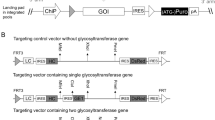
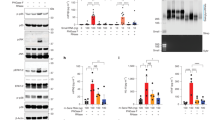
Abstract
Insect cells are attractive hosts for biopharmaceutical production due to their high productivity and mammalian-like post-translational modifications. However, their insect-specific N-glycans differ from mammalian types, thereby reducing product desirability. Here, with an emphasis on engineering N-glycosylation in insect cells, we aimed to develop a more tractable Spodoptera frugiperda Sf9 cell platform as a practical alternative to insect-based systems for engineering the production of tri-antennary N-glycans. A database search revealed that silkworm possesses N-acetylglucosaminyltransferase IV (GNTIV), a putative glycosyltransferase essential for tri-antennary N-glycan synthesis; however, it was not functional in vitro. Then, human GNTIV was introduced into insect cells, resulting in the production of small amounts of tri-antennary N-glycans. This suggested the need for additional factors to efficiently biosynthesize tri-antennary N-glycans. Subsequently, additional insect-derived glycosyltransferases, such as active GNTI and/or GNTII, were co-expressed with GNTIV. Co-expression of three N-acetylglucosaminyltransferases effectively led to the increased biosynthesis of tri-antennary N-glycans. On the other hand, trimming of the N-glycan structure was also observed due to the action of one or more endogenous glycosylhydrolases, which hydrolyze the terminal N-acetylglucosamine residue in insect cells. These facts indicate that effective tri-antennary N-glycan biosynthesis in insect cells requires not only the introduction of exogenous glycosyltransferases but also the knockdown or knockout of endogenous glycosylhydrolase(s).
Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. The gene sequences used in this study, *BmGNTI* , *BmGNTII* , *BmGNTIV* , and *hGNTIV* , were obtained from the databases of KEGG, SGID, and GenBank, with accession numbers KEGG Entry No. 100653407, SGID Entry KWMTBOMO06300 and KWMTBOMO00036, and NM\_012214.3.
References
Kesik-Brodacka, M. Progress in biopharmaceutical development. Biotechnol. Appl. Biochem. 65, 306–322 (2018).
Ayyar, B. V., Arora, S. & Ravi, S. S. Optimizing antibody expression: The nuts and bolts. Methods 116, 51–62 (2017).
Apweiler, R., Hermjakob, H. & Sharon, N. On the frequency of protein glycosylation, as deduced from analysis of the SWISS-PROT database. Biochim. Biophys. Acta Gen. Subj. 1473, 4–8 (1999).
Jacobs, P. & Callewaert, N. N-glycosylation engineering of biopharmaceutical expression systems. Curr. Mol. Med. 9, 774–800 (2009).
Chung, C. Y., Majewska, N. I., Wang, Q., Paul, J. T. & Betenbaugh, M. J. Snapshot: N-glycosylation processing pathways across kingdoms. Cell 171, 258–258 (2017).
Castilho, A. et al. In planta protein sialylation through overexpression of the respective mammalian pathway. J. Biol. Chem. 285, 15923–15930 (2010).
Kallolimath, S. et al. Engineering of complex protein sialylation in plants. Proc. Natl. Acad. Sci. U. S. A. 113, 9498–9503 (2016).
Castilho, A. et al. N-glycosylation engineering of plants for the biosynthesis of glycoproteins with bisected and branched complex N-glycans. Glycobiology 21, 813–823 (2011).
Dicker, M. & Strasser, R. Using glyco-engineering to produce therapeutic proteins. Expert Opin. Biol. Ther. 15, 1501–1516 (2015).
Suganuma, M. et al. N-glycan sialylation in a silkworm-baculovirus expression system. J. Biosci. Bioeng. 126, 9–14 (2018).
Bohlender, L. L. et al. Stable protein sialylation in Physcomitrella. Front. Plant Sci. 11, 610032 (2020).
Hamilton, S. R. et al. Humanization of yeast to produce complex terminally sialylated glycoproteins. Science 313, 1441–1443 (2006).
Shinkawa, T. et al. The absence of fucose but not the presence of galactose or bisecting N-acetylglucosamine of human IgG1 complex-type oligosaccharides shows the critical role of enhancing antibody-dependent cellular cytotoxicity. J. Biol. Chem. 278, 3466–3473 (2003).
Shields, R. L. et al. Lack of fucose on human IgG1 N-linked oligosaccharide improves binding to human FcγRIII and antibody-dependent cellular toxicity. J. Biol. Chem. 277, 26733–26740 (2002).
Umaña, P., Jean-Mairet, J., Moudry, R., Amstutz, H. & Bailey, J. E. Engineered glycoforms of an antineuroblastoma IgG1 with optimized antibody-dependent cellular cytotoxic activity. Nat. Biotechnol. 17, 176–180 (1999).
