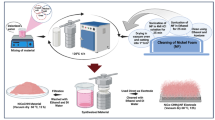
Abstract
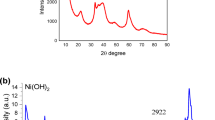
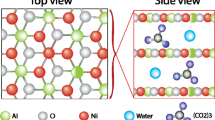
The free-standing Ni3S2 electrode was fabricated via a simple one-step hydrothermal method at 160 °C through the sulfidization of nickel foam (NF). The as-synthesized samples exhibited X-ray diffraction (XRD) patterns and Raman spectra consistent with the formation of Ni3S2 Although the XRD reflections of the Ni3S2 lattice remained unchanged after short-term electrochemical activation in 3.5 M KOH electrolyte, the Raman spectrum evolved to resemble that of oxidized nickel species, indicating a surface transformation of the electrode under electrochemical conditions. Prolonged redox cycling of the electrode for more than 10,000 cycles resulted in noticeable modifications of the XRD pattern. In addition to the characteristic reflections of the Ni3S2 phase, the formation of β-Ni(OH)2 nanoparticles was observed, with an average crystallite size of approximately 2.8 nm, as estimated using the Scherrer equation. The resulting heterostructured Ni(OH)2/Ni3S2/NF electrode exhibited an areal capacitance of 4.1 C cm–2 at a current density of 20 mA cm–2 when evaluated in a three-electrode configuration. The electrode demonstrated progressively enhanced capacitance stability with continued cycling. Capacitance retention increased from 19% after the first 10,000 cycles to 41% after the second 10,000 cycles, 66% after the third 8,000 cycles, and ultimately 79% after the fourth 6,000 cycles. Taking into account the electrochemical activation occurring between cycling intervals, the overall capacitance retention reached 72% after more than 30,000 cycles. Although the electrode capacitance decreased during galvanostatic charge–discharge (GCD) cycling, it could be partially recovered through subsequent cyclic voltammetry (CV) activation. Concurrently, a broad XRD reflection attributable to the α-Ni(OH)2 phase emerged, providing valuable insight into the structural evolution of the electrode and the underlying electrochemical energy-storage mechanism. The Ni(OH)2/Ni3S2/NF electrode aged through 10,000 GCD cycles was subsequently employed as the positive battery-type electrode in a hybrid supercapacitor, paired with an activated carbon negative electrode. The assembled device, with a Ni3S2/NF electrode area of 6 cm2, delivered an areal capacitance of 4.63 C cm–2 at a current density of 4 mA cm–2 and maintained 83.5% capacitance retention after more than 20,000 charge–discharge cycles.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
Sidhanath, V., Bhosale, S. V. & Bhosale Advancements in supercapacitors: breaking barriers and enabling amazing applications. Chem. Sci. 16, 10159. https://doi.org/10.1039/d5sc01955a (2025).
Kavishka & Dissanayake Dulsha Kularatna-Abeywardana. A review of supercapacitors: Materials, technology, challenges, and renewable energy applications. J. Energy Storage. 96, 112563. https://doi.org/10.1016/j.est.2024.112563 (2024).
Jayaprakash Meena, S., Sivasubramaniam, E. & David, Santhakumar, K. Green supercapacitors: review and perspectives on sustainable template-free synthesis of metal and metal oxide nanoparticles. RSC Sustain. 2, 1224–1245. https://doi.org/10.1039/d4su00009a (2024).
Iqbal Singh, R. et al. Ajay Vasishth. A review on supercapacitors: Development trends, challenges, and future perspectives. Next Res. 2, 100228. https://doi.org/10.1016/j.nexres.2025.100228 (2025).
Singh, N. et al. A comprehensive review on supercapacitors: Basics to recent advancements. J. Energy Storage. 121, 116498. https://doi.org/10.1016/j.est.2025.116498 (2025).
Mojtaba Mirzaeian, Q. et al. Hassan Fathinejad Jirandehi. Electrode and electrolyte materials for electrochemical capacitors. Int. J. Hydrog. Energy. 42, 25565–25587. https://doi.org/10.1016/j.ijhydene.2017.04.241 (2017).
Iqbal, M. F. et al. Faizan Ali. Supercapacitors: An Emerging Energy Storage System. Adv. Energy Sustain. Res. 6, 2400412. https://doi.org/10.1002/aesr.202400412 (2025).
