Abstract
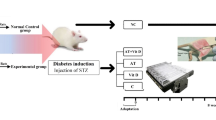
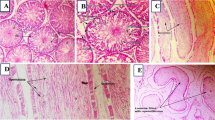
Bisphenol S (BPS) is a recognized environmental contaminant that harms reproductive organs and fertility, affecting human health worldwide. Over the last few decades, the search for a compound that mitigates its harmful effects has increased. Vitamin E has not been evaluated in diabetic rats as a study model. We assessed for the first time the use of Vitamin E as a potential ameliorant compound. We used 26 Wistar rats, and assigned it randomly in five groups: (1) healthy rats (Ctrl, n = 6); (2) diabetic rats without treatment (Ctrl-D, n = 5); (3) diabetic rats treated with Vitamin E (100 mg/kg bw/day, VitE-D, n = 5); (4) diabetic rats treated with BPS (100 mg/kg bw/day, BPS-D, n = 5); and, (5) diabetic rats receiving a combination of Vitamin E (100 mg/kg bw/day) and BPS (100 mg/kg bw/day) (VitE + BPS-D, n = 5). All doses were administered orally (p.o.). We evaluated its effect on serum estradiol and testosterone levels, testis cellular apoptosis, antioxidant enzyme activity, and sperm and testicular histologic characteristics. BPS increases oxidative stress, promotes cell apoptosis, provokes structural changes in seminiferous tubules, and negatively affects spermatogenesis and sperm quality. As a result of our study, co-administration of Vitamin E did not reduce the negative impact provoked by BPS; indeed, in some cases, the vitamin exacerbated the injury. The beneficial effects of VitE on testosterone serum levels were nullified when combined with BPS. Our results show the dangers of BPS to male reproductive health in the diabetes model and stress the necessity for improved strategies to mitigate its deleterious impacts.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Vandenberg, L. N., Hunt, P. A., Myers, J. P. & Vom Saal, F. S. Human exposures to bisphenol A: mismatches between data and assumptions. Rev. Environ. Health. 28 (1), 37–58. https://doi.org/10.1515/reveh-2012-0034 (2013).
Abouhamzeh, B., Zare, Z., Mohammadi, M., Moosazadeh, M. & Nourian, A. Bisphenol-S Influence on Oxidative Stress and Endocrine Biomarkers of Reproductive System: A Systematic Review and Meta-Analysis. Int. J. Prev. Med. 14, 37. https://doi.org/10.4103/ijpvm.ijpvm_271_21 (2023).
Ahbab, M. A., Barlas, N. & Karabulut, G. The toxicological effects of bisphenol A and octylphenol on the reproductive system of prepubertal male rats. Toxicol. Ind. Health. 33 (2), 133–146. https://doi.org/10.1177/0748233715603847 (2017).
Cimmino, I. et al. Potential Mechanisms of Bisphenol A (BPA) Contributing to Human Disease. Int. J. Mol. Sci. 21 (16). https://doi.org/10.3390/ijms21165761 (2020).
Siddique, S., Zhang, G. & Kubwabo, C. Exposure to bisphenol a and risk of developing type 2 diabetes: A mini review. Emerg. Contaminants. 6, 274–282. https://doi.org/10.1016/j.emcon.2020.07.005 (2020).
Lee, S., Kim, C., Shin, H., Kho, Y. & Choi, K. Comparison of thyroid hormone disruption potentials by bisphenols A, S, F, and Z in embryo-larval zebrafish. Chemosphere 221, 115–123. https://doi.org/10.1016/j.chemosphere.2019.01.019 (2019).
Cao, T. et al. The Effect of Exposure to Bisphenol A on Spermatozoon and the Expression of Tight Junction Protein Occludin in Male Mice. Dose Response. 18 (2), 1559325820926745. https://doi.org/10.1177/1559325820926745 (2020).
Xu, J., Huang, G., Nagy, T., Teng, Q. & Guo, T. L. Sex-dependent effects of bisphenol A on type 1 diabetes development in non-obese diabetic (NOD) mice. Arch. Toxicol. 93 (4), 997–1008. https://doi.org/10.1007/s00204-018-2379-5 (2019).
