individuals with and without tinnitus. Hear. Res. 344, 170–182 (2017).
Kujawa, S. G. & Liberman, M. C. Adding insult to injury: Cochlear nerve degeneration after “temporary” noise-induced hearing loss. J. Neurosci. 29, 14077–14085 (2009).
Furman, A. C., Kujawa, S. G. & Liberman, M. C. Noise-induced cochlear neuropathy is selective for fibers with low spontaneous rates. J. Neurophysiol. 110, 577–586 (2013).
Liberman, L. D., Suzuki, J. & Liberman, M. C. Dynamics of cochlear synaptopathy after acoustic overexposure. J. Assoc. Res. Otolaryngol. 16, 205–219 (2015).
Bauer, C. A., Brozoski, T. J. & Myers, K. Primary afferent dendrite degeneration as a cause of tinnitus. J. Neurosci. Res. 85, 1489–1498 (2007).
Knipper, M., Zimmermann, U. & Müller, M. Molecular aspects of tinnitus. Hear. Res. 266, 60–69 (2010).
Rüttiger, L. et al. The reduced cochlear output and the failure to adapt the central auditory response causes tinnitus in noise-exposed rats. PLoS ONE 8, e57247 (2013).
Johnsson, L. G. Sequence of degeneration of Corti’s organ and its first-order neurons. Ann. Otol. Rhinol. Laryngol. 83, 294–303 (1974).
Liberman, M. C. & Kiang, N. Y. Acoustic trauma in cats. Cochlear pathology and auditory-nerve activity. Acta Otolaryngol. Suppl. 358, 1–63 (1978).
Felix, H., Pollak, A., Gleeson, M. & Johnsson, L. G. Degeneration pattern of human first-order cochlear neurons. Adv. Otorhinolaryngol. 59, 116–123 (2002).
Chen, G., Shi, J., Hu, Z. & Hang, C. Inhibitory effect on cerebral inflammatory response following traumatic brain injury in rats: A potential neuroprotective mechanism of N-acetylcysteine. Mediators Inflamm. 2008, 716458 (2008).
Bavarsad Shahripour, R., Harrigan, M. R. & Alexandrov, A. V. N-acetylcysteine (NAC) in neurological disorders: Mechanisms of action and therapeutic opportunities. Brain Behav. 4, 108–122 (2014).
Kopke, R. et al. Efficacy and safety of N-acetylcysteine in prevention of noise-induced hearing loss: A randomized clinical trial. Hear. Res. 323, 40–50 (2015).
Williams, H. E., Claybourn, M. & Green, A. R. Investigating the free radical trapping ability of NXY-059, S-PBN and PBN. Free Radical Res. 41, 1047–1052 (2007).
Floyd, R. A., Chandru, H. K., He, T. & Towner, R. Anti-cancer activity of nitrones and observations on mechanism of action. Anti-Cancer Agents Med. Chem. 11, 373–379 (2011).
Tsuji, M., Inanami, O. & Kuwabara, M. Induction of neurite outgrowth in PC12 cells by alpha-phenyl-N-tert-butylnitron through activation of protein kinase C and the Ras-extracellular signal-regulated kinase pathway. J. Biol. Chem. 276, 32779–32785 (2001).
Yasui, H. et al. Induction of neurite outgrowth by alpha-phenyl-N-tert-butylnitrone through nitric oxide release and Ras-ERK pathway in PC12 cells. Free Radic. Res. 44, 645–654 (2010).
Chan, K. M., Gordon, T., Zochodne, D. W. & Power, H. A. Improving peripheral nerve regeneration: From molecular mechanisms to potential therapeutic targets. Exp. Neurol. 261, 826–835 (2014).
Ewert, D. L. et al. HPN-07, a free radical spin trapping agent, protects against functional, cellular and electrophysiological changes in the cochlea induced by acute acoustic trauma. PLoS ONE 12, e0183089. https://doi.org/10.1371/journal.pone.0183089 (2017).
Lu, J. et al. Electrophysiological assessment and pharmacological treatment of blast-induced tinnitus. PLoS ONE 16, e0243903 (2021).
Du, X. et al. Development of a temporary threshold shift model of chronic noise-induced tinnitus with evidence of altered auditory signaling pathway inhibition. Int. Tinnitus J. 28, 261–276 (2024).
Turner, J. G. & Larsen, D. Effects of noise exposure on development of tinnitus and hyperacusis: Prevalence rates 12 months after exposure in middle-aged rats. Hear. Res. 334, 30–36 (2016).