Ander González, E., Goikolea, J. A., Barrena, R. & Mysyk Review on supercapacitors: Technologies and materials. Renew. Sustain. Energy Rev. 58, 1189–1206. https://doi.org/10.1016/j.rser.2015.12.249 (2016).
Liang, R. et al. Transition Metal Oxide Electrode Materials for Supercapacitors: A Review of Recent Developments. Nanomaterials 11, 1248. https://doi.org/10.3390/nano11051248 (2021).
Vediyappan Thirumal, N., Mahato, K., Yoo, J. & Kim High performance Li-ion battery-type hybrid supercapacitor devices using antimony based composite anode and Ketjen black carbon cathode. J. Energy Storage. 61, 106756. https://doi.org/10.1016/j.est.2023.106756 (2023).
Su, D. Y. et al. Conducting Polymers in Supercapacitor Application. IOP Conf. Series: Earth Environ. Sci. 267, 042048. https://doi.org/10.1088/1755-1315/267/4/042048 (2019).
Subalakshmi Kumar, A. K. et al. Effect of fluoride (CoF2) based electrode material for high energy and power density asymmetric flexible supercapacitors. J. Energy Storage. 87, 111460. https://doi.org/10.1016/j.est.2024.111460 (2024).
Adil Emin, A. et al. An efficient electrodeposition approach for preparing CoMn-hydroxide on nickel foam as high-performance electrodes in aqueous hybrid supercapacitors. Fuel 381 Part. B. 133335. https://doi.org/10.1016/j.fuel.2024.133335 (2025).
Pranav, K., Katkar, M. B., Naikwade, S. W. & Lee A synergistic electrochemical properties of 3D marigold-like Fe-Cu-Ag phosphate/MWCNTs nanocomposite towards hybrid solid-state supercapatteries. J. Alloys Compd. 1038, 182815. https://doi.org/10.1016/j.jallcom.2025.182815 (2025).
Sakshi Sharma, P., Kadyan, R. K., Sharma, N., Kumar, S. & Grover Progressive updates on nickel hydroxide and its nanocomposite for electrochemical electrode material in asymmetric supercapacitor device. J. Energy Storage. 87, 111368. https://doi.org/10.1016/j.est.2024.111368 (2024).
Sajjad Gharanli, A. et al. Environmentally friendly nickel-based nanocomposites for energy storage: A review of supercapacitor and battery-type mechanisms. J. Energy Storage. 122, 116509. https://doi.org/10.1016/j.est.2025.116509 (2025).
Zhang, L., Shi, D., Liu, T., Jaroniec, M. & Yu, J. Nickel-based materials for supercapacitors. Mater. Today. 25, 35–65. https://doi.org/10.1016/j.mattod.2018.11.002 (2018).
Zhihan Liu, K. & Liu, X. Fabrication of customizable specific capacitance supercapacitor electrodes via in-situ oxidation of nickel foam by cyclic voltammetry process. J. Power Sources. 641, 236833. https://doi.org/10.1016/j.jpowsour.2025.236833 (2025).
Mengkang Zhu, D. et al. Shuyong Jiad Zhidong Chen. Recent advances in transition metal sulfide-based electrode materials for supercapacitors. Chem. Commun. 61, 8314–8326. https://doi.org/10.1039/D5CC01411E (2025).
Xuerui Yi, C., Kirk & Robertson, N. Nickel hydroxide-based energy storage devices nickel-metal hydride batteries vs. nickel hydroxide hybrid supercapacitors. Carbon Neutrality. 3, 39. https://doi.org/10.1007/s43979-024-00114-7 (2024).
Guan, Y. et al. Review of NiS-Based Electrode Nanomaterials for Supercapacitors. Nanomaterials 13, 979. https://doi.org/10.3390/nano13060979 (2023).
Ramyakrishna Pothu, R. et al. Jian-Min Ma. Nickel sulfide-based energy storage materials for high-performance electrochemical capacitors. Rare Met. 40, 353–373. https://doi.org/10.1007/s12598-020-01470-w (2021).
Asghar, A. et al. Recent progress in metal oxide-based electrode materials for safe and sustainable variants of supercapacitors. Front. Chem. 12, 1402563. https://doi.org/10.3389/fchem.2024.1402563 (2024).
