Abstract
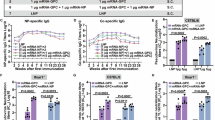
The Nairoviridae family of segmented negative-sense RNA viruses includes the serious human pathogen Crimean-Congo haemorrhagic fever virus (CCHFV), associated with a case/fatality rate of up to 40% for which no approved vaccines or treatments exist. Nairoviruses internalize via endocytosis and pass through the endolysosomal network, exploiting the changing ionic environment to promote envelope fusion. Fusion is influenced by hydrogen (H+) and potassium ions (K+), which increase in concentration as endosomes mature, regulated by host ion channels. Using the model nairovirus Hazara virus (HAZV) of the CCHFV serogroup, we performed an siRNA screen to identify cellular ion channels involved in nairovirus infection. Most high-ranking hits belonged to K+ and calcium (Ca2+) channel families. Consistent with this, we showed that clinically-approved K+ channel blockers quinidine, quinine and dronedarone and clinically-approved Ca2+ channel blockers tetrandrine and nifedipine significantly reduced HAZV activities. To further probe the role of K+ in HAZV infection, we used time-of-addition studies, showing K+ was required during entry. Biochemical experiments showed K+ expanded the pH range that promoted entry, potentially allowing endosome escape deeper within the endolysosomal network. These results show clinically-approved channel blockers effectively inhibit HAZV replication, suggesting repurposing existing therapies may represent promising avenues to block nairovirus infection.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
Surtees, R. et al. The crystal structure of the Hazara virus nucleocapsid protein. BMC Struct. Biol. 15, 24 (2015).
Frank, M. G. et al. Crimean-Congo Hemorrhagic Fever Virus for clinicians-Diagnosis, clinical management, and therapeutics. Emerg. Infect. Dis. 30, 864–873 (2024).
Hawman, D. W. & Feldmann, H. Crimean–Congo haemorrhagic fever virus. Nat. Rev. Microbiol. 21, 463–477 (2023).
D’Addiego, J. et al. Investigating the effect of ribavirin treatment on genetic mutations in Crimean-Congo haemorrhagic fever virus (CCHFV) through next-generation sequencing. J. Med. Virol. 95, e28548 (2023).
Hawman, D. W. et al. Favipiravir (T-705) but not ribavirin is effective against two distinct strains of Crimean-Congo hemorrhagic fever virus in mice. Antiviral Res. 157, 18–26 (2018).
Oestereich, L. et al. Evaluation of antiviral efficacy of ribavirin, arbidol, and T-705 (favipiravir) in a mouse model for Crimean-Congo hemorrhagic fever. PLoS Negl. Trop. Dis. 8, e2804 (2014).
Fels, J. M. et al. Protective neutralizing antibodies from human survivors of Crimean-Congo hemorrhagic fever. Cell 184, 3486-3501.e21 (2021).
Convalescent human plasma candidate reference materials protect against Crimean-Congo haemorrhagic fever virus (CCHFV) challenge in an A129 mouse model - ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0168170224001023.
Sorvillo, T. E. et al. Replicon particle vaccination induces non-neutralizing anti-nucleoprotein antibody-mediated control of Crimean-Congo hemorrhagic fever virus. Npj Vaccines 9, 88 (2024).
Leventhal, S. S. et al. Antibodies targeting the Crimean-Congo Hemorrhagic Fever Virus nucleoprotein protect via TRIM21. Nat. Commun. 15, 9236 (2024).
Dokuzoguz, B. et al. Severity scoring index for Crimean-Congo hemorrhagic fever and the impact of ribavirin and corticosteroids on fatality. Clin. Infect. Dis. 57, 1270–1274 (2013).
Punch, E. K. et al. Potassium is a trigger for conformational change in the fusion spike of an enveloped RNA virus. J. Biol. Chem. 293, 9937–9944 (2018).
Charlton, F. W. et al. Ion channels as therapeutic targets for viral infections: Further discoveries and future perspectives. Viruses 12, 844 (2020).
Scott, C. C. & Gruenberg, J. Ion flux and the function of endosomes and lysosomes: pH is just the start. BioEssays 33, 103–110 (2011).
Fuller, J. et al. Hazara nairovirus requires COPI components in both Arf1-dependent and Arf1-independent stages of its replication cycle. J. Virol. 94, e00766-e820 (2020).
Shaw, A. B. et al. Cellular endosomal potassium ion flux regulates arenavirus uncoating during virus entry. MBio 15, e01684 (2024).
Wang, B. et al. Dihydropyridine-derived calcium channel blocker as a promising anti-hantavirus entry inhibitor. Front. Pharmacol. 13, 940178 (2022).
Urata, S., Yoshikawa, R. & Yasuda, J. Calcium influx regulates the replication of several negative-strand RNA viruses including severe fever with thrombocytopenia syndrome virus. J. Virol. 97, e0001523 (2023).
Wang, G. & Lemos, J. R. Tetrandrine: A new ligand to block voltage-dependent Ca2+ and Ca(+)-activated K+ channels. Life Sci. 56, 295–306 (1995).
Dobson, S. J., Mankouri, J. & Whitehouse, A. Identification of potassium and calcium channel inhibitors as modulators of polyomavirus endosomal trafficking. Antiviral Res. https://doi.org/10.1016/j.antiviral.2020.104819 (2020).