Bansal, A. et al. Transgenerational effects of maternal bisphenol: a exposure on offspring metabolic health. J. Dev. Orig Health Dis. 10 (2), 164–175. https://doi.org/10.1017/S2040174418000764 (2019).
Yang, Y., Guan, J., Yin, J., Shao, B. & Li, H. Urinary levels of bisphenol analogues in residents living near a manufacturing plant in south China. Chemosphere 112, 481–486. https://doi.org/10.1016/j.chemosphere.2014.05.004 (2014).
Mandel, N., Gamboa, B., Cebrián, M. & Mérida-Ortega, Á. Challenges to regulate products containing bisphenol A: Implications for policy. Salud Pública de México. 61, 692. https://doi.org/10.21149/10411 (2019).
Kaptaner, B. et al. Bisphenol S leads to cytotoxicity-induced antioxidant responses and oxidative stress in isolated rainbow trout (Oncorhyncus mykiss) hepatocytes. Mol. Biol. Rep. 48, 1–10. https://doi.org/10.1007/s11033-021-06771-6 (2021).
Alharbi, H. F. et al. Exposure to Bisphenol A Substitutes, Bisphenol S and Bisphenol F, and Its Association with Developing Obesity and Diabetes Mellitus: A Narrative Review. Int. J. Environ. Res. Public. Health. 19 (23). https://doi.org/10.3390/ijerph192315918 (2022).
Azevedo, L. F. et al. Long-term exposure to bisphenol A or S promotes glucose intolerance and changes hepatic mitochondrial metabolism in male Wistar rats. Food Chem. Toxicol. 132, 110694. https://doi.org/10.1016/j.fct.2019.110694 (2019).
Peña-Corona, S. I. et al. Vitamin E (α-Tocopherol) Does Not Ameliorate the Toxic Effect of Bisphenol S on the Metabolic Analytes and Pancreas Histoarchitecture of Diabetic Rats. Toxics 11 (7). https://doi.org/10.3390/toxics11070626 (2023).
Mandrah, K., Jain, V., Ansari, J. A. & Roy, S. K. Metabolomic perturbation precedes glycolytic dysfunction and procreates hyperglycemia in a rat model due to bisphenol S exposure. Environ. Toxicol. Pharmacol. 77, 103372. https://doi.org/10.1016/j.etap.2020.103372 (2020).
Masenga, S. K., Kabwe, L. S., Chakulya, M. & Kirabo, A. Mechanisms of Oxidative Stress in Metabolic Syndrome. Int. J. Mol. Sci. 24 (9). https://doi.org/10.3390/ijms24097898 (2023).
Jakubiak, G. K. et al. Obesity and Insulin Resistance Is the Component of the Metabolic Syndrome Most Strongly Associated with Oxidative Stress. Antioxid. (Basel). 11 (1). https://doi.org/10.3390/antiox11010079 (2021).
Choi, S. I. et al. Potentilla rugulosa Nakai Extract Attenuates Bisphenol A-, S- and F-Induced ROS Production and Differentiation of 3T3-L1 Preadipocytes in the Absence of Dexamethasone. Antioxid. (Basel). 9 (2). https://doi.org/10.3390/antiox9020113 (2020).
Kose, O. et al. The effects of different bisphenol derivatives on oxidative stress, DNA damage and DNA repair in RWPE-1 cells: A comparative study. J. Appl. Toxicol. 40 (5), 643–654. https://doi.org/10.1002/jat.3934 (2020).
Peña-Corona, S. I. et al. Melatonin and Vitamins as Protectors against the Reproductive Toxicity of Bisphenols: Which Is the Most Effective? A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 24 (19). https://doi.org/10.3390/ijms241914930 (2023).
Traber, M. G. & Atkinson, J. Vitamin E, antioxidant and nothing more. Free Radic Biol. Med. 43 (1), 4–15. https://doi.org/10.1016/j.freeradbiomed.2007.03.024 (2007).