Yan, J., Lu, J., Sheng, Y., Sun, Y. & Zhang, D. Research Progress in the Preparation of Transition Metal Sulfide Materials and Their Supercapacitor Performance. Micromachines 15, 849. https://doi.org/10.3390/mi15070849 (2024).
Thomas, S. A., Cherusseri, J. & Deepthi, N. Rajendran. Nickel Disulfide (NiS2): A Sustainable Low-Cost Electrode Material for High-Performance Supercapacitors. Energy Technol. 12, 2400138. https://doi.org/10.1002/ente.202400138 (2025).
Li, Y., Shi, M., Wang, L. W. M., Li, J. & Cui, H. Tailoring synthesis of Ni3S2 nanosheets with high electrochemical performance by electrodeposition. Adv. Powder Technol. 29, 1092–1098. https://doi.org/10.1016/j.apt.2018.01.026 (2018).
Huang, H. et al. One-Step Synthesis of Self-Supported Ni3S2/NiS Composite Film on Ni Foam by Electrodeposition for High-Performance Supercapacitors. Nanomaterials 9, 1718. https://doi.org/10.3390/nano9121718 (2019).
Zhu, J. et al. Nanostructured Fe-Doped Ni3S2 Electrocatalyst for the Oxygen Evolution Reaction with High Stability at an Industrially-Relevant Current Density. ACS Appl. Mater. Interfaces. 16, 58520–58535. https://doi.org/10.1021/acsami.4c09821 (2024).
Lin, T. W., Dai, C. S. & Hung, K. C. High Energy Density Asymmetric Supercapacitor Based on NiOOH/Ni3S2/3D Graphene and Fe3O4/Graphene Composite Electrodes. Sci. Rep. 4, 7274. https://doi.org/10.1038/srep07274 (2014).
Xuerui Yi, C., Kirk & Robertson, N. Achieving Complete Conversion from Nickel Foam to Nickel Sulfide Foam for a Freestanding Hybrid-Supercapacitor Electrode. ChemElectroChem. 11, e202400383. (2024). https://doi.org/10.1002/celc.202400383
Tang, J. et al. High-rate and ultra-stable Na-ion storage for Ni3S2 nanoarrays via self-adaptive pseudocapacitance. Electrochim. Acta. 265, 709–716. https://doi.org/10.1016/j.electacta.2018.01.199 (2018).
Arun, T., Aravinth, K., Balaji Bhargav, P. & Francis, M. K. Ni-foam modified binder-free Ni3S2 electrode for improved cyclic performance in asymmetric supercapacitors. J. Phys. Chem. Solids. 184, 111723. https://doi.org/10.1016/j.jpcs.2023.111723 (2024).
Wu, B. et al. Ni3S2 nanorods growing directly on Ni foam for all-solid-state asymmetric supercapacitor and efficient overall water splitting. J. Energy Chem. 46, 178–186. https://doi.org/10.1016/j.jechem.2019.11.011 (2020).
Xunhui Xiong, B. et al. One-step synthesis of architectural Ni3S2 nanosheet-on-nanorods array for use as high-performance electrodes for supercapacitors. NPG Asia Mater. 8, e300. https://doi.org/10.1038/am.2016.126 (2016).
Huanhuan Huo, Y. & Zhao, C. Xu. 3D Ni3S2 nanosheet arrays supported on Ni foam for high-performance supercapacitor and nonenzymatic glucose detection. J. Mater. Chem. A. 2, 15111. (2014). https://doi.org/10.1039/c4ta02857k
Wensha Niu, Z. et al. High-performance asymmetric supercapacitor based on Ni3S2 nanoparticles immobilized on carbon nanosheets from sodium alginate. J. Alloys Compd. 885, 161194. https://doi.org/10.1016/j.jallcom.2021.161194 (2021).
Chen, X. et al. Hydrogen peroxide-induced growth of hierarchical Ni3S2 nanorod/sheet arrays for high performance supercapacitors. J. Colloid Interface Sci. 575, 168–176. https://doi.org/10.1016/j.jcis.2020.04.098 (2020).
