Abstract
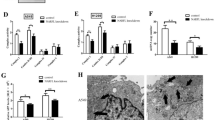
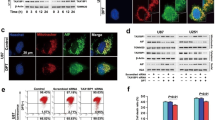
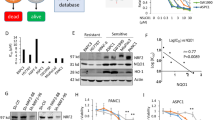
This study aimed to investigate the molecular mechanism by which propofol enhances chemosensitivity in non-small cell lung cancer (NSCLC). In vitro experiments were performed to evaluate the effects of propofol on NSCLC cell lines and to elucidate the underlying molecular mechanisms. In vivo, A549/DDP cells were subcutaneously injected into nude mice, followed by cisplatin (DDP) treatment, and tumor tissues were subsequently analyzed by hematoxylin–eosin staining and immunohistochemistry. Our results demonstrated that propofol significantly enhanced the sensitivity of A549/DDP cells to cisplatin by promoting Parthanatos. This process was characterized by increased apoptosis-inducing factor (AIF) and macrophage migration inhibitory factor (MIF) binding and nuclear translocation, loss of mitochondrial membrane potential, nicotinamide adenine dinucleotide (NAD+) depletion, accumulation of poly(ADP-ribose) (PAR), and increased expression of the DNA damage marker phosphorylated histone H2AX (γH2AX). Moreover, propofol treatment was associated with elevated interleukin-6 (IL-6) levels. Mechanistically, overexpression of methyltransferase-like 3 (METTL3) enhanced PARP-1 m6A modification and Parthanatos activation, whereas METTL3 knockdown exerted the opposite effects. Furthermore, propofol enhanced cisplatin sensitivity by regulating METTL3-mediated m6A modification of PARP-1 in vitro, which was further confirmed in vivo. In conclusion, propofol enhances cisplatin chemosensitivity in NSCLC by activating Parthanatos through modulation of METTL3-mediated PARP-1 m6A modification. These findings provide mechanistic insight into propofol-mediated reversal of chemoresistance and identify the METTL3–PARP-1–Parthanatos axis as a potential therapeutic target in NSCLC.
Similar content being viewed by others
Data availability
All data in this study are available from the corresponding author on reasonable request.
References
Gosney, J. R., Boothman, A. M., Ratcliffe, M. & Kerr, K. M. Cytology for PD-L1 testing: a systematic review. Lung Cancer. 141, 101–106 (2020).
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Qiu, H., Cao, S. & Xu, R. Cancer incidence, mortality, and burden in China: a time-trend analysis and comparison with the United States and United Kingdom based on the global epidemiological data released in 2020. Cancer Commun. (Lond.) 41, 1037–1048 (2021).
Siegel, M. K. D. et al. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 70, 145–164 (2020).
Zou, B., Zhou, X. L., Lai, S. Q. & Liu, J. C. Notch signaling and non-small cell lung cancer. Oncol. Lett. 15, 3415–3421 (2018).
Chen, Y. et al. Effects and mechanism of microRNA–218 against lung cancer. Mol. Med. Rep. 23, 8596 (2021).
Zheng, X. et al. Propofol affects non-small-cell lung cancer cell biology by regulating the miR-21/PTEN/AKT pathway in vitro and in vivo. Anesth. Analg. 131, 1270–1280 (2020).
Liu, J. et al. AS-IV enhances the antitumor effects of propofol in NSCLC cells by inhibiting autophagy. Open. Med. (Warsaw Poland). 18, 20230799 (2023).
Ling, Q., Wu, S., Liao, X., Liu, C. & Chen, Y. Anesthetic propofol enhances cisplatin-sensitivity of non-small cell lung cancer cells through N6-methyladenosine-dependently regulating the miR-486-5p/RAP1-NF-κB axis. BMC cancer. 22, 765 (2022).
Wang, Y. et al. A nuclease that mediates cell death induced by DNA damage and poly(ADP-ribose) polymerase-1. Science 354, 7485 (2016).
Huang, P. et al. Molecular mechanisms of Parthanatos and its role in diverse diseases. Int. J. Mol. Sci. 23, 2563 (2022).
Zhou, Y. et al. Parthanatos and its associated components: promising therapeutic targets for cancer. Pharmacol. Res. 163, 105299 (2021).
Zhang, Y. et al. Inhibition of AKT induces p53/SIRT6/PARP1-dependent parthanatos to suppress tumor growth. Cell. Commun. Signal. 20, 93 (2022).
Ribeiro, D. L. et al. The antitumoral/antimetastatic action of the flavonoid brachydin A in metastatic prostate tumor spheroids in vitro is mediated by (Parthanatos) PARP-related cell death. Pharmaceutics 14, 2563 (2022).
Ling, Q., Wu, S., Liao, X., Liu, C. & Chen, Y. Anesthetic propofol enhances cisplatin-sensitivity of non-small cell lung cancer cells through N6-methyladenosine-dependently regulating the miR-486-5p/RAP1-NF-kappaB axis. BMC Cancer. 22, 765 (2022).
