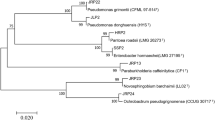
Abstract
In this study, endospore-forming bacteria were isolated, screened, and characterized from samples collected in Tan Phu protection forest for potential use in biofertilizer application. Twelve endospore-forming bacteria showed the ability to produce indole-3-acetic acid were evaluated. Based on the relative enzyme activity, siderophore production, phosphate and potassium solubilization, strain CN12 was selected. Strain CN12 was identified as Bacillus amyloliquefaciens based on 16 S rRNA gene sequencing. The liquid culture medium using inexpensive substrates was optimized for increasing its endospore production. The medium consisted of 10% mung bean sprouts extract solution, 0.87% molasses, 0.12% urea, and 0.06% MgSO4 produced the spore yield of 5.53 ± 3.5 (× 108 CFU/mL) for strain CN12. Malabar spinach plants treated with strain CN12 showed significantly increases of 39.4 ± 8.9 to 77.6 ± 11.8 g in fresh weight, 17.1 ± 3.0 to 65.8 ± 17.2 cm in plant height, and 2.9 ± 0.9 to 6.8 ± 2.3 g in root weight compared to the control. These observations revealed that strain CN12 is a promising candidate for biofertilizer production.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Sherpa, M. T., Sharma, L., Bag, N. & Das, S. Isolation, characterization, and evaluation of native rhizobacterial consortia developed from the rhizosphere of rice grown in organic state Sikkim, India, and their effect on plant growth. Front. Microbiol. 12, 713660. https://doi.org/10.3389/fmicb.2021.713660 (2021).
Wang, X. et al. Isolation and characterization of antagonistic bacteria Paenibacillus jamilae HS-26 and their effects on plant growth. BioMed Res. Int. 2019, 3638926. https://doi.org/10.1155/2019/3638926 (2019).
Tuan, N. N., Lin, Y. W. & Huang, S. L. Catabolism of 4-alkylphenols by Acinetobacter sp. OP5: genetic organization of the oph gene cluster and characterization of alkylcatechol 2, 3-dioxygenase. Bioresour. Technol. 131, 420–428. https://doi.org/10.1016/j.biortech.2012.12.086 (2013).
Hengl, T. et al. Soil nutrient maps of Sub-Saharan Africa: assessment of soil nutrient content at 250 m spatial resolution using machine learning. Nutr. Cycl. Agroecosyst. 109, 77–102. https://doi.org/10.1007/s10705-017-9870-x (2017).
Kaur, R. & Kaur, S. Plant growth-promoting potential of ‘Myroides gitamensis’ isolated from virgin soils of Punjab. Arch. Microbiol. 203, 2551–2561. https://doi.org/10.1007/s00203-021-02231-8 (2021).
Roslan, M. A. M. et al. Seed biopriming with P- and K-solubilizing Enterobacter hormaechei sp. improves the early vegetative growth and the P and K uptake of okra (Abelmoschus esculentus) seedling. PloS one. 15, e0232860. https://doi.org/10.1371/journal.pone.0232860 (2020).
Zhang, H., Han, L., Jiang, B. & Long, C. Identification of a phosphorus-solubilizing Tsukamurella tyrosinosolvens strain and its effect on the bacterial diversity of the rhizosphere soil of peanuts growth-promoting. World J. Microbiol. Biotechnol. 37, 109. https://doi.org/10.1007/s11274-021-03078-3 (2021).
Zhao, D. et al. Isolation and genome sequence of a novel phosphate-solubilizing rhizobacterium Bacillus altitudinis GQYP101 and its effects on rhizosphere microbial community structure and functional traits of corn seedling. Curr. Microbiol. 79, 249. https://doi.org/10.1007/s00284-022-02944-z (2022).
Goswami, M. & Deka, S. Isolation of a novel rhizobacteria having multiple plant growth promoting traits and antifungal activity against certain phytopathogens. Microbiol. Res. 240, 126516. https://doi.org/10.1016/j.micres.2020.126516 (2020).
Vassilev, N. et al. Formulation of microbial inoculants by encapsulation in natural polysaccharides: focus on beneficial properties of carrier additives and derivatives. Front. Plant. Sci. 11, 270. https://doi.org/10.3389/fpls.2020.00270 (2020).
Li, Y., Liu, X., Hao, T. & Chen, S. Colonization and maize growth promotion induced by phosphate solubilizing bacterial isolates. Int. J. Mol. Sci. 18, 965. https://doi.org/10.3390/ijms18071253 (2017).
