Abstract
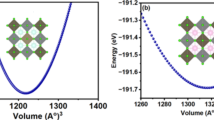
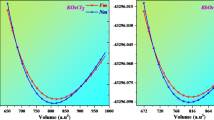
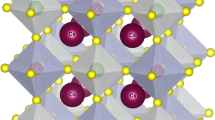
In this work, we use density functional theory (DFT) to thoroughly analyze the structural, electrical, optical, and thermoelectric characteristics of halide double perovskites Rb2AsAuX4 (X = Br, Cl). Both compounds’ structural stability is confirmed by the optimized lattice characteristics, with Rb2AsAuBr4 showing somewhat larger cell dimensions than Rb2AsAuCl4. Indirect band gaps of 0.338 eV (Br) and 0.885 eV (Cl), which are within the optimal range for solar applications, are shown by electronic band structure simulations. Strong absorption coefficients in the visible region above 1.2 × 101 cm−1 are shown in optical spectra, suggesting great potential for solar energy harvesting. High Seebeck coefficients of up to 310 μV/K (Br) and 285 μV/K (Cl) at ambient temperature are shown by thermoelectric analysis, together with electrical conductivities that facilitate effective charge transfer. Thermoelectric performance is further improved by the comparatively low thermal conductivity (0.9–1.1 W/m·K). Together, our findings demonstrate Rb2AsAuX4’s versatility and establish Rb2AsAuBr6 and Rb2AsAuCl4 as viable options for next optoelectronic and energy-harvesting applications.
Similar content being viewed by others
Data availability
Data underlying the results presented in this paper are not publicly available at this time but may be obtained from the corresponding author (fatmimessaud@yahoo.fr) upon reasonable request.
References
King, G. & Woodward, P. M. Cation ordering in perovskites. J. Mater. Chem. 20(28), 5785–5796. https://doi.org/10.1039/B926757C (2010).
Zhao, X. G. et al. Design of lead-free inorganic halide perovskites for solar cells via cation-transmutation. J. Am. Chem. Soc. 139(7), 2630–2638. https://doi.org/10.1021/jacs.6b09645 (2017).
Volonakis, G. et al. Lead-free halide double perovskites via heterovalent substitution of noble metals. J. Phys. Chem. Lett. 7(7), 1254–1259. https://doi.org/10.1021/acs.jpclett.6b00376 (2016).
Slavney, A. H., Hu, T., Lindenberg, A. M. & Karunadasa, H. I. A bismuth-halide double perovskite with long carrier recombination lifetime for photovoltaic applications. J. Am. Chem. Soc. 138(7), 2138–2141. https://doi.org/10.1021/jacs.5b13294 (2016).
Filip, M. R., Hillman, S., Haghighirad, A. A., Snaith, H. J. & Giustino, F. Band gaps of the lead-free halide double perovskites Cs2BiAgCl6 and Cs2BiAgBr6 from theory and experiment. J. Phys. Chem. Lett. 7(13), 2579–2585. https://doi.org/10.1021/acs.jpclett.6b01041 (2016).
Meng, W. et al. Parity-forbidden transitions and their impact on the optical absorption properties of lead-free metal halide perovskites and double perovskites. J. Phys. Chem. Lett. 7(23), 4848–4852. https://doi.org/10.1021/acs.jpclett.7b01042 (2016).
Wei, F. et al. The synthesis, structure and electronic properties of a lead-free hybrid inorganic–organic double perovskite (MA) 2 KBiCl 6 (MA= methylammonium). Mater. Horizons 4(6), 1091–1094. https://doi.org/10.1039/C6MH00053C (2017).
Tran, T. T., Panella, J. R., Chamorro, J. R., Morey, J. R. & Queen, T. M. Designing indirect–direct bandgap transitions in double perovskites. Mater. Horizons 4(4), 688–693. https://doi.org/10.1039/C7MH00239D (2017).
Xiao, Z. et al. Intrinsic instability of Cs2In (I) M (III) X6 (M= Bi, Sb; X= halogen) double perovskites: A combined density functional theory and experimental study. J. Am. Chem. Soc. 139(17), 6054–6057. https://doi.org/10.1021/jacs.7b02227 (2017).
Zhou, J. et al. Composition design, optical gap and stability investigations of lead-free halide double perovskite Cs2AgInCl6. J. Mater. Chem. A 5(29), 15031–15037. https://doi.org/10.1039/C7TA04690A (2017).
Zhao, S., Yamamoto, K., Iikubo, S., Hayashi, S. & Ma, T. First-principles study of electronic and optical properties of lead-free double perovskites Cs2NaBX6 (B= Sb, Bi; X= Cl, Br, I). J. Phys. Chem. Solids 117, 117–121. https://doi.org/10.1016/j.jpcs.2018.02.032 (2018).
