Abstract
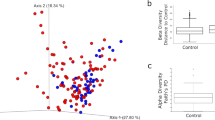
The microbiome-gut-brain axis has been increasingly recognized for its role in the pathophysiology of autism spectrum disorder (ASD), yet the underlying molecular mechanisms remain poorly understood. Neuropeptide Y (NPY), a key modulator of gut-brain communication, may play a pivotal role in this axis. This study investigated the sex-specific molecular profile of the NPY system in gut-brain communication via a genetic mouse model of ASD, the Nf1+/- mice. Quantitative real-time PCR was performed to assess the expression of NPY and its receptor transcripts in the amygdala, hippocampus, prefrontal cortex and intestinal tissue of juvenile male and female Nf1+/- mice. Additionally, gut microbiota analysis focused on Lactobacillus species in stool samples. Special emphasis was placed on sex differences, an area underexplored in ASD research. Sex-specific differences in NPY and its receptor expression were observed in both the brain and intestinal tissues of Nf1+/- mice. In mutant females, estrous cycle fluctuations were partly associated with changes in the NPY system. Notably, distinct correlations between the brain and intestinal NPY systems were identified in both sexes of wild-type (WT) and Nf1+/- mice. Microbiota analysis revealed sex-dependent alterations in Lactobacillus abundance, which correlated with the intestinal NPY system. Importantly, the Y2 receptor exhibited sex-specific expression patterns in both the gut and brain of Nf1+/- mice. This study provides novel evidence that the NPY system may play a critical role in gut-brain communication in ASD, with sex-dependent alterations in both the brain and gut. The intestinal Y2 receptor has emerged as a potential molecular biomarker for ASD, underscoring the importance of incorporating sex as a biological variable in future ASD research.
Similar content being viewed by others
Data availability
The datasets supporting the conclusions of this article are included within the article and its additional files.
Abbreviations
- ASD:
-
Autism spectrum disorder
- mRNA:
-
Messenger ribonucleic acid
- NF1:
-
Neurofibromatosis type 1
- NPY:
-
Neuropeptide Y
- PCA:
-
Principal component analysis
- PCR:
-
Polymerase chain reaction
- WT:
-
Wild-type
References
Zeidan, J. et al. Global prevalence of autism: A systematic review update. Autism Res. 15, 778–790 (2022).
Hartley, S. L. & Sikora, D. M. Sex differences in Autism Spectrum Disorder: An examination of developmental functioning, autistic symptoms, and coexisting behavior problems in toddlers. J. Autism Dev. Disord. 39, 1715–1722 (2009).
Matson, J. L. & Jang, J. Treating aggression in persons with Autism Spectrum Disorders: A review. Res. Dev. Disabil. 35, 3386–3391 (2014).
May, T., Cornish, K. & Rinehart, N. Does gender matter? A one year follow-up of autistic, attention and anxiety symptoms in high-functioning children with Autism Spectrum Disorder. J. Autism Dev. Disord. 44, 1077–1086 (2014).
May, T., Cornish, K. & Rinehart, N. J. Gender profiles of behavioral attention in children with Autism Spectrum Disorder. J. Atten. Disord. 20, 627–635 (2016).
Silva, G. et al. Oscillatory motor patterning is impaired in Neurofibromatosis Type 1: A behavioural, EEG and fMRI study. J. Neurodev. Disord. 10, 11 (2018).
Silva, G. et al. Peripheral attentional targets under covert attention lead to paradoxically enhanced alpha desynchronization in Neurofibromatosis Type 1. PLoS ONE 11, e0148600 (2016).
Haebich, K. M. et al. Socially oriented attention in young children with neurofibromatosis type 1: An eye-tracking study. Dev. Med. Child Neurol. https://doi.org/10.1111/dmcn.16497 (2025).
Santos, S. et al. Neurobehavioral sex-related differences in Nf1+/− mice: female show a “camouflaging”-type behavior. Biol. Sex Differ. 14, 24 (2023).
Ferreira, H., Santos, S., Martins, J., Castelo-Branco, M. & Gonçalves, J. Distinct early development trajectories in Nf1(±) and Tsc2(±) mouse models of autism. J. Neurodev. Disord. 17, 42 (2025).
Gonçalves, J. et al. Testing the excitation/inhibition imbalance hypothesis in a mouse model of the Autism Spectrum Disorder: In vivo neurospectroscopy and molecular evidence for regional phenotypes. Mol. Autism 8, 47 (2017).
Vellingiri, B. et al. An anxious relationship between Autism Spectrum Disorder and gut microbiota: A tangled chemistry?. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas. 99, 169–189 (2022).
Leader, G. et al. Gastrointestinal symptoms in Autism Spectrum Disorder: A systematic review. Nutrients https://doi.org/10.3390/nu14071471 (2022).