Muthukumarasamy, R., Revathi, G., Vadivelu, M. & Arun, K. Isolation of bacterial strains possessing nitrogen-fixation, phosphate and potassium-solubilization and their inoculation effects on sugarcane. Indian J. Exp. Biol. 55, 161–170 (2017).
Habibi, S. et al. Isolation and screening of indigenous plant growth-promoting rhizobacteria from different rice cultivars in Afghanistan soils. Microbes Environ. 34, 347–355. https://doi.org/10.1264/jsme2.ME18168 (2019).
Nguyen, N. T., Vo, V. T., Nguyen, T. H. P. & Kiefer, R. Isolation and optimization of a glyphosate-degrading Rhodococcus soli G41 for bioremediation. Arch. Microbiol. 204, 8596. https://doi.org/10.1007/s00203-022-02875-0 (2022).
Carrasco, M. et al. Screening and characterization of amylase and cellulase activities in psychrotolerant yeasts. BMC Microbiol. 16, 2542. https://doi.org/10.1186/s12866-016-0640-8 (2016).
Shrestha, S., Chio, C., Khatiwada, J. R., Kognou, A. L. M. & Qin, W. Optimization of multiple enzymes production by fermentation using lipid-producing Bacillus sp. Front. Microbiol. 13, 1049692. https://doi.org/10.3389/fmicb.2022.1049692 (2022).
Ren, H., Su, Y. T. & Guo, X. H. Rapid optimization of spore production from Bacillus amyloliquefaciens in submerged cultures based on dipicolinic acid fluorimetry assay. AMB Express. 8, 21. https://doi.org/10.1186/s13568-018-0555-x (2018).
Nierhaus, K. H. Mg2+, K+, and the ribosome. J. Bacteriol. 196, 3817–3819. https://doi.org/10.1128/JB.02297-14 (2014).
Chen, Z. M. et al. Greater enhancement of Bacillus subtilis spore yields in submerged cultures by optimization of medium composition through statistical experimental designs. Appl. Microbiol. Biotechnol. 85, 1353–1360. https://doi.org/10.1007/s00253-009-2162-x (2010).
Llado, S., Lopez-Mondejar, R. & Baldrian, P. F. Soil bacteria: diversity, involvement in ecosystem processes, and response to global change. Microbiol. Mol. Biol. Rev. 81, 2563. https://doi.org/10.1128/MMBR.00063-16 (2017).
Pham, V. H. & Le, V. C. The ecological interaction between endangered, precious and rare woody species in rich forest community of Tanphu protection forest, Vietnam. Biodiversitas 23, 6119–6127 (2022).
Cappuccino, J. G. & Welsh, C. Microbiology: a Laboratory Manual (Pearson, 2019).
Nguyen, N. T. et al. Identification and biodegradation potential of a novel strain of Kosakonia oryzae isolated from a polyoxyethylene tallow amine paddy soil. Curr. Microbiol. 78, 3173–3180. https://doi.org/10.1007/s00284-021-02592-9 (2021).
Nguyen, N. T. Acinetobacter soli SP2 capable of high-efficiency degradation of food emulsifier polysorbate 80. Curr. Microbiol. 75, 896–900. https://doi.org/10.1007/s00284-018-1463-7 (2018).
Lebrazi, S. et al. Screening and optimization of indole-3-acetic acid production and phosphate solubilization by rhizobacterial strains isolated from Acacia cyanophylla root nodules and their effects on its plant growth. J. Genetic Eng. Biotechnol. 18, 71. https://doi.org/10.1186/s43141-020-00090-2 (2020).
Salkowski, E. A microchemical method for the determination of indoleacetic acid. Plant Physiol. 32, 437–441 (1957).
Balla, A. et al. Screening of cellulolytic bacteria from various ecosystems and their cellulases production under multi-stress conditions. Catalysts 12, 2563 (2022).
Mohandas, A. et al. Production of Pectinase from Bacillus sonorensis MPTD1. Food Technol. Biotech. 56, 110–116. https://doi.org/10.17113/ftb.56.01.18.5477 (2018).
Pikovskaya, R. I. Mobilization of phosphorus in soil in connection with the vital activity of some microbial species. Mikrobiologiya 17, 362–370 (1948).
Nautiyal, C. S. An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol. Lett. 170, 265–270. https://doi.org/10.1111/j.1574-6968.1999.tb13383.x (1999).