Al Shamsi, M. S., Amin, A. & Adeghate, E. Beneficial effect of vitamin E on the metabolic parameters of diabetic rats. Mol. Cell. Biochem. 261 (1–2), 35–42. https://doi.org/10.1023/b:mcbi.0000028735.79172.9b (2004).
Mercuri, F., Quagliaro, L. & Ceriello, A. Oxidative stress evaluation in diabetes. Diabetes Technol. Ther. 2 (4), 589–600. https://doi.org/10.1089/15209150050502014 (2000).
Dickey, D. T. et al. Effect of N-acetylcysteine route of administration on chemoprotection against cisplatin-induced toxicity in rat models. Cancer Chemother. Pharmacol. 62 (2), 235–241. https://doi.org/10.1007/s00280-007-0597-2 (2008).
Sprong, R. C. et al. Low-dose N-acetylcysteine protects rats against endotoxin-mediated oxidative stress, but high-dose increases mortality. Am. J. Respir Crit. Care Med. 157 (4 Pt 1), 1283–1293. https://doi.org/10.1164/ajrccm.157.4.9508063 (1998).
Hybertson, B. M., Kitlowski, R. P., Jepson, E. K. & Repine, J. E. Supercritical fluid-aerosolized vitamin E pretreatment decreases leak in isolated oxidant-perfused rat lungs. J. Appl. Physiol. (1985). 84 (1), 263–268. https://doi.org/10.1152/jappl.1998.84.1.263 (1998).
CamurçaFD et al. Gastric emptying and gastrointestinal transit of liquid in awake rats is delayed after acute myocardial infarction. Dig. Dis. Sci. 49 (5), 757–762. https://doi.org/10.1023/b:ddas.0000030085.17586.06 (2004).
Pal, S. et al. Bisphenol S impairs blood functions and induces cardiovascular risks in rats. Toxicol. Rep. 4, 560–565 (2017).
Rashad, S., Ahmed, S., El-Sayed, M. & Ahmed, D. The Toxic Effect of Bisphenol A on Albino Rat Testicles and the Possible Protective Value of Vitamin E and Melatonin. Egyptian Society of. Clin. Toxicol. J. 9 (2), 1–12. https://doi.org/10.21608/esctj.2021.63294.1001 (2021).
Amraoui, W. et al. Modulatory Role of Selenium and Vitamin E, Natural Antioxidants, against Bisphenol A-Induced Oxidative Stress in Wistar Albinos Rats. Toxicol. Res. 34 (3), 231–239. https://doi.org/10.5487/TR.2018.34.3.231 (2018).
Al-Mousawi, A. M. et al. Impact of anesthesia, analgesia, and euthanasia technique on the inflammatory cytokine profile in a rodent model of severe burn injury. Shock 34 (3), 261–268. https://doi.org/10.1097/shk.0b013e3181d8e2a6 (2010).
Pérez-Rojas, J. M. et al. Preventive effect of tert-butylhydroquinone on cisplatin-induced nephrotoxicity in rats. Food Chem. Toxicol. 49 (10), 2631–2637. https://doi.org/10.1016/j.fct.2011.07.008 (2011).
Paglia, D. E. & Valentine, W. N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70 (1), 158–169 (1967).
Carlberg, I. & Mannervik, B. Glutathione reductase. Methods Enzymol. 113, 484–490. https://doi.org/10.1016/s0076-6879(85)13062-4 (1985).
Habig, W. H., Pabst, M. J. & Jakoby, W. B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249 (22), 7130–7139 (1974).
Aebi, H. Catalase in vitro. Methods Enzymol. 105, 121–126. https://doi.org/10.1016/s0076-6879(84)05016-3 (1984).
Beauchamp, C. & Fridovich, I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 44 (1), 276–287. https://doi.org/10.1016/0003-2697(71)90370-8 (1971).
Nishikimi, M., Appaji, N. & Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 46 (2), 849–854. https://doi.org/10.1016/s0006-291x(72)80218-3 (1972).
