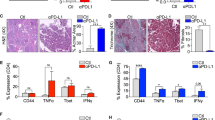
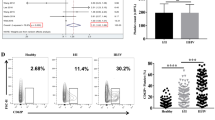
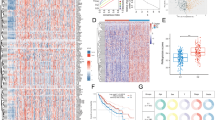
Abstract
Pancreatic ductal adenocarcinoma (PDAC) has a poor survival rate and limited treatments. Agonistic CD40 antibodies are promising, but clinical trials have shown only modest efficacy and significant hepatotoxicity. We previously reported that IL-1 pathway blockade enhances CD40 agonist efficacy against melanoma by depleting polymorphonuclear myeloid-derived suppressor cells (PMN-MDSCs; CD11b+Ly6C+Ly6G+). Because PMN-MDSCs also cause liver toxicity, we investigated the impact of IL-1R1 blockade on the efficacy and toxicity of agonistic CD40 antibody therapy in PDAC. Agonistic CD40 antibody therapy induced immune activation and significantly prolonged survival in orthotopic PDAC-bearing mice. The combination of an agonistic CD40 antibody and IL-1R1 blockade upregulated multiple immune-related pathways and enhanced innate and adaptive responses. However, it did not further improve CD40 efficacy or reduce liver toxicity. The efficacy of the CD40 agonist was partially dependent on CD8⁺ T cells. Our findings underscore the complex role of IL-1 signaling in modulating immune responses in PDAC and caution against pursuing IL-1R1 blockade, either as monotherapy or combined with agonistic CD40 antibodies, in clinical trials for PDAC.
Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request and deposited in NCBI Gene Expression Omnibus (GEO) database with accession number GEO: GSE301673 and will be publicly available as of the date of publication.
References
Mizrahi, J. D. et al. Pancreatic cancer. Lancet 395 (10242), 2008–2020 (2020).
Rahib, L. et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 74 (11), 2913–2921 (2014).
Kabacaoglu, D. et al. Immune checkpoint inhibition for pancreatic ductal adenocarcinoma: Current limitations and future options. Front. Immunol. 9, 1878 (2018).
Schizas, D. et al. Immunotherapy for pancreatic cancer: A 2020 update. Cancer Treat. Rev. 86, 102016 (2020).
Vonderheide, R. H. CD40 Agonist Antibodies in Cancer Immunotherapy. Annu. Rev. Med. 71, 47–58 (2020).
Beatty, G. L. et al. CD40 agonists alter tumor stroma and show efficacy against pancreatic carcinoma in mice and humans. Science 331 (6024), 1612–1616 (2011).
Li, D. K. & Wang, W. Characteristics and clinical trial results of agonistic anti-CD40 antibodies in the treatment of malignancies. Oncol. Lett. 20 (5), 176 (2020).
Padron, L. J. et al. Sotigalimab and/or nivolumab with chemotherapy in first-line metastatic pancreatic cancer: clinical and immunologic analyses from the randomized phase 2 PRINCE trial. Nat. Med. 28 (6), 1167–1177 (2022).
Weiss, S. A. et al. A phase II Trial of the CD40 agonistic antibody sotigalimab (APX005M) in combination with nivolumab in subjects with metastatic melanoma with confirmed disease progression on anti-PD-1 therapy. Clin. Cancer Res. 30(1), 74–81 (2024).
Johnson, P. et al. Clinical and biological effects of an agonist anti-CD40 antibody: a cancer research UK phase I study. Clin. Cancer Res. 21(6), 1321–1328 (2015).
Vonderheide, R. H. et al. Clinical activity and immune modulation in cancer patients treated with CP-870,893, a novel CD40 agonist monoclonal antibody. J. Clin. Oncol. 25 (7), 876–883 (2007).
Medina-Echeverz, J. et al. Systemic agonistic anti-CD40 treatment of tumor-bearing mice modulates hepatic myeloid-suppressive cells and causes immune-mediated liver damage. Cancer Immunol. Res. 3(5), 557–566 (2015).
Blake, S. J. et al. The immunotoxicity, but not anti-tumor efficacy, of anti-CD40 and anti-CD137 immunotherapies is dependent on the gut microbiota. Cell. Rep. Med. 2 (12), 100464 (2021).
Hussain, S. P. & Harris, C. C. Inflammation and cancer: an ancient link with novel potentials. Int. J. Cancer. 121 (11), 2373–2380 (2007).