Iglesias-Vázquez, L., Van Ginkel Riba, G., Arija, V. & Canals, J. Composition of gut microbiota in children with Autism Spectrum Disorder: A systematic review and meta-analysis. Nutrients https://doi.org/10.3390/nu12030792 (2020).
Sauer, A. K., Bockmann, J., Steinestel, K., Boeckers, T. M. & Grabrucker, A. M. Altered intestinal morphology and microbiota composition in the Autism Spectrum Disorders associated SHANK3 mouse model. Int. J. Mol. Sci. https://doi.org/10.3390/ijms20092134 (2019).
Lee, C. Y. Q., Franks, A. E. & Hill-Yardin, E. L. Autism-associated synaptic mutations impact the gut-brain axis in mice. Brain. Behav. Immun. 88, 275–282 (2020).
Tabouy, L. et al. Dysbiosis of microbiome and probiotic treatment in a genetic model of Autism Spectrum Disorders. Brain. Behav. Immun. 73, 310–319 (2018).
Buffington, S. A. et al. Microbial reconstitution reverses maternal diet-induced social and synaptic deficits in offspring. Cell 165, 1762–1775 (2016).
Dicks, L. M. T. Gut bacteria and neurotransmitters. Microorganisms 10, 1838 (2022).
Bettag, J. et al. Gut Microbiota to Microglia: Microbiome Influences Neurodevelopment in the CNS. Children (Basel Switzerland) 10, 1767 (2023).
Wei, P., Keller, C. & Li, L. Neuropeptides in gut-brain axis and their influence on host immunity and stress. Comput. Struct. Biotechnol. J. 18, 843–851 (2020).
Holzer, P., Reichmann, F. & Farzi, A. Neuropeptide Y, peptide YY and pancreatic polypeptide in the gut-brain axis. Neuropeptides 46, 261–274 (2012).
Sajdyk, T. J., Schober, D. A., Smiley, D. L. & Gehlert, D. R. Neuropeptide Y-Y2 receptors mediate anxiety in the amygdala. Pharmacol. Biochem. Behav. 71, 419–423 (2002).
Tasan, R. O. et al. The central and basolateral amygdala are critical sites of neuropeptide Y/Y2 receptor-mediated regulation of anxiety and depression. J. Neurosci. 30, 6282–6290 (2010).
Wood, J. et al. Structure and function of the amygdaloid NPY system: NPY Y2 receptors regulate excitatory and inhibitory synaptic transmission in the centromedial amygdala. Brain Struct. Funct. 221, 3373–3391 (2016).
Verma, D. et al. NPY Y2 receptors in the central amygdala reduce cued but not contextual fear. Neuropharmacology 99, 665–674 (2015).
Nahvi, R. J. et al. Intranasal neuropeptide Y as a potential therapeutic for depressive behavior in the rodent single prolonged stress model in females. Front. Behav. Neurosci. 15, 705579 (2021).
Reichmann, F. & Holzer, P. Neuropeptide Y: A stressful review. Neuropeptides 55, 99–109 (2016).
Gryp, T., Glorieux, G., Joossens, M. & Vaneechoutte, M. Comparison of five assays for DNA extraction from bacterial cells in human faecal samples. J. Appl. Microbiol. 129, 378–388 (2020).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Xie, F., Xiao, P., Chen, D., Xu, L. & Zhang, B. miRDeepFinder: A miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol. Biol. https://doi.org/10.1007/s11103-012-9885-2 (2012).
Pais, M. L., Martins, J., Castelo-Branco, M. & Gonçalves, J. Increased susceptibility to kainate-induced seizures in a mouse model of tuberous sclerosis complex: Importance of sex and circadian cycle. Epilepsia Open 9, 1710–1722 (2024).
Ajayi, A. F. & Akhigbe, R. E. Staging of the estrous cycle and induction of estrus in experimental rodents: An update. Fertil. Res. Pract. 6, 5 (2020).
Karl, T. & Herzog, H. Behavioral profiling of NPY in aggression and neuropsychiatric diseases. Peptides 28, 326–333 (2007).
Tural, U. & Iosifescu, D. V. Neuropeptide Y in PTSD, MDD, and chronic stress: A systematic review and meta-analysis. J. Neurosci. Res. 98, 950–963 (2020).
Enman, N. M., Sabban, E. L., McGonigle, P. & Van Bockstaele, E. J. Targeting the neuropeptide Y system in stress-related psychiatric disorders. Neurobiol. Stress 1, 33–43 (2015).
Zalla, T. & Sperduti, M. The amygdala and the relevance detection theory of autism: An evolutionary perspective. Front. Hum. Neurosci. 7, 894 (2013).
