Abstract
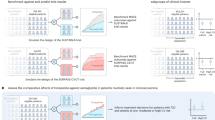
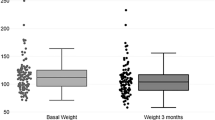
This study aimed to investigate the anti-atherosclerotic properties of tirzepatide, a dual glucose-dependent insulinotropic polypeptide (GIP)/glucagon-like peptide-1 (GLP-1) receptor agonist, in comparison with semaglutide, a selective GLP-1 receptor agonist. ApoE knockout mice were divided into early diabetes (dosed from 10 to 22 weeks of age), late diabetes (dosed from 18 to 30 weeks of age), and non-diabetic groups after streptozotocin treatment, and each group received semaglutide, tirzepatide, or saline for 12 weeks. In the early diabetes group, both agents significantly suppressed aortic plaque formation compared with control, while modestly improving glycemia and lipid levels. No significant vascular effects were observed in late diabetes or non-diabetic groups. Tirzepatide markedly reduced inflammatory mediators, including Mcp-1, Il-6, I-cam, and Cd68, whereas semaglutide showed partial overlap. Notably, these anti-inflammatory effects were also detected in non-diabetic mice, suggesting vascular protection may involve arterial actions independently of metabolic control. Taken together, our findings demonstrate that tirzepatide exerts anti-atherosclerotic effects comparable to semaglutide, supporting the concept that GIP and GLP-1 signaling can confer vascular benefits. These results highlight the potential clinical relevance of dual incretin receptor agonism for cardiovascular risk reduction, although further studies are required to clarify the specific role of GIP signaling.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Bjorkegren, J. L. M. & Lusis, A. J. Atherosclerosis: Recent developments. Cell 185, 1630–1645. https://doi.org/10.1016/j.cell.2022.04.004 (2022).
Kaur, R., Kaur, M. & Singh, J. Endothelial dysfunction and platelet hyperactivity in type 2 diabetes mellitus: Molecular insights and therapeutic strategies. Cardiovasc. Diabetol. 17, 121. https://doi.org/10.1186/s12933-018-0763-3 (2018).
Stratton, I. M. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): Prospective observational study. BMJ 321, 405–412. https://doi.org/10.1136/bmj.321.7258.405 (2000).
Edgar, L. et al. Hyperglycemia induces trained immunity in macrophages and their precursors and promotes atherosclerosis. Circulation 144, 961–982. https://doi.org/10.1161/CIRCULATIONAHA.120.046464 (2021).
Marso, S. P. et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 375, 311–322. https://doi.org/10.1056/NEJMoa1603827 (2016).
Marso, S. P. et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 375, 1834–1844. https://doi.org/10.1056/NEJMoa1607141 (2016).
Husain, M. et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 381, 841–851. https://doi.org/10.1056/NEJMoa1901118 (2019).
Hernandez, A. F. et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (harmony outcomes): A double-blind, randomised placebo-controlled trial. Lancet 392, 1519–1529. https://doi.org/10.1016/S0140-6736(18)32261-X (2018).
Gerstein, H. C. et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 394, 121–130. https://doi.org/10.1016/S0140-6736(19)31149-3 (2019).
Kimura, T. et al. Decreased glucagon-like peptide 1 receptor expression in endothelial and smooth muscle cells in diabetic db/db mice: TCF7L2 is a possible regulator of the vascular glucagon-like peptide 1 receptor. Diab. Vasc. Dis. Res. 14, 540–548. https://doi.org/10.1177/1479164117725898 (2017).
Kimura, T. et al. Down-regulation of vascular GLP-1 receptor expression in human subjects with obesity. Sci. Rep. 8, 10644. https://doi.org/10.1038/s41598-018-28849-1 (2018).
Taguchi, K. et al. Glucagon-like peptide-1 increased the vascular relaxation response via AMPK/Akt signaling in diabetic mice aortas. Eur. J. Pharmacol. 865, 172776. https://doi.org/10.1016/j.ejphar.2019.172776 (2019).
Taguchi, K., Kaneko, N., Okudaira, K., Matsumoto, T. & Kobayashi, T. GLP‐1 modulates insulin‐induced relaxation response through β‐arrestin2 regulation in diabetic mice aortas. Acta Physiol. (Oxf.) 231, e13573. https://doi.org/10.1111/apha.13573 (2021).
Gaspari, T. et al. A GLP-1 receptor agonist liraglutide inhibits endothelial cell dysfunction and vascular adhesion molecule expression in an ApoE-/- mouse model. Diab. Vasc. Dis. Res. 8, 117–124. https://doi.org/10.1177/1479164111404257 (2013).