Davies, J. et al. Expression of GnTIII in a recombinant anti-CD20 CHO production cell line: Expression of antibodies with altered glycoforms leads to an increase in ADCC through higher affinity for FcγRIII. Biotechnol. Bioeng. 74, 288–294 (2001).
Ohtsubo, K. Biological significance of N-acetylglucosaminyltransferase-IV-mediated protein glycosylation on the homeostasis of cellular physiological functions. Trends Glycsci Glycotechnol. 23, 103–105 (2011).
Taniguchi, N. & Korekane, H. Branched N-glycans and their implications for cell adhesion, signaling and clinical applications for cancer biomarkers and in therapeutics. BMB Rep. 44, 772–781 (2011).
Ohtsubo, K. Targeted genetic inactivation of N-acetylglucosaminyltransferase-IVa impairs insulin secretion from pancreatic β cells and evokes type 2 diabetes. Methods Enzymol. 479, 205–222 (2010).
Takeuchi, M. & Kobata, A. Structures and functional roles of the sugar chains of human erythropoietins. Glycobiology 1, 337–346 (1991).
Yin, B. et al. Glycoengineering of Chinese hamster ovary cells for enhanced erythropoietin N-glycan branching and sialylation. Biotechnol. Bioeng. 112, 2343–2351 (2015).
Nagels, B., van Damme, E. J. M., Pabst, M., Callewaert, N. & Weterings, K. Production of complex multiantennary N-glycans in Nicotiana benthamiana plants. Plant Physiol. 155, 1103–1112 (2011).
Nagels, B., Van Damme, E. J. M., Callewaert, N. & Weterings, K. Introduction of tri-antennary N-glycans in Arabidopsis thaliana plants. Plant Sci. 185–186, 161–168 (2012).
Walski, T., De Schutter, K., Van Damme, E. J. M. & Smagghe, G. Diversity and functions of protein glycosylation in insects. Insect Biochem. Mol. Biol. 83, 21–34 (2017).
Stanton, R. et al. The underestimated N-glycomes of lepidopteran species. Biochim. Biophys. Acta Gen. Subj. 1861, 699–714 (2017).
Hykollari, A., Malzl, D., Stanton, R., Eckmair, B. & Paschinger, K. Tissue-specific glycosylation in the honeybee: Analysis of the N-glycomes of Apis mellifera larvae and venom. Biochim. Biophys. Acta Gen. Subj. 1863, 129409 (2019).
Mortimer, N. T., Kacsoh, B. Z., Keebaugh, E. S. & Schlenke, T. A. Mgat1-dependent N-glycosylation of membrane components primes drosophila melanogaster blood cells for the cellular encapsulation response. PLoS Pathog. 8, 31 (2012).
Yamamoto-Hino, M. et al. Phenotype-based clustering of glycosylation-related genes by RNAi-mediated gene silencing. Genes Cells 20, 521–542 (2015).
Yamamoto-Hino, M. et al. Identification of genes required for neural-specific glycosylation using functional genomics. PLoS Genet. 6, 1–11 (2010).
Walski, T. et al. Protein N-glycosylation and N-glycan trimming are required for postembryonic development of the pest beetle Tribolium castaneum. Sci. Rep. 6, 1–15 (2016).
Yang, Q., De Schutter, K., Chen, P., Van Damme, E. J. M. & Smagghe, G. RNAi of the N-glycosylation-related genes confirms their importance in insect development and α-1,6-fucosyltransferase plays a role in the ecdysis event for the hemimetabolous pest insect Nilaparvata lugens. Insect Sci. 29, 91–99 (2022).
Kajiura, H. et al. Bombyx mori β1,4-N-acetylgalactosaminyltransferase possesses relaxed donor substrate specificity in N-glycan synthesis. Sci. Rep. 11, 1–16 (2021).
Kajiura, H., Hamaguchi, Y., Mizushima, H., Misaki, R. & Fujiyama, K. Sialylation potentials of the silkworm, Bombyx mori; B. mori possesses an active α2,6-sialyltransferase. Glycobiology 25, 1441–1453 (2015).
Kajiura, H. et al. Characterization of Bombyx mori N-acetylglucosaminyltransferase II splicing variants. Biochem. Biophys. Res. Commun. 529, 404–410 (2020).
Mabashi-Asazuma, H. et al. Targeted glycoengineering extends the protein N-glycosylation pathway in the silkworm silk gland. Insect Biochem. Mol. Biol. 65, 20–27 (2015).
Mabashi-Asazuma, H. & Jarvis, D. L. CRISPR-Cas9 vectors for genome editing and host engineering in the baculovirus–insect cell system. Proc. Natl. Acad. Sci. U. S. A. 114, 9068–9073 (2017).