Hill, J. W., Urban, J. H., Xu, M. & Levine, J. E. Estrogen induces Neuropeptide Y (NPY) Y1 receptor gene expression and responsiveness to NPY in gonadotrope-enriched pituitary cell cultures. Endocrinology 145, 2283–2290 (2004).
Acosta-Martinez, M., Horton, T. & Levine, J. E. Estrogen receptors in Neuropeptide Y neurons: At the crossroads of feeding and reproduction. Trends Endocrinol. Metab. 18, 48–50 (2007).
Morton, J. T. et al. Multi-level analysis of the gut-brain axis shows autism spectrum disorder-associated molecular and microbial profiles. Nat. Neurosci. 26, 1208–1217 (2023).
Bellone, C. & Lüscher, C. Bugs R Us: Restoring sociability with microbiota in autism. Cell reports. Med. 2, 100256 (2021).
Chen, Z. et al. Neuropeptide Y-mediated gut microbiota alterations aggravate postmenopausal osteoporosis. Adv. Sci. 10, 2303015 (2023).
Iliodromiti, Z. et al. Gut microbiome and neurodevelopmental disorders: A link yet to be disclosed. Microorganisms https://doi.org/10.3390/microorganisms11020487 (2023).
Sgritta, M. et al. Mechanisms underlying microbial-mediated changes in social behavior in mouse models of Autism Spectrum Disorder. Neuron 101, 246-259.e6 (2019).
Alamoudi, M. U. et al. Comparing the gut microbiome in Autism and preclinical models: A systematic review. Front. Cell. Infect. Microbiol. 12, 905841 (2022).
Fröhlich, E. E. et al. Cognitive impairment by antibiotic-induced gut dysbiosis: Analysis of gut microbiota-brain communication. Brain. Behav. Immun. 56, 140–155 (2016).
Nahvi, R. J. & Sabban, E. L. Sex differences in the Neuropeptide Y system and implications for stress related disorders. Biomolecules 10, 1–21 (2020).
Comeras, L. B., Hörmer, N., Mohan Bethuraj, P. & Tasan, R. O. NPY released from GABA neurons of the Dentate Gyrus specially reduces contextual fear without affecting cued or trace fear. Front. Synaptic Neurosci. 13, 635726 (2021).
Koponen, E., Lakso, M. & Castrén, E. Overexpression of the full-length neurotrophin receptor trkB regulates the expression of plasticity-related genes in mouse brain. Brain Res. Mol. Brain Res. 130, 81–94 (2004).
Méndez-Couz, M., González-Pardo, H., Arias, J. L. & Conejo, N. M. Hippocampal neuropeptide Y2 receptor blockade improves spatial memory retrieval and modulates limbic brain metabolism. Neurobiol. Learn. Mem. https://doi.org/10.1016/j.nlm.2021.107561 (2022).
Gonçalves, J. et al. Methamphetamine-induced changes in the mice hippocampal neuropeptide Y system: Implications for memory impairment. J. Neurochem. 123, 1041–1053 (2012).
Tang, T. et al. Structural basis for ligand recognition of the neuropeptide Y Y(2) receptor. Nat. Commun. 12, 737 (2021).
Casello, S. M. et al. Neuropeptide system regulation of prefrontal cortex circuitry: Implications for neuropsychiatric disorders. Front. Neural Circuits 16, 796443 (2022).
Sah, R., Ekhator, N. N., Jefferson-Wilson, L., Horn, P. S. & Geracioti, T. D. J. Cerebrospinal fluid neuropeptide Y in combat veterans with and without posttraumatic stress disorder. Psychoneuroendocrinology 40, 277–283 (2014).
Sah, R. et al. Low cerebrospinal fluid neuropeptide Y concentrations in posttraumatic stress disorder. Biol. Psychiatry 66, 705–707 (2009).
Wang, W. et al. NPY receptor 2 mediates NPY antidepressant effect in the mPFC of LPS rat by suppressing NLRP3 signaling pathway. Mediators Inflamm. 2019, 7898095 (2019).
Sharma, A., Ren, X., Zhang, H. & Pandey, G. N. Effect of depression and suicidal behavior on neuropeptide Y (NPY) and its receptors in the adult human brain: A postmortem study. Prog. Neuropsychopharmacol. Biol. Psychiatry 112, 110428 (2022).
Carboni, L., El Khoury, A., Beiderbeck, D. I., Neumann, I. D. & Mathé, A. A. Neuropeptide Y, calcitonin gene-related peptide, and neurokinin A in brain regions of HAB rats correlate with anxiety-like behaviours. Eur. Neuropsychopharmacol. 57, 1–14 (2022).
Paris, J. J. & Frye, C. A. Estrous cycle, pregnancy, and parity enhance performance of rats in object recognition or object placement tasks. Reproduction 136, 105–115 (2008).