Johnsen, S. G. Testicular biopsy score count–a method for registration of spermatogenesis in human testes: normal values and results in 335 hypogonadal males. Hormones 1 (1), 2–25. https://doi.org/10.1159/000178170 (1970).
Ws, R. ImageJ, U.S. National Institutes of Health, Bethesda, Maryland, USA. (2011).
WHO. WHO laboratory manual for the examination and processing of human semen, 6th ed. (2021).
Menkveld, R. Sperm Morphology Assessment Using Strict (Tygerberg) Criteria. In: (eds Carrell, D. T. & Aston, K. I.) Spermatogenesis: Methods and Protocols. Humana, Totowa, NJ, 39–50. doi:https://doi.org/10.1007/978-1-62703-038-0_5 (2013).
Meng, J., Holdcraft, R. W., Shima, J. E., Griswold, M. D. & Braun, R. E. Androgens regulate the permeability of the blood-testis barrier. Proc. Natl. Acad. Sci. U S A. 102 (46), 16696–16700. https://doi.org/10.1073/pnas.0506084102 (2005).
Wen, Q. et al. Dynein 1 supports spermatid transport and spermiation during spermatogenesis in the rat testis. Am. J. Physiol. Endocrinol. Metab. 315 (5), E924–E948. https://doi.org/10.1152/ajpendo.00114.2018 (2018).
Mok, K. W., Mruk, D. D., Lee, W. M. & Cheng, C. Y. A study to assess the assembly of a functional blood-testis barrier in developing rat testes. Spermatogenesis 1 (3), 270–280. https://doi.org/10.4161/spmg.1.3.17998 (2011).
Vigueras-Villaseñor, R. M. et al. Effect of allopurinol on damage caused by free radicals to cryptorchid testes. Acta Histochem. 111 (2), 127–137. https://doi.org/10.1016/j.acthis.2008.05.004 (2009).
Roser, M., Ritchie, H. & Spooner, F. Burden of disease (Our World Data, 2021).
Selvin, E. & Juraschek, S. P. Diabetes Epidemiology in the COVID-19 Pandemic. Diabetes Care. 43 (8), 1690–1694. https://doi.org/10.2337/dc20-1295 (2020).
Martínez-Pinna, J. et al. Endocrine disruptors in plastics alter β-cell physiology and increase the risk of diabetes mellitus. Am. J. Physiol. Endocrinol. Metab. 324 (6), E488–E505. https://doi.org/10.1152/ajpendo.00068.2023 (2023).
Ogurtsova, K. et al. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 128, 40–50. https://doi.org/10.1016/j.diabres.2017.03.024 (2017).
Dai, W., He, Q. Z., Zhu, B. Q. & Zeng, H. C. Oxidative stress-mediated apoptosis is involved in bisphenol S-induced reproductive toxicity in male C57BL/6 mice. J. Appl. Toxicol. 41 (11), 1839–1851. https://doi.org/10.1002/jat.4170 (2021).
Shi, M., Sekulovski, N., MacLean, J. A. & Hayashi, K. Prenatal Exposure to Bisphenol A Analogues on Male Reproductive Functions in Mice. Toxicol. Sci. 163 (2), 620–631. https://doi.org/10.1093/toxsci/kfy061 (2018).
Sahu, A. & Verma, R. Bisphenol S dysregulates thyroid hormone homeostasis; Testicular survival, redox and metabolic status: Ameliorative actions of melatonin. Environ. Toxicol. Pharmacol. 104, 104300. https://doi.org/10.1016/j.etap.2023.104300 (2023).
Kumar, J., Verma, R. & Haldar, C. Melatonin ameliorates Bisphenol S induced testicular damages by modulating Nrf-2/HO-1 and SIRT-1/FOXO-1 expressions. Environ. Toxicol. 36 (3), 396–407. https://doi.org/10.1002/tox.23045 (2021).
Fenclová, T. et al. Nursing Exposure to Bisphenols as a Cause of Male Idiopathic Infertility. Front. Physiol. 13, 725442. https://doi.org/10.3389/fphys.2022.725442 (2022).