Geisler, C., Mabashi-Asazuma, H., Kuo, C. W., Khoo, K. H. & Jarvis, D. L. Engineering β1,4-galactosyltransferase I to reduce secretion and enhance N-glycan elongation in insect cells. J. Biotechnol. 193, 52–65 (2015).
Kato, T. et al. N-glycan modification of a recombinant protein via coexpression of human glycosyltransferases in silkworm pupae. Sci. Rep. 7, 4–5 (2017).
Kato, T., Kajikawa, M., Maenaka, K. & Park, E. Y. Silkworm expression system as a platform technology in life science. Appl. Microbiol. Biotechnol. 85, 459–470 (2010).
Shimomura, M. et al. KAIKObase: An integrated silkworm genome database and data mining tool. BMC Genomics 10, 486 (2009).
Xu, H. The advances and perspectives of recombinant protein production in the silk gland of silkworm Bombyx mori. Transgenic Res. 23, 697–706 (2014).
Paschinger, K. & Wilson, I. B. H. Comparisons of N-glycans across invertebrate phyla. Parasitology 146, 1733–1742 (2019).
Kajiura, H. et al. Temporal analysis of N-acetylglucosamine extension of N-glycans in the middle silk gland of silkworm Bombyx mori. J. Biosci. Bioeng. 133, 533–540 (2022).
Oka, N., Mori, S., Ikegaya, M., Park, E. Y. & Miyazaki, T. Crystal structure and sugar-binding ability of the C-terminal domain of N -acetylglucosaminyltransferase IV establish a new carbohydrate-binding module family. Glycobiology 32, 1153–1163 (2022).
Maghodia, A. B., Geisler, C. & Jarvis, D. L. A new bacmid for customized protein glycosylation pathway engineering in the baculovirus-insect cell system. ACS Chem. Biol. 16, 1941–1950 (2021).
Yan, S. et al. Glycoproteomic and single-protein glycomic analyses reveal zwitterionic N-glycans on natural and recombinant proteins derived from insect cells. Mol. Cell. Proteomics 24, 100981 (2025).
Aoki, K. et al. Dynamic developmental elaboration of N-linked glycan complexity in the Drosophila melanogaster embryo. J. Biol. Chem. 282, 9127–9142 (2007).
Kurz, S. et al. Targeted release and fractionation reveal glucuronylated and sulphated N- and O-glycans in larvae of dipteran insects. J. Proteomics 126, 172–188 (2015).
Juliant, S. et al. Engineering the baculovirus genome to produce galactosylated antibodies in lepidopteran cells. Methods Mol. Biol. 988, 59–77 (2013).
Geisler, C. & Jarvis, D. L. Substrate specificities and intracellular distributions of three N-glycan processing enzymes functioning at a key branch point in the insect N-glycosylation pathway. J. Biol. Chem. 287, 7084–7097 (2012).
Aumiller, J. J., Hollister, J. R. & Jarvis, D. L. Molecular cloning and functional characterization of β-N-acetylglucosaminidase genes from Sf9 cells. Protein Expr. Purif. 47, 571–590 (2006).
Geisler, C., Aumiller, J. J. & Jarvis, D. L. A fused lobes gene encodes the processing β-N-acetylglucosaminidase in Sf9 cells. J. Biol. Chem. 283, 11330–11339 (2008).
Friesen, P., Scotti, P., Longworth, J. & Rueckert, R. Black beetle virus: Propagation in Drosophila line 1 cells and an infection-resistant subline carrying endogenous black beetle virus-related particles. J. Virol. 35, 741–747 (1980).
Li, T.-C., Scotti, P. D., Miyamura, T. & Takeda, N. Latent infection of a new alphanodavirus in an insect cell line. J. Virol. 81, 10890–10896 (2007).
Ma, H., Galvin, T. A., Glasner, D. R., Shaheduzzaman, S. & Khan, A. S. Identification of a novel rhabdovirus in Spodoptera frugiperda cell lines. J. Virol. 88, 6576–6585 (2014).
Maghodia, A. B., Geisler, C. & Jarvis, D. L. Characterization of an Sf-rhabdovirus-negative Spodoptera frugiperda cell line as an alternative host for recombinant protein production in the baculovirus-insect cell system. Protein Expr. Purif. 122, 45–55 (2016).
Tamura, T. et al. Germline transformation of the silkworm Bombyx mori L. using a piggyBac transposon-derived vector. Nat. Biotechnol. 18, 559 (2000).
Tatematsu, K. et al. Construction of a binary transgenic gene expression system for recombinant protein production in the middle silk gland of the silkworm Bombyx mori. Transgenic Res. 19, 473–487 (2010).