Du, K. Z., Meng, W., Wang, X., Yan, Y. & Mitzi, D. B. Bandgap engineering of lead-free double perovskite Cs2AgBiBr6 through trivalent metal alloying. Angew. Chem. 129(28), 8270–8274. https://doi.org/10.1002/ange.201703970 (2017).
Greul, E., Petrus, M. L., Binek, A., Docampo, P. & Bein, T. Highly stable, phase pure Cs2AgBiBr6 double perovskite thin films for optoelectronic applications. J. Mater. Chem. A 5(37), 19972–19981. https://doi.org/10.1039/C7TA06816F (2017).
Ning, W. et al. Long electron–hole diffusion length in high-quality lead-free double perovskite films. Adv. Mater. 30(7), 1706246. https://doi.org/10.1002/adma.201706246 (2018).
Yang, X. et al. Multifunctional dye interlayers: Simultaneous power conversion efficiency and stability enhancement of Cs2AgBiBr6 lead-free inorganic perovskite solar cell through adopting a multifunctional dye interlayer. Adv. Func. Mater. 30(16), 2001557. https://doi.org/10.1002/adfm.202001557 (2017).
Pantaler, M. et al. Hysteresis-free lead-free double-perovskite solar cells by interface engineering. ACS Energy Lett. 3(8), 1781–1786. https://doi.org/10.1021/acsenergylett.8b00871 (2018).
Deng, Z. et al. Exploring the properties of lead-free hybrid double perovskites using a combined computational-experimental approach. J. Mater. Chem. A 4(31), 12025–12029. https://doi.org/10.1039/C6TA05817E (2016).
Wu, C. et al. The dawn of lead‐free perovskite solar cell: Highly stable double perovskite Cs2AgBiBr6 film. Adv. Sci. 5(3), 1700759. https://doi.org/10.1002/advs.201700759 (2018).
Li, Q. et al. High‐pressure band‐gap engineering in lead‐free Cs2AgBiBr6 double perovskite. Angew. Chem. 129(50), 15969–15973. https://doi.org/10.1002/anie.201708684 (2017).
Savory, C. N., Walsh, A. & Scanlon, D. O. Can Pb-free halide double perovskites support high-efficiency solar cells?. ACS Energy Lett. 1(5), 949–955. https://doi.org/10.1021/acsenergylett.6b00471 (2016).
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131(17), 6050–6051. https://doi.org/10.1021/ja809598r (2009).
Zhao, X. G. et al. Design of lead-free inorganic halide perovskites for solar cells via cation-transmutation. J. Am. Chem. Soc. 139(7), 2630–2638. https://doi.org/10.1021/jacs.6b09645 (2017).
Chen, M., Ju, M.-G. & Carl, A. D. Cesium Titanium(IV) Bromide thin films based stable lead-free perovskite solar cells. Joule 2(3), 558–570. https://doi.org/10.1016/j.joule.2018.01.009 (2018).
Hu, S., Ren, Z., Djurišić, A. B. & Rogach, A. L. Metal halide perovskites as emerging thermoelectric materials. ACS Energy Lett. 6(11), 3882–3905. https://doi.org/10.1021/acsenergylett.1c02015 (2021).
Zhan, X. Q. et al. Can lead-free double halide perovskites serve as proper photovoltaic absorber?. J. Phys. Chem. Lett. 14(48), 10784–10793. https://doi.org/10.1021/acs.jpclett.3c02663 (2023).
Shi, Y. et al. Tunable molecular dipole moments and orientations for efficient and stable perovskite solar cells. Joule https://doi.org/10.1016/j.joule.2025.102009 (2025).
Aron Walsh, Alexey A. Sokol, John Buckeridge, David O. Scanlon & C. Richard A. Catlow, Oxidation states and ionicity. Nature Materials, 17, 958–964.https://doi.org/10.1038/s41563-018-0165-7 (2018).
Blaha, P., Schwarz, K., Madsen, G. K. H. & Kvasnicka, D. Wien2K. Augment. Plane Wave+ Local Orbitals Program Calc. Cryst. Prop. 60(1), 155–169 (2001).
Burke, K., Perdew, J. P. & Ernzerhof, M. Why semilocal functionals work: Accuracy of the on-top pair density and importance of system averaging. J. Chem. Phys. 109(10), 3760–3771. https://doi.org/10.1063/1.476976 (1998).
Becke, A. D. & Johnson, E. R. A simple effective potential for exchange. J. Chem. Phys. 124(22), 221101. https://doi.org/10.1063/1.2213970 (2006).