Murata, M. & Kang, J. H. Bisphenol A (BPA) and cell signaling pathways. Biotechnol. Adv. 36 (1), 311–327. https://doi.org/10.1016/j.biotechadv.2017.12.002 (2018).
Barbagallo, F. et al. Effects of Bisphenols on Testicular Steroidogenesis. Front. Endocrinol. (Lausanne). 11, 373. https://doi.org/10.3389/fendo.2020.00373 (2020).
Daian, L. M., Tanko, G., Vacaru, A. M., Ghila, L. & Chera, S. Modulation of Unfolded Protein Response Restores Survival and Function of β-Cells Exposed to the Endocrine Disruptor Bisphenol A. Int. J. Mol. Sci. 24 (3). https://doi.org/10.3390/ijms24032023 (2023).
Cao, S. S. & Kaufman, R. J. Endoplasmic reticulum stress and oxidative stress in cell fate decision and human disease. Antioxid. Redox Signal. 21 (3), 396–413. https://doi.org/10.1089/ars.2014.5851 (2014).
Adegoke, E. O., Rahman, M. S. & Pang, M. G. Bisphenols Threaten Male Reproductive Health via Testicular Cells. Front. Endocrinol. (Lausanne). 11, 624. https://doi.org/10.3389/fendo.2020.00624 (2020).
Amjad, S., Rahman, M. S. & Pang, M. G. Role of Antioxidants in Alleviating Bisphenol A Toxicity. Biomolecules 10 (8). https://doi.org/10.3390/biom10081105 (2020).
Jiang, W. et al. Potential effects of bisphenol A on diabetes mellitus and its chronic complications: A narrative review. Heliyon 9 (5), e16340. https://doi.org/10.1016/j.heliyon.2023.e16340 (2023).
Vivarelli, F. et al. Co-carcinogenic effects of vitamin E in prostate. Sci. Rep. 9 (1), 11636. https://doi.org/10.1038/s41598-019-48213-1 (2019).
Lee, Y. Y. et al. Activities of antioxidant and redox enzymes in human normal hepatic and hepatoma cell lines. Mol. Cells. 14 (2), 305–311 (2002).
Aitken, R. J. & Roman, S. D. Antioxidant Systems and Oxidative Stress in the Testes (Madame Curie Bioscience Database, 2013).
Darghouthi, M., Rezg, R., Boughmadi, O. & Mornagui, B. Low-dose bisphenol S exposure induces hypospermatogenesis and mitochondrial dysfunction in rats: A possible implication of StAR protein. Reprod. Toxicol. 107, 104–111. https://doi.org/10.1016/j.reprotox.2021.11.007 (2022).
Awadallah, S. Protein Antioxidants in Thalassemia. vol 60 Advances in Clinical Chemistry. (2013). https://doi.org/10.1016/B978-0-12-407681-5.00003-9
Golestani, A. et al. Paradoxical dose- and time-dependent regulation of superoxide dismutase and antioxidant capacity by vitamin E in rat. Clin. Chim. Acta. 365 https://doi.org/10.1016/j.cca.2005.08.008 (2006).
Zheng, M. et al. The Applications and Mechanisms of Superoxide Dismutase in Medicine, Food, and Cosmetics. Antioxid. (Basel). 12 (9). https://doi.org/10.3390/antiox12091675 (2023).
Traber, M. G. & Bruno, R. S. Vitamin E. Present Knowledge in Nutrition. Academic Press. (2020). https://doi.org/10.1016/B978-0-323-66162-1.00007-X
Chen, X., Touyz, R. M., Park, J. B. & Schiffrin, E. L. Antioxidant effects of vitamins C and E are associated with altered activation of vascular NADPH oxidase and superoxide dismutase in stroke-prone SHR. Hypertension 38 (3 Pt 2), 606–611. https://doi.org/10.1161/hy09t1.094005 (2001).
