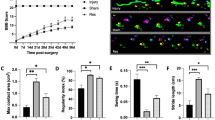
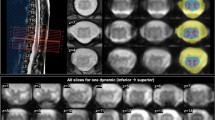
Abstract
A cascade of biological responses to spinal cord injury (SCI), including neuroinflammation, plays a pivotal role in determining long-term outcomes and has become a primary therapeutic target. Riluzole, a neuroprotective agent, has demonstrated efficacy in preserving tissue integrity and improving motor function following SCI. The study aims to use this established treatment to verify that resting-state fMRI (rsfMRI) functional connectivity (rsFC) and TSPO PET metrics are reliable biomarkers of SCI severity, progression, and treatment response. 16 male rats with a moderate lumbar contusion injury were administered Riluzole or HBC vehicle. rsfMRI and TSPO PET scans were collected post-SCI alongside motor-sensory behavioral tests. After SCI, significantly stronger rsFC between dorsal-to-dorsal gray matter horns rostral to the SCI was observed in the riluzole group, compared to the vehicle group. A majority of horn pairs rostral and caudal to injury exhibited significant decrease in rsFC over time for both groups and correlated with post-injury behavioral deficits and recovery. TSPO-PET detected increased SCI neuroinflammatory activity. Our results demonstrate reductions in rsFC disruption, validating the role of rsFC as biomarkers of SCI severity and progression. The imaging biomarkers can be used to evaluate the responsiveness to treatment and efficacy of novel therapies in preclinical studies.
Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Alizadeh, A., Dyck, S. M. & Karimi-Abdolrezaee, S. Traumatic spinal cord injury: An overview of pathophysiology, models and acute injury mechanisms. Front. Neurol. 10, 282 (2019).
Stammers, A. T., Liu, J. & Kwon, B. K. Expression of inflammatory cytokines following acute spinal cord injury in a rodent model. J. Neurosci. Res. 90, 782–790 (2012).
Dickens, A. M. et al. Detection of microglial activation in an acute model of neuroinflammation using PET and radiotracers 11C-(R)-PK11195 and 18F-GE-180. J. Nucl. Med. 55, 466–472 (2014).
Park, E., Velumian, A. A. & Fehlings, M. G. The role of excitotoxicity in secondary mechanisms of spinal cord injury: A review with an emphasis on the implications for white matter degeneration. J. Neurotrauma 21, 754–774 (2004).
Tator, C. H. & Fehlings, M. G. Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J. Neurosurg. 75, 15–26 (1991).
Donnelly, D. J. & Popovich, P. G. Inflammation and its role in neuroprotection, axonal regeneration and functional recovery after spinal cord injury. Exp. Neurol. 209, 378–388 (2008).
Blesch, A. & Tuszynski, M. H. Spinal cord injury: Plasticity, regeneration and the challenge of translational drug development. Trends Neurosci. 32, 41–47 (2009).
Raineteau, O. & Schwab, M. E. Plasticity of motor systems after incomplete spinal cord injury. Nat. Rev. Neurosci. 2, 263–273 (2001).
Curt, A., Van Hedel, H. J. A., Klaus, D. & Dietz, V. Recovery from a spinal cord injury: Significance of compensation, neural plasticity, and repair. J. Neurotrauma 25, 677–685 (2008).
Onifer, S. M., Smith, G. M. & Fouad, K. Plasticity after spinal cord injury: Relevance to recovery and approaches to facilitate it. Neurotherapeutics 8, 283–293 (2011).
Wu, Y. et al. Delayed post-injury administration of riluzole is neuroprotective in a preclinical rodent model of cervical spinal cord injury. J. Neurotrauma 30, 441–452 (2013).
Nagoshi, N., Nakashima, H. & Fehlings, M. Riluzole as a neuroprotective drug for spinal cord injury: From bench to bedside. Molecules 20, 7775–7789 (2015).
Turkheimer, F. E. et al. The methodology of TSPO imaging with positron emission tomography. Biochem. Soc. Trans. 43, 586–592 (2015).
