Abstract
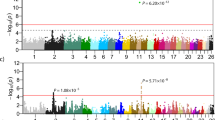
Body axis patterning in vertebrates is controlled by HOX genes during embryogenesis, with their expression gradients defining spatial identity along the anterior–posterior axis. While the developmental roles of HOX genes in axial patterning are well established, it remains unclear to what extent embryonically defined anterior–posterior HOX expression patterns are retained as transcriptional signatures in adult tissues. Tail length in sheep provides a model for studying the phenotypic effects of HOX gene variants. We used Improved Jezersko–Solčava sheep, which segregate for both tail length and HOXB13 promoter variants, to examine genotype–phenotype associations. The HOXB13 genotype was the main determinant of adult tail length, acting primarily by modulating caudal vertebral number. RNA‑seq and qPCR analyses revealed a clear anterior–posterior HOXB13 expression gradient in adult tail skin and bones, consistent with retained positional patterning, with stronger distal expression in short‑tailed animals. These results provide in vivo evidence that spatially patterned HOX gene expression is retained in adulthood, consistent with residual transcriptional signatures of embryonic anterior–posterior identity.
Similar content being viewed by others
Data availability
RNA-Seq data were deposited into the Gene Expression Omnibus database under accession number GSE311823 and are available at the following URL: [https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE311823].
References
Wellik, D. M. Hox patterning of the vertebrate axial skeleton. Dev. dynamics: official publication Am. Association Anatomists. 236, 2454–2463 (2007).
Hubert, K. A. & Wellik, D. M. Hox genes in development and beyond. Development 150, 1–10 (2023).
Mallo, M. The vertebrate tail: a gene playground for evolution. Cell. Mol. Life Sci. 77, 1021–1030 (2020).
Kingsley, E. P. et al. Adaptive tail-length evolution in deer mice is associated with differential Hoxd13 expression in early development. Nat. Ecol. Evol. 8, 791–805 (2024).
Aires, R. et al. Tail bud progenitor activity relies on a network comprising Gdf11, Lin28, and Hox13 genes. Dev. Cell. 48, 383–395 (2019). e388.
Zeltser, L., Desplan, C. & Heintz, N. Hoxb-13: a new Hox gene in a distant region of the HOXB cluster maintains colinearity. Development 122, 2475–2484 (1996).
Young, T. et al. Cdx and Hox genes differentially regulate posterior axial growth in mammalian embryos. Dev. Cell. 17, 516–526 (2009).
Hung, Y. S. et al. Protogenin facilitates trunk-to-tail HOX code transition via modulating GDF11/SMAD2 signaling in mammalian embryos. Commun. Biology. 7, 1669 (2024).
Kalds, P., Huang, S., Chen, Y. & Wang, X. Ovine HOXB13: expanding the gene repertoire of sheep tail patterning and implications in genetic improvement. Commun. Biology. 5, 1196 (2022).
Li, X. et al. Whole-genome resequencing of wild and domestic sheep identifies genes associated with morphological and agronomic traits. Nat. Commun. 11, 2815 (2020).
Manzari, Z., Mehrabani-Yeganeh, H., Nejati‐Javaremi, A., Moradi, M. & Gholizadeh, M. Detecting selection signatures in three Iranian sheep breeds. Anim. Genet. 50, 298–302 (2019).
Ahbara, A. M. et al. Natural adaptation and human selection of northeast African sheep genomes. Genomics 114, 110448 (2022).
Li, R. et al. A sheep pangenome reveals the spectrum of structural variations and their effects on tail phenotypes. Genome Res. 33, 463–477 (2023).
Lagler, D. K. et al. Fine-mapping and identification of candidate causal genes for tail length in the Merinolandschaf breed. Commun. biology. 5, 918 (2022).
Shelton, M. Studies on tail length of Rambouillet and Mouflon sheep. J. heredity. 68, 128–130 (1977).
Zhi, D. et al. Whole genome sequencing of Hulunbuir short-tailed sheep for identifying candidate genes related to the short-tail phenotype. G3: Genes Genomes Genet. 8, 377–383 (2018).