Tsubota, T. & Sezutsu, H. Genome editing of silkworms. Methods Mol. Biol. 1630, 205–218 (2017).
Fujita, R. et al. Efficient production of recombinant SARS-CoV-2 spike protein using the baculovirus-silkworm system. Biochem. Biophys. Res. Commun. 529, 257–262 (2020).
Martínez-Solís, M., Herrero, S. & Targovnik, A. M. Engineering of the baculovirus expression system for optimized protein production. Appl. Microbiol. Biotechnol. 103, 113–123 (2019).
Van Die, I., Van Tetering, A., Bakker, H., Van Den Eijnden, D. H. & Joziasse, D. H. Glycosylation in lepidopteran insect cells: Identification of a β1 → 4-N-acetylgalactosaminyltransferase involved in the synthesis of complex-type oligosaccharide chains. Glycobiology 6, 157–164 (1996).
Haines, N. & Irvine, K. D. Functional analysis of Drosophila β1,4-N- acetlygalactosaminyltransferases. Glycobiology 15, 335–346 (2005).
Paschinger, K., Staudacher, E., Stemmer, U., Fabini, G. & Wilson, I. B. H. Fucosyltransferase substrate specificity and the order of fucosylation in invertebrates. Glycobiology 15, 463–474 (2005).
Bakker, H. et al. An antibody produced in tobacco expressing a hybrid β-1,4- galactosyltransferase is essentially devoid of plant carbohydrate epitopes. Proc. Natl. Acad. Sci. U. S. A. 103, 7577–7582 (2006).
Hesselink, T. et al. Expression of natural human β1,4-GalT1 variants and of non-mammalian homologues in plants leads to differences in galactosylation of N-glycans. Transgenic Res. 23, 717–728 (2014).
Castilho, A. et al. Rapid high yield production of different glycoforms of Ebola virus monoclonal antibody. PLoS ONE 6, e26040 (2011).
Strasser, R. et al. Improved virus neutralization by plant-produced anti-HIV antibodies with a homogeneous β1,4-galactosylated N-glycan profile. J. Biol. Chem. 284, 20479–20485 (2009).
Frey, A. D., Karg, S. R. & Kallio, P. T. Expression of rat β(1,4)-N-acetylglucosaminyltransferase III in Nicotiana tabacum remodels the plant-specific N-glycosylation. Plant Biotechnol. J. 7, 33–48 (2009).
Léonard, R. et al. The Drosophila fused lobes gene encodes an N-acetylglucosaminidase involved in N-glycan processing. J. Biol. Chem. 281, 4867–4875 (2006).
Kim, N. Y. et al. Short-hairpin RNA-mediated gene expression interference in Trichoplusia ni cells. J. Microbiol. Biotechnol. 22, 190–198 (2012).
Watanabe, S. et al. Sialylation of N-glycans on the recombinant proteins expressed by a baculovirus-insect cell system under β-N-acetylglucosaminidase inhibition. J. Biol. Chem. 277, 5090–5093 (2002).
Nomura, T. et al. Improvement of glycosylation structure by suppression of β-N-acetylglucosaminidases in silkworm. J. Biosci. Bioeng. 119, 131–136 (2015).
Kokuho, T., Yasukochi, Y., Watanabe, S. & Inumaru, S. Molecular cloning and expression profile analysis of a novel β-D-N-acetylhexosaminidase of domestic silkworm (Bombyx mori). Genes Cells 15, 525–536 (2010).
Kajiura, H., Seki, T. & Fujiyama, K. Arabidopsis thaliana ALG3 mutant synthesizes immature oligosaccharides in the ER and accumulates unique N-glycans. Glycobiology 20, 736–751 (2010).
Acknowledgements
We gratefully thank Dr. Ken-ichiro Tatematsu and Hideki Sezutsu (Division of Silk-Producing Insect Biotechnology, Institute of Agrobiological Sciences, National Agriculture and Food Research Organization, Japan) for providing the silkworm type strain used to prepare B. mori cDNA.
Funding
This work was supported by the Ministry of Agriculture, Forestry and Fisheries (MAFF), Japan, under the commissioned research project “Scientific technique research promotion program for agriculture, forestry, fisheries and food industry” (Project Nos. 1301 and 2201).
Author information
Authors and Affiliations
Contributions
H.K. and K.F. designed the research with assistance from R.M. H.K, N.N., and Y.S. produced the recombinant proteins. R.L.S.S.-C. performed MS analysis of the reaction products. H.K. and N.N. performed all the other experiments. H.K. wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kajiura, H., Nishiguchi, N., Sawada-Choi, R.L.S. et al. N-Glycoengineering of insect cells for tri-antennary N-Glycan biosynthesis. Sci Rep (2026). https://doi.org/10.1038/s41598-026-41152-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-41152-8