Slack, G. A. Nonmetallic crystals with high thermal conductivity. J. Phys. Chem. Solids 34(2), 321–335. https://doi.org/10.1016/0022-3697(73)90168-7 (1973).
Berman, R. & Klemens, P. G. Thermal conduction in solids. Phys. Today. 31(9), 56–57. https://doi.org/10.1063/1.2994996 (1978).
Julian, C. L. Theory of heat conduction in rare-gas crystals. Phys. Rev. 137(1A), A128–A137. https://doi.org/10.1103/PhysRev.137.A128 (1965).
Murnaghan, F. D. The compressibility of media under extreme pressures. Proc. Natl. Acad. Sci. U. S. A. 30(9), 244–247. https://doi.org/10.1073/pnas.30.9.244 (1944).
Hasan, M. M., Sarker, M. A., Islam, M. R. & Islam, M. R. First-principles analysis of the effects of halogen variation on the properties of lead-free novel perovskites AlGeX3 (X = F, Cl, Br, and I). ACS Omega 9(33), 35301–35312. https://doi.org/10.1021/acsomega.4c00209 (2024).
Zheng, F., Tan, L. Z., Liu, S. & Rappe, A. M. Rashba spin–orbit coupling enhanced carrier lifetime in CH3NH3PbI3. Nano Lett. 15(12), 7794–7800. https://doi.org/10.1021/acs.nanolett.5b01854 (2015).
Barman, N. & Sarkar, U. Theoretical insights into the structural, electronic, photocatalytic and supercapacitor applications of pentahexoctite. Phys. Chem. Chem. Phys. https://doi.org/10.1039/D5CP04410C (2025).
Simoncelli, M., Marzari, N. & Mauri, F. Unified theory of thermal transport in crystals and glasses. Nat. Phys. 15(8), 809–813. https://doi.org/10.1038/s41567-019-0520-x (2019).
Barman, N., Dua, H. & Sarkar, U. First principles investigation of thermoelectric properties of Naphyne. Appl. Surf. Sci. 682, 161649. https://doi.org/10.1016/j.apsusc.2024.161649 (2025).
Barman, N. & Sarkar, U. Pentagraphyne: A high-performance thermoelectric material with high Seebeck coefficient. ACS Appl. Energy Mater. https://doi.org/10.1021/acsaem.5c01477 (2025).
Muscarella, L. A. & Hutter, E. M. Halide double-perovskite semiconductors beyond photovoltaics. ACS Energy Lett. 7(6), 2128–2135. https://doi.org/10.1021/acsenergylett.2c00811 (2022).
Ju, M.-G. et al. Toward eco-friendly and stable perovskite materials for photovoltaics. Joule 2(7), 1231–1241. https://doi.org/10.1016/j.joule.2018.04.026 (2018).
Duan, L., Cui, X., Xu, C., Chen, Z. & Zheng, J. Monolithic perovskite/perovskite/silicon triple-junction solar cells: Fundamentals, progress, and prospects. Nano-Micro Lett. 18(1), 8. https://doi.org/10.1007/s40820-025-01836-8 (2025).
Monir, M. E. et al. Half-metallicity of novel halide double perovskites K2CuVCl6 and Rb2CuVCl6: Application in next-generation spintronic devices. RSC Adv. 15, 17685. https://doi.org/10.1039/D5RA02811F (2025).
Fatmi, M. et al. High performance double perovskites of Cs2InAgBr6 and Cs2InAgCl6 structural electronic optical and thermoelectric properties for next generation photovoltaics. Sci. Rep. 15, 20851. https://doi.org/10.1038/s41598-025-04600-5 (2025).
Ghebouli, M. A. et al. Exploring the mechanical, dynamical, and thermal stability of Cs2AgBiX6 (X = Br, Cl) for optoelectronic and thermoelectric applications. Sci. Rep. 15, 20993. https://doi.org/10.1038/s41598-025-05549-1 (2025).
Benlakhdar, F. et al. Electronic and optical properties of lead-free K2AgSbBr6 double perovskite tuned by doping elements Cu+, Bi3+, and I-. Sci. Rep. 15, 40362. https://doi.org/10.1038/s41598-025-24417-6 (2025).
Funding
No funding was provided.
Author information
Authors and Affiliations
Contributions
Conceptualization: K. Bouferrache, Mustafa Jaipallah Abdelmageed Abualreish. Data curation: M.A. Ghebouli, Aseel Smerat. Formal analysis: Murat Yaylacı, S. Alomairy, Ghadah Sheetah. Validation: M. Fatmi.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bouferrache, K., Ghebouli, M.A., Fatmi, M. et al. Synergistic optoelectronic and thermoelectric performance in Rb2AsAuBr6 and Rb2AsAuCl6 double perovskites for multifunctional energy conversion. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42440-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42440-z