Guo, C. et al. Glucagon-like peptide 1 improves insulin resistance in vitro through anti-inflammation of macrophages. Braz. J. Med. Biol. Res. 49, e5826. https://doi.org/10.1590/1414-431X20165826 (2016).
Vinue, A. et al. The GLP-1 analogue lixisenatide decreases atherosclerosis in insulin-resistant mice by modulating macrophage phenotype. Diabetologia 60, 1801–1812. https://doi.org/10.1007/s00125-017-4330-3 (2017).
Gaspari, T., Welungoda, I., Widdop, R. E., Simpson, R. W. & Dear, A. E. The GLP-1 receptor agonist liraglutide inhibits progression of vascular disease via effects on atherogenesis, plaque stability and endothelial function in an ApoE(-/-) mouse model. Diab. Vasc. Dis. Res. 10, 353–360. https://doi.org/10.1177/1479164113481817 (2013).
Li, P., Tang, Z., Wang, L. & Feng, B. Glucagon-like peptide-1 analogue liraglutide ameliorates atherogenesis via inhibiting advanced glycation end product-induced receptor for advanced glycosylation end product expression in Apolipoprotein-E deficient mice. Mol. Med. Rep. 16, 3421–3426. https://doi.org/10.3892/mmr.2017.6978 (2017).
Jojima, T. et al. Liraglutide, a GLP-1 receptor agonist, inhibits vascular smooth muscle cell proliferation by enhancing AMP-activated protein kinase and cell cycle regulation, and delays atherosclerosis in ApoE deficient mice. Atherosclerosis 261, 44–51. https://doi.org/10.1016/j.atherosclerosis.2017.04.001 (2017).
Park, B. et al. GLP-1 receptor agonists and atherosclerosis protection: The vascular endothelium takes center stage. Am. J. Physiol. Heart Circ. Physiol. 326, H1159–H1176. https://doi.org/10.1152/ajpheart.00574.2023 (2024).
Samms, R. J. et al. GIPR agonism mediates weight-independent insulin sensitization by tirzepatide in obese mice. J. Clin. Invest. https://doi.org/10.1172/JCI146353 (2021).
Nogi, Y. et al. Glucose-dependent insulinotropic polypeptide prevents the progression of macrophage-driven atherosclerosis in diabetic Apolipoprotein E-null mice. PLoS ONE 7, e35683. https://doi.org/10.1371/journal.pone.0035683 (2012).
Kahles, F. et al. The incretin hormone GIP is upregulated in patients with atherosclerosis and stabilizes plaques in ApoE(-/-) mice by blocking monocyte/macrophage activation. Mol. Metab. 14, 150–157. https://doi.org/10.1016/j.molmet.2018.05.014 (2018).
Pujadas, G. et al. Genetic disruption of the Gipr in Apoe(-/-) mice promotes atherosclerosis. Mol. Metab. 65, 101586. https://doi.org/10.1016/j.molmet.2022.101586 (2022).
Xia, Y. et al. Tirzepatide’s role in targeting adipose tissue macrophages to reduce obesity-related inflammation and improve insulin resistance. Int. Immunopharmacol. 143, 113499. https://doi.org/10.1016/j.intimp.2024.113499 (2024).
Regmi, A. et al. Tirzepatide modulates the regulation of adipocyte nutrient metabolism through long-acting activation of the GIP receptor. Cell Metab. 36, 1898–1899. https://doi.org/10.1016/j.cmet.2024.06.012 (2024).
Xu, G. et al. Downregulation of GLP-1 and GIP receptor expression by hyperglycemia: Possible contribution to impaired incretin effects in diabetes. Diabetes 56, 1551–1558. https://doi.org/10.2337/db06-1033 (2007).
Rakipovski, G. et al. The GLP-1 analogs liraglutide and semaglutide reduce atherosclerosis in ApoE(-/-) and LDLr(-/-) mice by a mechanism that includes inflammatory pathways. JACC Basic Transl. Sci. 3, 844–857. https://doi.org/10.1016/j.jacbts.2018.09.004 (2018).
Pan, X. et al. Semaglutide alleviates inflammation-induced endothelial progenitor cells injury by inhibiting MiR-155 expression in macrophage exosomes. Int. Immunopharmacol. 119, 110196. https://doi.org/10.1016/j.intimp.2023.110196 (2023).