Han, J. et al. Two linked TBXT (brachyury) gene polymorphisms are associated with the tailless phenotype in fat-rumped sheep. Anim. Genet. 50, 772–777 (2019).
Li, X. et al. Genomic analyses of wild argali, domestic sheep, and their hybrids provide insights into chromosome evolution, phenotypic variation, and germplasm innovation. Genome Res. 32, 1669–1684 (2022).
Dong, K. et al. Genomic analysis of worldwide sheep breeds reveals PDGFD as a major target of fat-tail selection in sheep. BMC Genom. 21, 1–12 (2020).
Pan, Z. et al. Rapid evolution of a retro-transposable hotspot of ovine genome underlies the alteration of BMP2 expression and development of fat tails. BMC Genom. 20, 1–15 (2019).
Zagožen, F. Ovčereja (ČZP Kmečki glas, 1982).
Cividini, A. et al. Rejski program za oplemenjeno jezersko-solčavsko ovco. (2024).
Savšek, R. et al. Poročilo o opravljenem delu znotraj STRP na področju reje drobnice v letu 2023. (2024).
Eck, K., Kunz, E., Mendel, C., Lühken, G. & Medugorac, I. Morphometric measurements in lambs as a basis for future mapping studies. Small Ruminant Res. 181, 57–64 (2019).
R Core Team. R: A language and environment for statistical computing, (2023). https://www.R-project.org/
Fox, J. & Weisberg, S. An R companion to applied regression (Sage, 2018).
Lenth, R. Estimated Marginal Means, aka Least-Squares Means_. R package version 1.8. 5 (2023).
Burnham, K. P. & Anderson, D. R. Model selection and multimodel inference: a practical information-theoretic approach (Springer, 2002).
Lynch, M. & Walsh, B. Genetics and analysis of quantitative traits Vol. 1 (Sinauer Sunderland, 1998).
Hümmelchen, H. et al. Frequency and characterisation of anomalies and fractures of the caudal spine in sheep with undocked tails. Animals 13, 1419 (2023).
Andrews, S. FastQC: A quality control tool for high throughput sequence data, (2010). https://www.bioinformatics.babraham.ac.uk/projects/fastqc/
Bonfert, T., Kirner, E., Csaba, G., Zimmer, R. & Friedel, C. C. ContextMap 2: fast and accurate context-based RNA-seq mapping. BMC Bioinform. 16, 1–15 (2015).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Kluge, M. & Friedel, C. C. Watchdog–a workflow management system for the distributed analysis of large-scale experimental data. BMC Bioinform. 19, 1–13 (2018).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 1–21 (2014).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. Roy. Stat. Soc.: Ser. B (Methodol.). 57, 289–300 (1995).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics: J. Integr. biology. 16, 284–287 (2012).
Murtagh, F. & Legendre, P. Ward’s hierarchical agglomerative clustering method: which algorithms implement Ward’s criterion? J. Classif. 31, 274–295 (2014).
Ahbara, A. et al. Genome-wide variation, candidate regions and genes associated with fat deposition and tail morphology in Ethiopian indigenous sheep. Front. Genet. 9, 699 (2019).
Burke, A. C., Nelson, C. E., Morgan, B. A. & Tabin, C. Hox genes and the evolution of vertebrate axial morphology. Development 121, 333–346 (1995).
Economides, K. D., Zeltser, L. & Capecchi, M. R. Hoxb13 mutations cause overgrowth of caudal spinal cordand tail vertebrae. Dev. Biol. 256, 317–330 (2003).
Denans, N., Iimura, T. & Pourquié, O. Hox genes control vertebrate body elongation by collinear Wnt repression. Elife 4, e04379 (2015).
Yamada, K. et al. An atlas of seven zebrafish hox cluster mutants provides insights into sub/neofunctionalization of vertebrate Hox clusters. Development 148, dev198325 (2021).
Deschamps, J. & van Nes, J. Developmental regulation of the Hox genes during axial morphogenesis in the mouse. (2005).