Narayanaswami, V. et al. Emerging PET radiotracers and targets for imaging of neuroinflammation in neurodegenerative diseases: Outlook beyond TSPO. Mol. imaging 17, 1–25. https://doi.org/10.1177/1536012118792317 (2018).
Liu, B. et al. In vivo detection of age- and disease-related increases in neuroinflammation by 18F-GE180 TSPO MicroPET imaging in wild-type and Alzheimer’s transgenic mice. J. Neurosci. 35, 15716–15730 (2015).
Gargiulo, S. et al. Imaging of brain TSPO expression in a mouse model of amyotrophic lateral sclerosis with 18F-DPA-714 and micro-PET/CT. Eur. J. Nucl. Med. Mol. Imaging 43, 1348–1359 (2016).
Lucot, K. L. et al. Tracking innate immune activation in a mouse model of Parkinson’s disease using TREM1 and TSPO PET tracers. J. Nucl. Med. 63, 1570–1578 (2022).
Tremoleda, J. L. et al. In vivo PET imaging of the neuroinflammatory response in rat spinal cord injury using the TSPO tracer [18F]GE-180 and effect of docosahexaenoic acid. Eur. J. Nucl. Med. Mol. Imaging 43, 1710–1722 (2016).
Mu, C. et al. Spatiotemporal dynamics of neuroinflammation relate to behavioral recovery in rats with spinal cord injury. Mol. Imaging Biol. 26, 240–252 (2023).
Schölvinck, M. L., Maier, A., Ye, F. Q., Duyn, J. H. & Leopold, D. A. Neural basis of global resting-state fMRI activity. Proc. Natl. Acad. Sci. U. S. A. 107, 10238–10243 (2010).
van den Heuvel, M. P. & Hulshoff Pol, H. E. Exploring the brain network: A review on resting-state fMRI functional connectivity. Eur. Neuropsychopharmacol. 20, 519–534 (2010).
Stroman, P. W. et al. The current state-of-the-art of spinal cord imaging: Methods. Neuroimage 84, 1070–1081 (2014).
Bican, O., Minagar, A. & Pruitt, A. A. The spinal cord. A review of functional neuroanatomy. Neurol. Clin. 31, 1–18 (2013).
Figley, C. R. & Stroman, P. W. Investigation of human cervical and upper thoracic spinal cord motion: Implications for imaging spinal cord structure and function. Magn. Reson. Med. 58, 185–189 (2007).
Ford, J. C. et al. A method for in vivo high resolution MRI of rat spinal cord injury. Magn. Reson. Med. 31, 218–223 (1994).
Pierre-Jerome, C., Arslan, A. & Bekkelund, S. I. MRI of the spine and spinal cord: Imaging techniques, normal anatomy, artifacts, and pitfalls. J. Manipulative Physiol. Ther. 23, 470–475 (2000).
Wu, T. L. et al. Resting-state functional connectivity in the rat cervical spinal cord at 9.4 T. Magn. Reson. Med. 79, 2773–2783 (2018).
Conrad, B. N. et al. Multiple Sclerosis lesions affect intrinsic functional connectivity of the spinal cord. Brain 141, 1650–1664 (2018).
Chen, L. M., Mishra, A., Yang, P. F., Wang, F. & Gore, J. C. Injury alters intrinsic functional connectivity within the primate spinal cord. Proc. Natl. Acad. Sci. U. S. A. 112, 5991–5996 (2015).
Wu, T. L. et al. Longitudinal assessment of recovery after spinal cord injury with behavioral measures and diffusion, quantitative magnetization transfer and functional magnetic resonance imaging. NMR Biomed. 33, e4216 (2020).
Barry, R. L., Smith, S. A., Dula, A. N. & Gore, J. C. Resting state functional connectivity in the human spinal cord. Elife 2014, 1–15 (2014).
Wei, P. et al. Resting state networks in human cervical spinal cord observed with fMRI. Eur. J. Appl. Physiol. 108, 265–271 (2010).
Kjell, J. & Olson, L. Rat models of spinal cord injury: From pathology to potential therapies. DMM Dis. Models Mech. 9, 1125–1137 (2016).
Cheriyan, T. et al. Spinal cord injury models: A review. Spinal Cord 52, 588–595 (2014).