Zhong, H. et al. Propofol inhibits parthanatos via ROS-ER-calcium-mitochondria signal pathway in vivo and vitro. Cell Death Dis. 9, 932 (2018).
Zhang, Z. et al. m(6)A regulators as predictive biomarkers for chemotherapy benefit and potential therapeutic targets for overcoming chemotherapy resistance in small-cell lung cancer. J. Hematol. Oncol. 14, 190 (2021).
Guan, S. et al. Germline USP36 mutation confers resistance to EGFR-TKIs by upregulating MLLT3 expression in patients with non-small cell lung cancer. Clin. Cancer Res. 30, 1382–1396 (2024).
Li, Z., Li, Y., Shen, L., Shen, L. & Li, N. Molecular characterization, clinical relevance and immune feature of m7G regulator genes across 33 cancer types. Front. Genet. 13, 981567 (2022).
Lu, Y. et al. Propofol-induced MiR-20b expression initiates endogenous cellular signal changes mitigating hypoxia/re-oxygenation-induced endothelial autophagy in vitro. Cell. Death Dis. 11, 681 (2020).
Li, H. et al. METTL3 promotes oxaliplatin resistance of gastric cancer CD133 + stem cells by promoting PARP1 mRNA stability. Cell. Mol. Life Sci. 79, 135 (2022).
Li, D. et al. Oxaliplatin induces the PARP1-mediated parthanatos in oral squamous cell carcinoma by increasing production of ROS. Aging 13, 4242–4257 (2021).
Liu, H. & Wang, P. CRISPR screening and cell line IC50 data reveal novel key genes for trametinib resistance. Clin. Exp. Med. 25, 21 (2024).
Chen, Z. & Er Saw, P. Integration in biomedical science 2024: emerging trends in the post-pandemic era. BIO Integr. 5, 1–2 (2024).
Liu, H., Dilger, J. P. & Lin, J. Effects of local anesthetics on cancer cells. Pharmacol. Ther. 212, 107558 (2020).
Li, R. et al. Effects of local anesthetics on breast cancer cell viability and migration. BMC Cancer. 18, 666 (2018).
Liu, H., Dilger, J. P. & Lin, J. Lidocaine suppresses viability and migration of human breast cancer cells: TRPM7 as a target for some breast cancer cell lines. Cancers (Basel) 13, 2563 (2021).
Li, R., Liu, H., Dilger, J. P. & Lin, J. Effect of propofol on breast cancer cell, the immune system, and patient outcome. BMC Anesthesiol. 18, 77 (2018).
Chen, D. et al. Allosteric inhibitors of macrophage migration inhibitory factor (MIF) interfere with apoptosis-inducing factor (AIF) co-localization to prevent Parthanatos. J. Med. Chem. 66, 8767–8781 (2023).
Medrano, M. et al. Cannabinoids induce cell death in leukaemic cells through Parthanatos and PARP-related metabolic disruptions. Br. J. Cancer. 130, 1529–1541 (2024).
Li, R. et al. The potential effect of general anesthetics in cancer surgery: meta-analysis of postoperative metastasis and inflammatory cytokines. Cancers (Basel) 15, 14523 (2023).
Jin, Z. et al. Potential therapeutic application of local anesthetics in cancer treatment. Recent. Pat. Anticancer Drug Discov. 17, 326–342 (2022).
Kang, Z. R. et al. Deficiency of BCAT2-mediated branched-chain amino acid catabolism promotes colorectal cancer development. Biochim. Biophys. Acta Mol. Basis Dis. 1870, 166941 (2024).
Lee, S. J. et al. hsa-miR-CHA2, a novel microRNA, exhibits anticancer effects by suppressing cyclin E1 in human non-small cell lung cancer cells. Biochim. Biophys. Acta Mol. Basis Dis. 1870, 167250 (2024).
Qian, X. J., Wang, J. W., Liu, J. B. & Yu, X. The mediating role of miR-451/ETV4/MMP13 signaling axis on epithelialmesenchymal transition in promoting non-small cell lung cancer progression. Curr. Mol. Pharmacol. 17, e210723218988 (2024).
Xie, M., Liu, B., Chen, Z., Cao, T. & Zhang, X. Transcriptomic signatures in TP53 positive and negative tumor samples in NSCLC. Curr. Gene Ther. (2025).
Acknowledgements
The authors express their appreciation to staff in Zhongshan People’s Hospital, for their technical assistance.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Quan Ling, Yong Chen: study design, data analysis, drafting the manuscript and revision of the manuscript. Kepeng Liu, Junlin Wen, Jin Liu: data collection and analysis, drafting the manuscript, investigation. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Our study has been approved by laboratory animal care and ethics committee of Guangdong Laidi Biomedical Research lnstitute Co, LTD (Approval No. 2024027-3).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ling, Q., Liu, K., Wen, J. et al. Propofol regulates METTL3-mediated PARP-1 m6A modification to promote Parthanatos to improve NSCLC chemotherapy resistance. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42665-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42665-y