Dinarello, C. A. Proinflammatory cytokines. Chest 118 (2), 503–508 (2000).
Voronov, E. et al. IL-1 is required for tumor invasiveness and angiogenesis. Proc. Natl. Acad. Sci. U.S.A. 100(5), 2645–2650 (2003).
Lewis, A. M. et al. Interleukin-1 and cancer progression: the emerging role of interleukin-1 receptor antagonist as a novel therapeutic agent in cancer treatment. J. Transl Med. 4, 48 (2006).
Lee, P. Y. et al. IL-1alpha modulates neutrophil recruitment in chronic inflammation induced by hydrocarbon oil. J. Immunol. 186 (3), 1747–1754 (2011).
Brinster, C. & Shevach, E. M. Costimulatory effects of IL-1 on the expansion/differentiation of CD4 + CD25+Foxp3 + and CD4 + CD25+Foxp3- T cells. J. Leukoc. Biol. 84 (2), 480–487 (2008).
Cahill, C. M. & Rogers, J. T. Interleukin (IL) 1beta induction of IL-6 is mediated by a novel phosphatidylinositol 3-kinase-dependent AKT/IkappaB kinase alpha pathway targeting activator protein-1. J. Biol. Chem. 283 (38), 25900–25912 (2008).
Jung, Y. D. et al. Role of P38 MAPK, AP-1, and NF-kappaB in interleukin-1beta-induced IL-8 expression in human vascular smooth muscle cells. Cytokine 18 (4), 206–213 (2002).
Zhang, D. et al. STAT3 participates in transcriptional activation of the C-reactive protein gene by interleukin-6. J. Biol. Chem. 271 (16), 9503–9509 (1996).
Amlani, A. et al. Successful treatment of cytokine release syndrome with IL-6 blockade in a patient transitioning from immune-checkpoint to MEK/BRAF inhibition: A case report and review of literature. Oncologist 25(7), e1120–e1123 (2020).
Yoshino, K. et al. Severe colitis after PD-1 blockade with nivolumab in advanced melanoma patients: Potential role of Th1-dominant immune response in immune-related adverse events: Two case reports. BMC Cancer 19(1), 1019 (2019).
Schalper, K. A. et al. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and reduced clinical benefit of immune-checkpoint inhibitors. Nat. Med. 26 (5), 688–692 (2020).
Zhang, Y. et al. Interleukin-17-induced neutrophil extracellular traps mediate resistance to checkpoint blockade in pancreatic cancer. J. Exp. Med. https://doi.org/10.1084/jem.20190354 (2020).
Tarhini, A. A. et al. Baseline circulating IL-17 predicts toxicity while TGF-beta1 and IL-10 are prognostic of relapse in ipilimumab neoadjuvant therapy of melanoma. J. Immunother Cancer. 3, 39 (2015).
Singh, S. et al. IL-1alpha mediates innate and acquired resistance to immunotherapy in melanoma. J. Immunol. 206(8), 1966–1975 (2021).
Jaillon, S. et al. Neutrophil diversity and plasticity in tumour progression and therapy. Nat. Rev. Cancer. 20 (9), 485–503 (2020).
Kapanadze, T. et al. Tumor-induced CD11b(+) Gr-1(+) myeloid-derived suppressor cells exacerbate immune-mediated hepatitis in mice in a CD40-dependent manner. Eur. J. Immunol. 45 (4), 1148–1158 (2015).
Ager, C. R. et al. High potency STING agonists engage unique myeloid pathways to reverse pancreatic cancer immune privilege. J. Immunother. Cancer https://doi.org/10.1136/jitc-2021-003246 (2021).
Kanehisa, M. et al. KEGG: biological systems database as a model of the real world. Nucleic Acids Res. 53 (D1), D672–D677 (2025).
Fan, Y. C. et al. Tumor-derived interleukin-1 receptor antagonist exhibits immunosuppressive functions and promotes pancreatic cancer. Cell. Biosci. 13 (1), 147 (2023).
Das, S. et al. Tumor cell-derived IL1beta promotes desmoplasia and immune suppression in pancreatic cancer. Cancer Res. 80(5), 1088–1101 (2020).
Takahashi, R. et al. Interleukin-1beta-induced pancreatitis promotes pancreatic ductal adenocarcinoma via B lymphocyte-mediated immune suppression. Gut 70 (2), 330–341 (2021).