Zhang, H., Liu, Y., Zhu, C. & Ma, X. Influence of annealing process on the electrochemical properties of Ni3S2 electrode for stable supercapacitors. J. Energy Storage. 32, 101946. https://doi.org/10.1016/j.est.2020.101946 (2020).
Shao, M., Li, J., Li, J., Yan, Y. & Li, R. Synthesis of Ni3S2 and MOF–Derived Ni(OH)2 Composite Electrode Materials on Ni Foam for High–Performance Supercapacitors. Nanomaterials 13, 493. https://doi.org/10.3390/nano13030493 (2023).
Appiagyei, A. B. Bonsu, J. O. & Han, J. I. Construction of NiCo-OH/Ni3S2 core-shell heterostructure wrapped in rGO nanosheets as efficient supercapacitor electrode enabling high stability up to 20,000 cycles. J. Electroanal. Chemistry 889, 115226. (2021). https://doi.org/10.1016/j.jelechem.2021.115226
Kimothi, S., Ahmad, W., Gautam, A. S. & Singh, S. P. Roopesh Kumar and Mohd. Yusuf. High-performance Electrode Material Ni(OH)2 for Supercapacitors in View of Energy Storage Applications. Curr. Mater. Sci. 18, 274–280. https://doi.org/10.2174/0126661454274174231228101732 (2025).
Patel, K. K., Singhal, T., Pandey, V., Sumangala, T. P. & Sreekanth, M. S. Evolution and recent developments of high performance electrode material for supercapacitors: A review. J. Energy Storage. 44, 103366. https://doi.org/10.1016/j.est.2021.103366 (2021).
Ash, B., Nalajala, V. S., Popuri, A. K., Subbaiah, T. & Minakshi, M. Perspectives on Nickel Hydroxide Electrodes Suitable for Rechargeable Batteries: Electrolytic vs. Chem. Synthesis Routes Nanomaterials. 10, 1878. https://doi.org/10.3390/nano10091878 (2020).
Li, J., Liu, Y., Cao, W. & Chen, N. Rapid in situ growth of β-Ni(OH)2 nanosheet arrays on nickel foam as an integrated electrode for supercapacitors exhibiting high energy density. Dalton Trans. 49, 4956–4966. https://doi.org/10.1039/d0dt00687d (2020).
Liang Zhao, S. et al. Phase-controlled growth of nickel hydroxide nanostructures on nickel foam for enhanced supercapacitor performance. J. Energy Storage. 43, 103171. https://doi.org/10.1016/j.est.2021.103171 (2021).
Chen, J. S., Guan, C. & Gui, Y. Daniel John Blackwood. Rational Design of Self-Supported Ni3S2 Nanosheets Array for Advanced Asymmetric Supercapacitor with a Superior Energy Density. ACS Appl. Mater. Interfaces. 9, 496–504. https://doi.org/10.1021/acsami.6b14746 (2017).
Weijia Zhou, X. et al. One-step synthesis of Ni3S2 nanorod@Ni(OH)2 nanosheet core–shell nanostructures on a threedimensional graphene network for high-performance supercapacitors. Energy Environ. Sci. 6, 2216. https://doi.org/10.1039/c3ee40155c (2013).
Xiaobing, W., Hao, J. H. Y. S. J., Liu, F., Han, S. & An, J. Ni Foam-Ni3S2@Ni(OH)2-Graphene Sandwich Structure Electrode Materials: Facile Synthesis and High Supercapacitor Performance. Chem. Eur. J. 23, 4128–4136. https://doi.org/10.1002/chem.201605212 (2017).
Cao, Y. Q. et al. Self-formed porous Ni(OH)2 on Ni3S2/Ni foam during electrochemical cycling for high performance supercapacitor with ultrahigh areal capacitance. Electrochim. Acta. 303, 148–156. https://doi.org/10.1016/j.electacta.2019.02.075 (2019).
Sha Liu, K. et al. Three-dimensional interconnected composite nanoarchitectonics with Ni(OH)2/Ni3S2 nanosheets and hierarchical porous carbon for high-performance supercapacitor electrodes. Diam. Relat. Mater. 155, 112326. https://doi.org/10.1016/j.diamond.2025.112326 (2025).
Jinghuang Lin, X. et al. Weidong Fei and Jicai Feng. Rational construction of core–shell Ni3S2@Ni(OH)2 nanostructures as battery-like electrodes for supercapacitors. Inorg. Chem. Front. 5, 1985–1991. https://doi.org/10.1039/c8qi00347e (2018).