Kontush, A., Finckh, B., Karten, B., Kohlschutter, A. & Beisiegel, U. Antioxidant and prooxidant activity of a-tocopherol in human plasma and low density lipoprotein. vol 37. Journal of Lipid Research.
Aitken, R. J. & Roman, S. D. Antioxidant Systems and Oxidative Stress in the Testes. (2013).
Nna, V. U., Abu Bakar, A. B., Ahmad, A., Eleazu, C. O. & Mohamed, M. Oxidative Stress, NF-κB-Mediated Inflammation and Apoptosis in the Testes of Streptozotocin-Induced Diabetic Rats: Combined Protective Effects of Malaysian Propolis and Metformin. Antioxid. (Basel). 8(10). https://doi.org/10.3390/antiox8100465 (2019).
Kumar, N. & Singh, A. K. Trends of male factor infertility, an important cause of infertility: A review of literature. J. Hum. Reprod. Sci. 8 (4), 191–196. https://doi.org/10.4103/0974-1208.170370 (2015).
Wu, H. et al. Bisphenol S perturbs Sertoli cell junctions in male rats via alterations in cytoskeletal organization mediated by an imbalance between mTORC1 and mTORC2 (Science of The Total Environment, 2021).
Naderi, M., Wong, M. Y. & Gholami, F. Developmental exposure of zebrafish (Danio rerio) to bisphenol-S impairs subsequent reproduction potential and hormonal balance in adults. Aquat. Toxicol. 148, 195–203. https://doi.org/10.1016/j.aquatox.2014.01.009 (2014).
Ghayda, R. A. et al. Urinary bisphenol S concentrations: Potential predictors of and associations with semen quality parameters among men attending a fertility center. Environ. Int. 131, 105050. https://doi.org/10.1016/j.envint.2019.105050 (2019).
Pathak, M., Sharma, S., Kushwaha, P. & Kumar, S. Functional lead compounds and targets for the development of drugs for the treatment of male infertility. Phytochemicals as Lead Compounds for New Drug Discovery. (2020). https://doi.org/10.1016/B978-0-12-817890-4.00022-6
Pei, J., Pan, X., Wei, G. & Hua, Y. Research progress of glutathione peroxidase family (GPX) in redoxidation. Front. Pharmacol. 14, 1147414. https://doi.org/10.3389/fphar.2023.1147414 (2023).
Aitken, R. J. & Roman, S. D. Antioxidant systems and oxidative stress in the testes. Oxid. Med. Cell. Longev. 1 (1), 15–24. https://doi.org/10.4161/oxim.1.1.6843 (2008).
Umeda, F., Kato, K., Muta, K. & Ibayashi, H. Effect of vitamin E on function of pituitary-gonadal axis in male rats and human subjects. Endocrinol. Jpn. 29 (3), 287–292. https://doi.org/10.1507/endocrj1954.29.287 (1982).
Molangiri, A., Varma, S., Kambham, M. S., Duttaroy, S., Basak, A. K. & S Prenatal exposure to bisphenol S and bisphenol A differentially affects male reproductive system in the adult offspring. Food Chem. Toxicol. 167, 113292. https://doi.org/10.1016/j.fct.2022.113292 (2022).
Ullah, H. et al. Chronic exposure of bisphenol S (BPS) affect hypothalamic-pituitary-testicular activities in adult male rats: possible in estrogenic mode of action. Environ. Health Prev. Med. 26 (1), 31. https://doi.org/10.1186/s12199-021-00954-0 (2021).
Řimnáčová, H. et al. Low doses of Bisphenol S affect post-translational modifications of sperm proteins in male mice. Reprod. Biol. Endocrinol. 18 (1), 56. https://doi.org/10.1186/s12958-020-00596-x (2020).
Dutta, S., Sengupta, P. & Muhamad, S. Male reproductive hormones and semen quality. Asian Pac. J. Reprod. 8 https://doi.org/10.4103/2305-0500.268132 (2019).