Mu, C. et al. Validation of qMT and CEST MRI as biomarkers of response to treatment after lumbar spinal cord injury in rats. NMR Biomed. 38, e70015 (2025).
Brooks, J. C. W., Büchel, C., Winkler, A. M., Andersson, J. L. & Tracey, I. Investigating resting-state functional connectivity in the cervical spinal cord at 3 T. Neuroimage 147, 589–601 (2017).
Werry, E. L. et al. Recent developments in TSPO PET imaging as a biomarker of neuroinflammation in neurodegenerative disorders. Int. J. Mol. Sci. 20, 3161 (2019).
Badhiwala, J. H., Ahuja, C. S. & Fehlings, M. G. Time is spine: A review of translational advances in spinal cord injury. J. Neurosurg. Spine 30, 1–18 (2019).
Lu, M. et al. Design and construction of an interchangeable RF coil system for rodent spinal cord MR imaging at 9.4 T. Magn. Reson. Imaging 84, 124–131 (2021).
Zou, Q. H. et al. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: Fractional ALFF. J. Neurosci. Methods 172, 137–141 (2008).
Cheung, Y. Y., Nickels, M. L., Tang, D., Buck, J. R. & Manning, H. C. Facile synthesis of SSR180575 and discovery of 7-chloro-N,N,5-trimethyl-4-oxo-3(6-[18F]fluoropyridin-2-yl)-3,5-dihydro-4H-pyridazino[4,5-b]indole-1-acetamide, a potent pyridazinoindole ligand for PET imaging of TSPO in cancer. Bioorg. Med. Chem. Lett. 24, 4466–4471 (2014).
Basso, D. M., Beattie, M. S. & Bresnahan, J. C. A sensitive and reliable locomotor rating scale for open field testing in rats. J. Neurotrauma 12, 1–21 (1995).
Walsh, R. N. & Cummins, R. A. The open-field test: A critical review. Psychol. Bull. 83, 482–504 (1976).
Deuis, J. R., Dvorakova, L. S. & Vetter, I. Methods used to evaluate pain behaviors in rodents. Front. Mol. Neurosci. 10, 284 (2017).
Ahmed, Z. et al. Actin-binding proteins coronin-1a and IBA-1 are effective microglial markers for immunohistochemistry. J. Histochem. Cytochem. 55, 687–700 (2007).
Eng, L. F. Glial fibrillary acidic protein (GFAP): The major protein of glial intermediate filaments in differentiated astrocytes. J. Neuroimmunol. 8, 203–214 (1985).
Bankhead, P. et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 7, 1–7 (2017).
Acknowledgements
The authors gratefully acknowledge Zou Yue and Chaohui Tang for their assistance with surgical procedures and perfusion of animals, the Center for Small Animal Imaging (CSAI) staff for imaging support, and the Vanderbilt University Medical Center (VUMC) Translational Pathology Shared Resource Core (NCI/NIH Cancer Center Support Grant 2P30 CA068485-14) for their expertise with processing the spinal cord tissue histology.
Funding
This study is supported by DOD Grant SC190134 (awarded to LMC), NIH Grant F31 NS113949 (awarded to CM), and acknowledges the support also of NIH award 1S10OD025085 (awarded to JCG). We acknowledge additional NIH support for instrumentation grants 1S10OD016245-01 and 1S10RR023784-01.
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to the work. CM, LMC, and JCG designed the experimental studies and procedures. XY and ML designed and tested the MRI coil hardware used in the study. CM and JLR performed surgical procedures, animal care, and collected behavior data. CM and FW collected MR data and optimized MR sequences. MNT collected and processed PET imaging data, with the assistance of CM. CM, JLR, and LMC performed perfusion and spinal cord tissue extraction. CM analyzed the MRI and behavioral data with the assistance of FW. and J.L.R. CM and JLR performed histology image quantification. CM drafted the manuscript and prepared the figures. All authors provided feedback and revisions for the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mu, C., Reed, J.L., Wang, F. et al. Anti-inflammatory treatment confirms rsfMRI and TSPO PET as biomarkers of functional connectivity and neuroinflammation in rat contusion spinal cord injuries. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42844-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42844-x