Barth, C., Villringer, A. & Sacher, J. Sex hormones affect neurotransmitters and shape the adult female brain during hormonal transition periods. Front. Neurosci. 9, 37 (2015).
Martini, M., Sica, M., Gotti, S., Eva, C. & Panzica, G. C. Effects of estrous cycle and sex on the expression of neuropeptide Y Y1 receptor in discrete hypothalamic and limbic nuclei of transgenic mice. Peptides 32, 1330–1334 (2011).
Xu, M., Urban, J. H., Hill, J. W. & Levine, J. E. Regulation of hypothalamic neuropeptide Y Y1 receptor gene expression during the estrous cycle: Role of progesterone receptors. Endocrinology 141, 3319–3327 (2000).
Conrad, M., Hubold, C., Fischer, B. & Peters, A. Modeling the hypothalamus-pituitary-adrenal system: Homeostasis by interacting positive and negative feedback. J. Biol. Phys. 35, 149–162 (2009).
Lebron-Milad, K. & Milad, M. R. Sex differences, gonadal hormones and the fear extinction network: Implications for anxiety disorders. Biol. Mood Anxiety Disord. 2, 3 (2012).
Gehlert, D. R. Introduction to the reviews on neuropeptide Y. Neuropeptides 38, 135–140 (2004).
Redrobe, J. P., Dumont, Y., St-Pierre, J. A. & Quirion, R. Multiple receptors for neuropeptide Y in the hippocampus: Putative roles in seizures and cognition. Brain Res. 848, 153–166 (1999).
Wolcott, N. S., Redman, W. T., Karpinska, M., Jacobs, E. G. & Goard, M. J. The estrous cycle modulates hippocampal spine dynamics, dendritic processing, and spatial coding. bioRxiv Prepr. Serv. Biol. https://doi.org/10.1101/2024.08.02.606418 (2024).
Sotelo-Orozco, J. & Hertz-Picciotto, I. The association between gastrointestinal issues and psychometric scores in children with Autism Spectrum Disorder, developmental delays, Down Syndrome, and typical development. J. Autism Dev. Disord. https://doi.org/10.1007/s10803-024-06387-2 (2024).
Kushak, R. I. & Winter, H. S. Gut microbiota and gender in Autism Spectrum Disorders. Curr. Pediatr. Rev. 16, 249–254 (2020).
Bargiela, S., Steward, R. & Mandy, W. The experiences of late-diagnosed women with Autism Spectrum Conditions: An investigation of the female Autism Phenotype. J. Autism Dev. Disord. 46, 3281–3294 (2016).
Dean, M., Harwood, R. & Kasari, C. The art of camouflage: Gender differences in the social behaviors of girls and boys with autism spectrum disorder. Autism 21, 678–689 (2017).
Hull, L., Petrides, K. V. & Mandy, W. The female autism phenotype and camouflaging: A narrative review. Rev. J. Autism Dev. Disord. 7, 306–317 (2020).
Chang, C. & Lin, H. Dysbiosis in gastrointestinal disorders. Best Pract. Res. Clin. Gastroenterol. 30, 3–15 (2016).
Coretti, L. et al. Sex-related alterations of gut microbiota composition in the BTBR mouse model of autism spectrum disorder. Sci. Rep. 7, 45356 (2017).
Joo, M.-K. et al. Regulation of colonic neuropeptide Y expression by the gut microbiome in patients with ulcerative colitis and its association with anxiety- and depression-like behavior in mice. Gut Microbes 16, 2319844 (2024).
Acknowledgements
We thank André Alves for excellent technical support and assistance. The authors are grateful to the animal support staff at the Institute of Nuclear Sciences Applied to Health.
Funding
This work was supported by the Ph.D. Fellow UI/BD/150837/2021 (https://doi.org/10.54499/UI/BD/150837/2021) from FCT, FCT Exploratory Project 2022.01066.PTDC, Strategic Plan FCT/UIDP&B/04950/2025, COMPETE and FEDER funds, FCT, Portugal.
Author information
Authors and Affiliations
Contributions
BM: experimental design, conducted experiments, data collection/analysis/interpretation, manuscript preparation; JM: experimental design, data collection, manuscript editing; MCB: experimental design, data interpretation, manuscript editing, funding acquisition; JG: experimental design, data interpretation, manuscript preparation, manuscript editing, funding acquisition. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All animal procedures adhered to protocols approved by the ORBEA of the Institute of Nuclear Science Applied to Health (1/2017 and 8/2022).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martins, B., Martins, J., Castelo-Branco, M. et al. Sex-dependent dysregulation of the gut-brain NPYergic system in a mouse model of autism spectrum disorder. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42601-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42601-0