Heffner, H. E., Heffner, R. S., Contos, C. & Ott, T. Audiogram of the hooded rat (Rattus norvegicus). Hear. Res. 73, 244–247 (1994).
Du, X. et al. Identification and quantification of GABAAR-α1-positive cells in the DCN of rats with behavioral evidence of noise-induced tinnitus. IBRO Neurosci. Rep. 19, 332–344. https://doi.org/10.1016/j.ibneur.2025.07.005 (2025).
Lin, H. W., Furman, A. C., Kujawa, S. G. & Liberman, M. C. Primary neural degeneration in the guinea pig cochlea after reversible noise-induced threshold shift. J. Assoc. Res. Otolaryngol. 12, 605–616 (2011).
Du, X. et al. Antioxidants reduce neurodegeneration and accumulation of pathologic Tau proteins in the auditory system after blast exposure. Free Radic. Biol. Med. 108, 627–643 (2017).
Hesse, L. L. et al. Non-monotonic relation between noise exposure severity and neuronal hyperactivity in the auditory midbrain. Front. Neurol. 7, 133. https://doi.org/10.3389/fneur.2016.00133 (2016).
Fernandez, K. A. et al. Noise-induced cochlear synaptopathy with and without sensory cell loss. Neuroscience 427, 43–57. https://doi.org/10.1016/j.neuroscience.2019.11.051 (2020).
Chen, F., Zhao, F., Mahafza, N. & Lu, W. Detecting noise-induced cochlear synaptopathy by auditory brainstem response in tinnitus patients with normal hearing thresholds: A meta-analysis. Front. Neurosci. 15, 778197. https://doi.org/10.3389/fnins.2021.778197 (2021).
Møller, A. R. & Jannetta, P. J. Neural generators of the auditory brainstem response. In The Auditory Brainstem Response (ed. Jacobson, J. T.) 13–31 (College Hill, 1985).
Sergeyenko, Y., Lall, K., Liberman, M. C. & Kujawa, S. G. Age-related cochlear synaptopathy: An early-onset contributor to auditory functional decline. J. Neurosci. 33, 13686–13694 (2013).
Brozoski, T. J., Bauer, C. A. & Caspary, D. M. Elevated fusiform cell activity in the dorsal cochlear nucleus of chinchillas with psychophysical evidence of tinnitus. J. Neurosci. 22, 2383–2390 (2002).
Kaltenbach, J. A. & Afman, C. E. Hyperactivity in the dorsal cochlear nucleus after intense sound exposure and its resemblance to tone-evoked activity: A physiological model for tinnitus. Hear. Res. 140, 165–172 (2000).
Kaltenbach, J. A., Zacharek, M. A., Zhang, J. & Frederick, S. Activity in the dorsal cochlear nucleus of hamsters previously tested for tinnitus following intense tone exposure. Neurosci. Lett. 355, 121–125 (2004).
Wu, C., Martel, D. T. & Shore, S. E. Increased synchrony and bursting of dorsal cochlear nucleus fusiform cells correlate with tinnitus. J. Neurosci. 36, 2068–2073. https://doi.org/10.1523/JNEUROSCI.3960-15.2016 (2016).
Milbrandt, J. C., Holder, T. M., Wilson, M. C., Salvi, R. J. & Caspary, D. M. GAD levels and muscimol binding in rat inferior colliculus following acoustic trauma. Hear. Res. 147, 251–260 (2000).
Wang, H. et al. Plasticity at glycinergic synapses in dorsal cochlear nucleus of rats with behavioral evidence of tinnitus. Neuroscience 164, 747–759 (2009).
Dong, S., Rodger, J., Mulders, W. H. & Robertson, D. Tonotopic changes in GABA receptor expression in guinea pig inferior colliculus after partial unilateral hearing loss. Brain Res. 1342, 24–32 (2010).
Fetoni, A. R. et al. Protective effects of N-acetylcysteine on noise-induced hearing loss in guinea pigs. Acta Otorhinolaryngol. Ital. 29, 70–75 (2009).
Reid, A. J., Shawcross, S. G., Hamilton, A. E., Wiberg, M. & Terenghi, G. N-acetylcysteine alters apoptotic gene expression in axotomised primary sensory afferent subpopulations. Neurosci. Res. 65, 148–155 (2009).
Lu, J. et al. Antioxidants reduce cellular and functional changes induced by intense noise in the inner ear and cochlear nucleus. J. Assoc. Res. Otolaryngol. 15, 353–372 (2014).