Rajawat, M. V. S., Singh, S., Tyagi, S. P. & Saxena, A. K. A modified plate assay for rapid screening of potassium-solubilizing bacteria. Pedosphere 26, 768–773. https://doi.org/10.1016/S1002-0160(15)60080-7 (2016).
Hu, Q. P. & Xu, J. G. A simple double-layered chrome azurol S agar (SDCASA) plate assay to optimize the production of siderophores by a potential biocontrol agent Bacillus. Afr. J. Microbiol. Res. 5, 4321–4327 (2011).
Arora, N. K. & Verma, M. Modified microplate method for rapid and efficient estimation of siderophore produced by bacteria. 3 Biotech. 7, 381. https://doi.org/10.1007/s13205-017-1008-y (2017).
Ausubel, F. M. Short Protocols in Molecular Biology : A Compendium of Methods From Current Protocols in Molecular Biology (Wiley, 2002).
Hou, Q. et al. Design of primers for evaluation of lactic acid Bacteria populations in complex biological samples. Front. Microbiol. 9, 2045. https://doi.org/10.3389/fmicb.2018.02045 (2018).
Tian, Z. et al. Optimization of sporulation conditions for Bacillus subtilis BSNK-5. Processes 10, 1133 (2022).
Kumar, K., Amaresan, N., Bhagat, S., Madhuri, K. & Srivastava, R. C. Isolation and characterization of rhizobacteria associated with coastal agricultural ecosystem of rhizosphere soils of cultivated vegetable crops. World J. Microbiol. Biotechnol. 27, 1625–1632 (2011).
Wagi, S. & Ahmed, A. Bacillus spp.: potent microfactories of bacterial IAA. PeerJ 7, e7258. https://doi.org/10.7717/peerj.7258 (2019).
Khianngam, S., Meetum, P., Chiangmai, P. N. & Tanasupawat, S. Identification and optimisation of Indole-3-acetic acid production of endophytic bacteria and their effects on plant growth. Trop. life Sci. Res. 34, 219–239. https://doi.org/10.21315/tlsr2023.34.1.12 (2023).
Du, H. et al. Effects of Bacillus amyloliquefaciens TL106 isolated from Tibetan pigs on probiotic potential and intestinal microbes in weaned piglets. Microbiol. Spectr. 10, e0120521. https://doi.org/10.1128/spectrum.01205-21 (2022).
Mehta, N., Rao, P. & Saini, R. A review on metabolites and pharmaceutical potential of food legume crop mung bean (Vigna radiata L. Wilczek). BioTechnologia (Pozn). 102, 425–435. https://doi.org/10.5114/bta.2021.111107 (2021).
Shasaltaneh, M. D., Moosavi-Nejad, Z., Gharavi, S. & Fooladi, J. Cane molasses as a source of precursors in the bioproduction of tryptophan by Bacillus subtilis. Iran. J. Microbiol. 5, 285–292 (2013).
Wu, L. et al. Green synthesis of isomaltulose from cane molasses by Bacillus subtilis WB800-pHA01-palI in a biologic membrane reactor. Food Chem. 229, 761–768. https://doi.org/10.1016/j.foodchem.2017.03.001 (2017).
Bashan, Y. & de-Bashan, L. E. Fresh-weight measurements of roots provide inaccurate estimates of the effects of plant growth-promoting bacteria on root growth: a critical examination. Soil Biol. Biochem. 37, 1795–1804. https://doi.org/10.1016/j.soilbio.2005.02.013 (2005).
Reich, R. R. Bacillus stearothermophilus spore suspensions: effect of storage conditions and time on viability and moist heat resistance. J. Parenter. Sci. Technol. 35, 74–78 (1981).
Funding
This research was funded by PetroVietnam Fertilizer and Chemicals Corporation under grant number 138 − 24/PVFCCo/KHCN-KHVLUD/D_UreVSV.
Author information
Authors and Affiliations
Contributions
Tuan Ngoc Nguyen has designed the study. Tu Cam Ly, Nghi Tran, Nhung Hong Nguyen, Thu Thien Pham, Hien My Tran, Thuan Phuoc Dinh, Hung Phi Nguyen, Tuan Ngoc Nguyen performed the experiments. Tuan Ngoc Nguyen conducted the statistical analysis. Tuan Ngoc Nguyen and Rudolf Kiefer wrote the main manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nguyen, T.N., Ly, T.C., Tran, N. et al. Cost-effective plant-based medium for enhanced spore production of B. amyloliquefaciens CN12 for biofertilizer application. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42679-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42679-6