Porter, V., Alderson, L., Hall, S. J. G. & Sponenberg, D. P. Mason’s World Encyclopedia of Livestock Breeds and Breeding, CABI, 2, (2016).
Gilbert, S. F. & Barresi, M. J. F. Developmental Biology. 12th Edition ednOxford University Press, (2024).
Sadler, T. W. Langman’s Medical Embryology. 14th Edition ednWolters Kluwer, (2019).
Wellik, D. M. & Capecchi, M. R. Hox10 and Hox11 genes are required to globally pattern the mammalian skeleton. Science 301, 363–367 (2003).
Duboule, D. The rise and fall of Hox gene clusters. (2007).
Riddle, R. D., Johnson, R. L., Laufer, E. & Tabin, C. Sonic hedgehog mediates the polarizing activity of the ZPA. Cell 75, 1401–1416 (1993).
Kawakami, Y. et al. Sp8 and Sp9, two closely related buttonhead-like transcription factors, regulate Fgf8 expression and limb outgrowth in vertebrate embryos. (2004).
Galli, L. M. et al. Differential inhibition of Wnt-3a by Sfrp‐1, Sfrp‐2, and Sfrp‐3. Dev. dynamics: official publication Am. Association Anatomists. 235, 681–690 (2006).
Arteaga-Solis, E. et al. Regulation of limb patterning by extracellular microfibrils. J. Cell Biol. 154, 275–282 (2001).
Barna, M., Hawe, N., Niswander, L. & Pandolfi, P. P. Plzf regulates limb and axial skeletal patterning. Nat. Genet. 25, 166–172 (2000).
Bahnamiri, H. Z., Zali, A., Ganjkhanlou, M., Sadeghi, M. & Shahrbabak, H. M. Regulation of lipid metabolism in adipose depots of fat-tailed and thin-tailed lambs during negative and positive energy balances. Gene 641, 203–211 (2018).
Wild, J. P. Ryder: Sheep and man(Book Review). Antiquity 58, 142 (1984).
Verdugo, M. P. et al. Ancient cattle genomics, origins, and rapid turnover in the Fertile Crescent. Science 365, 173–176 (2019).
Hajihosseinlo, A., Jafari, S. & Ajdary, M. The relationship of GH and LEP gene polymorphisms with fat-tail measurements (fat-tail dimensions) in fat-tailed Makooei breed of Iranian sheep. Adv. Biomedical Res. 4, 172 (2015).
Moioli, B., Pilla, F. & Ciani, E. Signatures of selection identify loci associated with fat tail in sheep. J. Anim. Sci. 93, 4660–4669 (2015).
Yuan, Z. et al. Selection signature analysis reveals genes associated with tail type in Chinese indigenous sheep. Anim. Genet. 48, 55–66 (2017).
Bakhtiarizadeh, M. R., Salehi, A., Alamouti, A. A., Abdollahi-Arpanahi, R. & Salami, S. A. Deep transcriptome analysis using RNA-Seq suggests novel insights into molecular aspects of fat-tail metabolism in sheep. Sci. Rep. 9, 9203 (2019).
Acknowledgements
At the University of Ljubljana, Biotechnical Faculty, we thank Ana Petek, Ana Jakopič, Ana Brunčič, Stane Karner, Domen Drašler, Marko Bizjak, Gregor Šen, and Dušan Birtič for their valuable technical assistance with animal measurements and sampling procedures at the slaughterhouse. Funding: core grant P4-0220 from Slovenian Research and Innovation Agency (ARIS). We are also thankful to the DAAD funding programs for supporting our work through the project number DAAD ID-57514909.
Author information
Authors and Affiliations
Contributions
S.H.: original draft preparation. S.H., I.M., M.S., C.F.: conceptualization, methodology, supervision. R.E., M.Č., U.D., S.K., N.P., M.U. V.B., M.F.: investigation, methodology. All authors read, contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Horvat, S., Ellenrieder, R., Simčič, M. et al. Retention of embryonic positional identity signatures in the adult sheep tail: evidence from HOXB13 spatial RNA expression gradients. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42438-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42438-7