Wang, J. H., Cheng, Z. & Brédas, J. L. Electronic and vibrational properties of nickel sulfides from first principles. J. Chem. Phys. 127, 214705. https://doi.org/10.1063/1.2801985 (2007).
Adil Emin, Q., Li, J., Li, Y., Li, D. & He Rational design of hedgehog-like NiMn2S4 architectures for energy storage devices. Int. J. Electrochem. Sci. 19, 100881. https://doi.org/10.1016/j.ijoes.2024.100881 (2024).
Nesbitt, H., Legrand, D. & Bancroft, G. Interpretation of Ni2p XPS spectra of Ni conductors and Ni insulators. Phys. Chem. Min. 27, 357–366. https://doi.org/10.1007/s002690050265 (2000).
Buckley, A. N. & Woods, R. Electrochemical and XPS studies of the surface oxidation of synthetic heazlewoodite (Ni3S2). J. Appl. Electrochem. 21, 575–582. https://doi.org/10.1007/BF01024844 (1991).
Bing, Y. et al. Surface Restructuring of Nickel Sulfide Generates Optimally Coordinated Active Sites for Oxygen Reduction Catalysis. Joule 1, 600–612. (2017). https://doi.org/10.1016/j.joule.2017.08.020
Julia Gallenberger, H. M. et al. Stability and decomposition pathways of the NiOOH OER active phase of NiOx electrocatalysts at open circuit potential traced by ex situ and in situ spectroscopies. Catal. Sci. Technol. 13, 4693–4700. https://doi.org/10.1039/d3cy00674c (2023).
Kostecki, R. Frank McLarnon. Electrochemical and In Situ Raman Spectroscopic Characterization of Nickel Hydroxide Electrodes: I. Pure Nickel Hydroxide. J. Electrochem. Soc. 144, 485. https://doi.org/10.1149/1.1837437 (1997).
Hall, D. S., Lockwood, D. J., Bock, C. & MacDougall, B. R. Nickel hydroxides and related materials: a review of their structures, synthesis and properties. Proc. R. Soc. A 471, 20140792. (2015). https://doi.org/10.1098/rspa.2014.0792
Lee, M., Jang, Y., Yoon, G. & Lee, S. Gyeong Hee Ryu. Synthesis and electrochemical evaluation of nickel hydroxide nanosheets with phase transition to nickel oxide. RSC Adv. 14, 10172. https://doi.org/10.1039/d4ra01120a (2024).
Ferrus-Suspedra, O. D. M. D., Marc, T. M. & Koper The importance of nickel oxyhydroxide deprotonation on its activity towards electrochemical water oxidation. Chem. Sci. 7, 2639. https://doi.org/10.1039/c5sc04486c (2016).
Acknowledgements
The study was carried out with the financial support of the Ministry of Science and Higher Education of the Republic of Kazakhstan (grant No AP23488976).
Funding
The study was carried out with the financial support of the Ministry of Science and Higher Education of the Republic of Kazakhstan (grant No AP23488976).
Author information
Authors and Affiliations
Contributions
Conceptualization, K.A.A., M.T.G., D.Z. and M.M.; methodology, K.A.A., Z.K.K., L.V.G., and A.A.M.; software, D.Z. and M.T.G.; validation, L.V.G., Z.K.K., A.A.M., R.R.N. and D.Z.; formal analysis, Z.K.K., L.V.G. A.A.M., Z.S.K. and R.R.N.; investigation, K.A.A., R.R.N., Z.S.K. and D.Z.; resources, K.A.A. and M.T.G.; data curation, Z.K.K., L.V.G., A.A.M., Z.S.K. and D.Z.; writing—original draft preparation, K.A.A., D.Z., Z.S.K. and M.M.; writing—review and editing, K.A.A.; supervision, K.A.A.; project administration, K.A.A. and M.T.G.; funding acquisition, K.A.A. and M.T.G. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdullin, K.A., Gabdullin, M.T., Gritsenko, L.V. et al. In situ formation of nanocrystalline Ni(OH)2 in alkaline electrolyte explains superior capacitance and cycling stability of Ni3S2/NF electrodes. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42576-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42576-y