Karlidag, T. et al. Evaluation of the effect of methylprednisolone and N-acetylcystein on anastomotic degeneration and regeneration of the facial nerve. Auris Nasus Larynx 39, 145–150 (2012).
Welin, D., Novikova, L. N., Wiberg, M., Kellerth, J. O. & Novikov, L. N. Effects of N-acetyl-cysteine on the survival and regeneration of sural sensory neurons in adult rats. Brain Res. 1287, 58–66. https://doi.org/10.1016/j.brainres.2009.06.038 (2009).
Qian, H. R. & Yang, Y. Neuron differentiation and neuritogenesis stimulated by N-acetylcysteine (NAC). Acta Pharmacol. Sin. 30, 907–912. https://doi.org/10.1038/aps.2009.72 (2009).
Suzuki, J., Corfas, G. & Liberman, M. C. Round-window delivery of neurotrophin 3 regenerates cochlear synapses after acoustic overexposure. Sci. Rep. 6, 24907. https://doi.org/10.1038/srep24907 (2016).
Bramhall, N. F., Bharadwaj, H. M., Verhulst, S. & Heinz, M. G. Perceptual consequences of cochlear deafferentation in humans. Trends Hear. 26, 23312165241239541 (2022).
Bramhall, N. F., McMillan, G. P., Gallun, F. J. & Konrad-Martin, D. Auditory brainstem response demonstrates that reduced peripheral auditory input is associated with self-report of tinnitus. J. Acoust. Soc. Am. 146, 3849 (2019).
Park, E. et al. Evidence of cochlear synaptopathy and the effect of systemic steroid in acute idiopathic tinnitus with normal hearing. Otol. Neurotol. 42, 978–984 (2021).
Guest, H. et al. Impairment of auditory brainstem response wave I in tinnitus patients: Evidence and confounds. Hear. Res. 354, 40–50 (2017).
Buran, B. N., McMillan, G. P., Keshishzadeh, S., Verhulst, S. & Bramhall, N. F. Predicting synapse counts in living humans by combining computational models with auditory physiology. J. Acoust. Soc. Am. 151, 561 (2022).
Schaette, R. & Kempter, R. Development of tinnitus-related neuronal hyperactivity through homeostatic plasticity after hearing loss: A computational model. Eur. J. Neurosci. 23, 3124–3138 (2006).
Wang, Y., Hirose, K. & Liberman, M. C. Dynamics of noise-induced cellular injury and repair in the mouse cochlea. J. Assoc. Res. Otolaryngol. 3, 248–268 (2002).
Sitko, A. A., Frank, M. M., Romero, G. E., Hunt, M. & Goodrich, L. V. Lateral olivocochlear neurons modulate cochlear responses to noise exposure. Proc. Natl. Acad. Sci. U.S.A. 122, e2404558122 (2025).
Guinan, J. J. Jr. Olivocochlear efferents: Their action, effects, measurement and uses, and the impact of the new conception of cochlear mechanical responses. Hear. Res. 362, 38–47 (2018).
Eggermont, J. J. & Roberts, L. E. Tinnitus: Animal models and findings in humans. Cell Tissue Res. 361, 311–336 (2015).
Du, X. et al. Effects of antioxidant treatment on blast-induced brain injury. PLoS ONE 8, e80138. https://doi.org/10.1371/journal.pone.0080138 (2013).
Du, X. et al. Selective degeneration of synapses in the dorsal cochlear nucleus of chinchilla following acoustic trauma and effects of antioxidant treatment. Hear. Res. 283, 1–13 (2012).
Idrizbegovic, E., Bogdanovic, N. & Canlon, B. Modulating calbindin and parvalbumin immunoreactivity in the cochlear nucleus by moderate noise exposure in mice. A quantitative study on the dorsal and posteroventral cochlear nucleus. Brain Res. 800, 86–96 (1998).
Acknowledgements
This research was supported by The Oklahoma Center for the Advancement of Science and Technology (OCAST) grant # AR20-014.
Author information
Authors and Affiliations
Contributions
J.L., X.D., R.D.K. and M.B.W. designed research; J.L., X.D., Z.Y., Q.C., W.C., D.N., W.L. and X.H. performed research; J.L. analyzed data; J.L., X.D. and M.B.W. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
Richard D. Kopke has a financial interest in Otologic Pharmaceutics, Inc. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lu, J., Du, X., Yokell, Z. et al. A delayed pharmacological treatment strategy attenuates noise-induced tinnitus in rats. Sci Rep (2026). https://doi.org/10.1038/s41598-026-40960-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-40960-2