Shekhar, S., Gupta, N., Kirubakaran, R. & Pareek, P. Oral nifedipine versus intravenous labetalol for severe hypertension during pregnancy: A systematic review and meta-analysis. BJOG Int. J. Obstet. Gynaecol. 123, 40–47 (2016).
Sherman, L. G. & Liang, C. S. Nifedipine in chronic stable angina: A double-blind placebo-controlled crossover trial. Am. J. Cardiol. 51, 706–711 (1983).
Patel, P. & Goyal, A. Antiarrhythmic Medications. in StatPearls (StatPearls Publishing, Treasure Island (FL), 2025).
Armstrong, C. M. Interaction of tetraethylammonium ion derivatives with the potassium channels of giant axons. J. Gen. Physiol. 58, 413–437 (1971).
Fatherazi, S. & Cook, D. L. Specificity of tetraethylammonium and quinine for three K channels in insulin-secreting cells. J. Membr. Biol. 120, 105–114 (1991).
Revenko, S. V., Khodorov, B. I. & Shapovalova, L. M. [Blockade of the sodium and potassium channels of a myelinated nerve fiber by quinidine]. Neirofiziologiia Neurophysiol. 14, 324–330 (1982).
Hover, S. et al. Modulation of potassium channels inhibits bunyavirus infection. J. Biol. Chem. 291, 3411–3422 (2016).
Liu, P. et al. A tris (2-carboxyethyl) phosphine (TCEP) related cleavage on cysteine-containing proteins. J. Am. Soc. Mass Spectrom. 21, 837–844 (2010).
Helenius, A., Marsh, M. & White, J. Inhibition of Semliki Forest Virus penetration by lysosomotropic weak bases. J. Gen. Virol. 58, 47–61 (1982).
Schmidt, C. et al. Novel electrophysiological properties of dronedarone: Inhibition of human cardiac two-pore-domain potassium (K2P) channels. Naunyn-Schmiedeberg’s Arch. Pharmacol. 385, 1003–1016 (2012).
Thomas, D. et al. Acute effects of dronedarone on both components of the cardiac delayed rectifier K+ current, HERG and KvLQT1/minK potassium channels. Br. J. Pharmacol. 140, 996 (2003).
Hover, S. et al. Bunyavirus requirement for endosomal K+ reveals new roles of cellular ion channels during infection. PLoS Pathog. 14, e1006845 (2018).
Hover, S. et al. Organisation of the orthobunyavirus tripodal spike and the structural changes induced by low pH and K+ during entry. Nat. Commun. 14, 5885 (2023).
Simon, M., Johansson, C. & Mirazimi, A. Crimean-Congo hemorrhagic fever virus entry and replication is clathrin-, pH- and cholesterol-dependent. J. Gen. Virol. 90, 210–215 (2009).
Jin, M. et al. Hantaan Virus enters cells by clathrin-dependent receptor-mediated endocytosis. Virology 294, 60–69 (2002).
Koch, J. et al. The Phenuivirus Toscana virus makes an atypical use of vacuolar acidity to enter host cells. PLoS Pathog. 19, e1011562 (2023).
Bowden, T. A. et al. Orthobunyavirus ultrastructure and the curious tripodal glycoprotein spike. PLoS Pathog. 9, e1003374 (2013).
Hover, S., Foster, B., Barr, J. N. & Mankouri, J. Viral dependence on cellular ion channels – An emerging anti-viral target?. J. Gen. Virol. 98, 345–351 (2017).
Russell, T., Gangotia, D. & Barry, G. Assessing the potential of repurposing ion channel inhibitors to treat emerging viral diseases and the role of this host factor in virus replication. Biomed. Pharmacother. Biomedecine Pharmacother. 156, 113850 (2022).
Vitali Serdoz, L., Rittger, H., Furlanello, F. & Bastian, D. Quinidine-A legacy within the modern era of antiarrhythmic therapy. Pharmacol. Res. 144, 257–263 (2019).
Fuller, J. et al. Rescue of infectious recombinant Hazara Nairovirus from cDNA reveals the nucleocapsid protein DQVD caspase cleavage motif performs an essential role other than cleavage. J. Virol. https://doi.org/10.1128/jvi.00616-19 (2019).
Schindelin, J. et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Acknowledgements
We acknowledge Wellcome trust equipment grant 221538/Z/20/Z, which supports the use of the IncuCyte live cell imaging platform.
Funding
This work was supported by a University of Leeds PhD studentship to FWC, MRC project Grant MR/T016159/1 to JNB, JF, JM and TAE, BBSRC project grant BB/V007467/1 to JNB, JM and HP, grant PID2023-149259NB-I00 funded by MICIU/AEI/ https://doi.org/10.13039/501100011033, a Human Frontiers Science Programme grant RGP0040/2019 awarded to JF, and by “ERDF A way of making Europe” to JF.
Author information
Authors and Affiliations
Contributions
FWC, HMP, SEH and AA acquired the data. FWC, MS, JF and JNB analysed and interpreted the data. TAE and JM made contributions to the conception and design of the work. FWC, JF and JNB wrote the manuscript. FWC made the figures. All authors approved the submitted version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Charlton, F.W., Hover, S.E., Alyahyawi, A. et al. Identification of cellular ion channels that facilitate Hazara nairovirus infection enables selection of clinically approved compounds with anti-nairoviral properties. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42810-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42810-7