Liu, Q., Zhu, J., Kong, B., Shuai, W. & Huang, H. Tirzepatide attenuates lipopolysaccharide-induced left ventricular remodeling and dysfunction by inhibiting the TLR4/NF-kB/NLRP3 pathway. Int. Immunopharmacol. 120, 110311. https://doi.org/10.1016/j.intimp.2023.110311 (2023).
van Eenige, R. et al. Combined glucose-dependent insulinotropic polypeptide receptor and glucagon-like peptide-1 receptor agonism attenuates atherosclerosis severity in APOE*3-Leiden.CETP mice. Atherosclerosis 372, 19–31. https://doi.org/10.1016/j.atherosclerosis.2023.03.016 (2023).
Willard, F. S. et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight https://doi.org/10.1172/jci.insight.140532 (2020).
Liu, Q. K. Mechanisms of action and therapeutic applications of GLP-1 and dual GIP/GLP-1 receptor agonists. Front. Endocrinol. (Lausanne) 15, 1431292. https://doi.org/10.3389/fendo.2024.1431292 (2024).
Berglund, L. M. et al. Glucose-dependent insulinotropic polypeptide stimulates osteopontin expression in the vasculature via endothelin-1 and CREB. Diabetes 65, 239–254. https://doi.org/10.2337/db15-0122 (2016).
Koska, J. Incretins and preservation of endothelial function. Cardiovasc. Hematol. Agents Med. Chem. 10, 295–308. https://doi.org/10.2174/187152512803530289 (2012).
Mori, Y. et al. Glucose-dependent insulinotropic polypeptide suppresses peripheral arterial remodeling in male mice. Endocrinology 159, 2717–2732. https://doi.org/10.1210/en.2018-00336 (2018).
Heimburger, S. M. et al. Glucose-dependent insulinotropic polypeptide (GIP) and cardiovascular disease. Peptides 125, 170174. https://doi.org/10.1016/j.peptides.2019.170174 (2020).
Sanada, J. et al. Dulaglutide exerts beneficial anti atherosclerotic effects in ApoE knockout mice with diabetes: The earlier, the better. Sci. Rep. 11, 1425. https://doi.org/10.1038/s41598-020-80894-x (2021).
Iwamoto, Y. et al. Tirzepatide, a dual glucose‐dependent insulinotropic polypeptide/glucagon‐like peptide 1 receptor agonist, exhibits favourable effects on pancreatic β‐cells and hepatic steatosis in obese type 2 diabetic db/db mice. Diabetes Obes. Metab. 26, 5982–5994. https://doi.org/10.1111/dom.15972 (2024).
Ma, T. et al. Distinct effects of semaglutide and tirzepatide on metabolic and inflammatory gene expression in brown adipose tissue of mice fed a high-fat, high-fructose diet. Front. Nutr. 12, 1659233. https://doi.org/10.3389/fnut.2025.1659233 (2025).
Acknowledgements
We thank central research institute of Kawasaki Medical School for technical supports of experiments.
Funding
This study was partially supported by a Research Project Grant from the Japan Arteriosclerosis Prevention Fund (Grant No. 2024-1-014) and by a grant from Kawasaki Medical School (Grant No. R05G004).
Author information
Authors and Affiliations
Contributions
J.S, T.K. and H.K. designed this experiment. K.D., J.S., Y.N. and Y.S. performed the experiments. J.S. and T.K. supervised the experimental procedures. K.D., J.S., and T.K. wrote the manuscript. Y.I., T.S., H.I., Y.F., M.S., S.N. and K.K. participated in discussion. H.K. participated in discussion and reviewed the manuscript. All authors approved the submission of the final version of this manuscript.
Corresponding author
Ethics declarations
Competing interests
T.K. has received honoraria for lectures from Novo Nordisk Pharma and Sumitomo Pharma. S. N. has received honoraria for lectures from Novo Nordisk Pharma and Daiichi Sankyo. K.K. has served as an advisor to, received honoraria for lectures from and received scholarship grants from Novo Nordisk Pharma, Sanwa Kagaku, Taisho Pharma, Kowa, Sumitomo Pharma, Mitsubishi Tanabe Pharma, Astellas and Boehringer Ingelheim. H.K. has received honoraria for lectures, scholarship grants and research grants from Novo Nordisk Pharma, Sanofi, Eli Lilly, Boehringer Ingelheim, Taisho Pharma, Sumitomo Pharma, Takeda Pharma, Ono Pharma, Daiichi Sankyo, Mitsubishi Tanabe Pharma, Kissei Pharma, MSD, AstraZeneca, Astellas, Novartis, Kowa and Abbott. All other authors have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dan, K., Sanada, J., Kimura, T. et al. Early intervention with tirzepatide or semaglutide influences anti-atherosclerotic effects in ApoE knockout mice. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42437-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42437-8