Aggen, D. H. et al. Blocking IL1 beta promotes tumor regression and remodeling of the myeloid compartment in a renal cell carcinoma model: Multidimensional analyses. Clin. Cancer Res. 27(2), 608–621 (2021).
Herremans, K. M. et al. The interleukin-1 axis and the tumor immune microenvironment in pancreatic ductal adenocarcinoma. Neoplasia 28, 100789 (2022).
Singh, M. et al. Intratumoral CD40 activation and checkpoint blockade induces T cell-mediated eradication of melanoma in the brain. Nat. Commun. 8 (1), 1447 (2017).
Dosch, A. R. et al. Targeting tumor-stromal IL6/STAT3 signaling through IL1 receptor inhibition in pancreatic cancer. Mol. Cancer Ther. 20(11), 2280–2290 (2021).
Barbier, L. et al. Interleukin-1 family cytokines: Keystones in liver inflammatory diseases. Front. Immunol. 10, p2014 (2019).
Acknowledgements
We thank Erica Goodoff, Senior Scientific Editor in the Research Medical Library at The University of Texas MD Anderson Cancer Center, for editing this article.
Funding
This work was supported by the University Cancer Foundation via the Institutional Research Grant program at The University of Texas MD Anderson Cancer Center and Hirshberg Foundation for Pancreatic Cancer Research (to M.S.) and National Institutes of Health/National Cancer Institute Grant P30CA016672, which supports the flow cytometry facility at The University of Texas MD Anderson Cancer Center. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Conception, design and study supervision: MS, Development of methodology: MS, AB, INB, SC, SA, AA, VM, NWF, JR, Resources and critical comments on manuscript: MAC, VKM, SK, Writing the manuscript: MS.
Corresponding author
Ethics declarations
Competing interests
MAC received grants from ImmunoMet, Inc. and personal fees from ImmunoGenesis, Inc., Alligator Bioscience, Inc., ImmunoOS, Inc., ImmunoMet, Inc., Oncoresponse, Inc., Pieris, Inc., Nurix, Inc., Apetevo, Inc., Servier, Inc., Kineta, Inc., Salarius, Inc., Xencor, Inc., Agenus, Inc., and Mereo. In addition, MAC also has a patent Methods and Composition for Localized Secretion of Anti-CTLA-4 Antibodies that has been issued and licensed to multiple licensees, and a patent Dual specifically antibodies which bind both PD-L1 and PD-L2 and prevent their binding to PD-1 that is pending and licensed with ImmunoGenesis, Inc. SK has ownership interest in Lutris, Iylon, Frontier Medicines, Xilis, Navire and is a consultant for Genentech, EMD Serono, Merck, Holy Stone Healthcare, Novartis, Lilly, Boehringer Ingelheim, AstraZeneca/MedImmune, Bayer Health, Redx Pharma, Ipsen, HalioDx, Lutris, Jacobio, Pfizer, Repare Therapeutics, Inivata, GlaxoSmithKline, Jazz Pharmaceuticals, Iylon, Xilis, Abbvie, Amal Therapeutics, Gilead Sciences, Mirati Therapeutics, Flame Biosciences, Servier, Carina Biotech, Bicara Therapeutics, Endeavor BioMedicines, Numab, Johnson & Johnson/Janssen, Genomic Health, Frontier Medicines, Replimune, TaihoPharmaceutical, Cardiff Oncology, Ono Pharmaceutical, Bristol-Myers Squibb-Medarex, Amgen, Tempus, Foundation Medicine, Harbinger Oncology, Inc, Takeda, CureTeq, Zentalis, Black Stone Therapeutics, NeoGenomics Laboratories, Accademia Nazionale Di Medicina, Tachyon Therapeutics and receive research funding from Sanofi, Biocartis, Guardant Health, Array BioPharma, Genentech/Roche, EMD Serono, MedImmune, Novartis, Amgen, Lilly, Daiichi Sankyo. VKM reports research support to BioNTech, Pfizer, Bristol Myers Squibb, Genentech, RedX Pharma, and Apollo Therapeutics institutions and consulting for Regeneron, Bristol Myers Squibb, and Novartis outside the submitted work. MS, AB, INB, SC, SA, AA, VM, NWF, and JR declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Boda, A., Bandey, I.N., Chowdhury, S. et al. IL1R1 blockade augments CD40 agonist mediated immunity in pancreatic cancer. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42259-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42259-8