Ichihara, I. The fine structure of testicular interstitial cells in the mouse administered with vitamin E. Okajimas Folia Anat. Jpn. 43 (3), 203–217. https://doi.org/10.2535/ofaj1936.43.3-4_203 (1967).
de Freitas, A. T. A. G. et al. Regulatory and junctional proteins of the blood-testis barrier in human Sertoli cells are modified by monobutyl phthalate (MBP) and bisphenol A (BPA) exposure. Toxicol. Vitro. 34, 1–7. https://doi.org/10.1016/j.tiv.2016.02.017 (2016).
Akintunde, J. K., Farouk, A. A. & Mogbojuri, O. Metabolic treatment of syndrome linked with Parkinson’s disease and hypothalamus pituitary gonadal hormones by turmeric curcumin in Bisphenol-A induced neuro-testicular dysfunction of wistar rat. Biochem. Biophys. Rep. 17, 97–107. https://doi.org/10.1016/j.bbrep.2018.12.004 (2019).
Gonçalves, G. D., Semprebon, S. C., Biazi, B. I., Mantovani, M. S. & Fernandes, G. S. A. Bisphenol A reduces testosterone production in TM3 Leydig cells independently of its effects on cell death and mitochondrial membrane potential. Reprod. Toxicol. 76, 26–34. https://doi.org/10.1016/j.reprotox.2017.12.002 (2018).
Wu, H. et al. Bisphenol S perturbs Sertoli cell junctions in male rats via alterations in cytoskeletal organization mediated by an imbalance between mTORC1 and mTORC2. Sci. Total Environ. 762 https://doi.org/10.1016/j.scitotenv.2020.144059 (2021).
Ji, K., Hong, S., Kho, Y. & Choi, K. Effects of bisphenol s exposure on endocrine functions and reproduction of zebrafish. Environ. Sci. Technol. 47 (15), 8793–8800. https://doi.org/10.1021/es400329t (2013).
Meccariello, R. et al. Intra-testicular signals regulate germ cell progression and production of qualitatively mature spermatozoa in vertebrates. Front. Endocrinol. (Lausanne). 5, 69. https://doi.org/10.3389/fendo.2014.00069 (2014).
Feng, Y. et al. Effects of bisphenol analogues on steroidogenic gene expression and hormone synthesis in H295R cells. vol 147. Chemosphere. https://doi.org/10.1016/j.chemosphere.2015.12.081 (2016)
Acknowledgements
We acknowledge MSc. Omar Noel Medina for providing technical support in sample processing for antioxidant evaluation, and MVZ Lucía Macias and Francisco Sánchez for their assistance in animal handling. Sheila Irais Peña-Corona (CVU:495850) acknowledges the Postdoctoral Program Scholarship of Secretaría de Ciencia, Humanidades, Tecnología e Innovación ‘Estancias Posdoctorales por México 2022; Estancia PosdoctoralAcadémica Inicial 2022’. Juan Isaac Chávez-Corona is a PhD student in the Programa de Doctorado en Ciencias de la Producción y de la Salud Animal de la Facultad de Medicina Veterinaria y Zootecnia de la Universidad Nacional Autónoma de México.
Funding
This paper was supported by the Programa de Apoyo a la Investigación y al Posgrado (PAIP; grants 5000–9141 and 5000–9105), Facultad de Química, UNAM; by Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT, IN202725, IN204722), UNAM; by Programa de Apoyo a Proyectos para Innovar y Mejorar la Educación (PAPIME, PE205524), and grant 319631 Fronteras de la Ciencia 2022, CONACyT.
Author information
Authors and Affiliations
Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas. That is revising or critically reviewing the article; giving final approval of the version to be published; agreeing on the journal to which the article has been submitted; and, confirming to be accountable for all aspects of the work.
Corresponding authors
Ethics declarations
Competing interests
Authors wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Ethics approval and consent to participate
Not Applicable.
Consent for publication
Not Applicable.
Clinical trial number
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Peña-Corona, S.I., Chávez-Corona, J.I., Ruiz-García, O.V. et al. Vitamin E fails to prevent bisphenol S induced testicular damage in diabetic rats. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42624-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42624-7