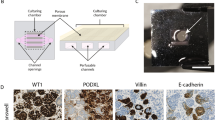
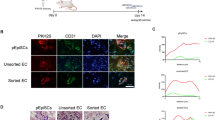
Abstract
Blood vessel organoids (BVOs) represent a promising tool for modeling vascular diseases, drug screening, and regenerative therapies. However, current protocols for BVO generation are complex, labor-intensive, and reliant on animal-derived extracellular matrices (ECM) such as Matrigel, limiting reproducibility, scalability, and clinical applicability. We developed a simplified, animal-origin-free protocol for BVO generation that addresses current limitations and enables high-throughput automated workflows. The method employs ultra-low attachment 96-well U-bottom plates for standardized aggregation and differentiation of human induced pluripotent stem cells (hiPSCs) in a human derived collagen-based extracellular matrix. Unlike conventional protocols where aggregates are embedded in a two-layer ECM, our approach utilizes a single-layer, which we termed “sitting drop”. This innovative approach requires considerably fewer materials and handling steps and is compatible with high-throughput automated machines. BVO generation utilizing the here described optimized protocol resulted in the formation of BVOs with reproducible morphology and cellular composition. Flow cytometry confirmed the presence of CD31⁺ endothelial cells and PDGFRβ⁺ pericytes in BVOs, generated in sitting drops in ultra-low adhesive U-bottom shaped 96 well plates, with cell population percentages comparable to those observed in traditional two-layer BVO cultures. In vivo transplantation of mature BVOs in a mouse full-thickness skin wound model demonstrated integration of BVO derived cells into host vessels, highlighting their potential in cell-based therapies. Our study presents a robust and animal-origin-free method for BVO generation based on single-layer “sitting drop” cultures. This protocol maintains cellular integrity while enhancing reproducibility and automation-readiness, paving the way for high-throughput screening and clinical translation of vascular organoid technology.
Similar content being viewed by others
Data availability
The data, presented in this study, are available from the corresponding author on reasonable request.
References
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459(7244), 262–5 (2009).
Huch, M. et al. In vitro expansion of single Lgr5 + liver stem cells induced by Wnt-driven regeneration. Nature 494(7436), 247–50 (2013).
Sachs, N. et al. Long-term expanding human airway organoids for disease modeling. EMBO J. https://doi.org/10.15252/embj.2018100300 (2019).
Boj, S. F. et al. Organoid models of human and mouse ductal pancreatic cancer. Cell 160 (1–2), 324–338 (2015).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501(7467), 373–9 (2013).
Wimmer, R. A., Leopoldi, A., Aichinger, M., Kerjaschki, D. & Penninger, J. M. Generation of blood vessel organoids from human pluripotent stem cells. Nat. Protoc. 14(11), 3082–100 (2019).
Wimmer, R. A. et al. Human blood vessel organoids as a model of diabetic vasculopathy. Nature 565(7740), 505–10 (2019).
Kong, D. et al. In vitro modeling of atherosclerosis using iPSC-derived blood vessel organoids. Adv. Healthc. Mater. 14(1), e2400919 (2025).
Aisenbrey, E. A. & Murphy, W. L. Synthetic alternatives to Matrigel. Nat. Rev. Mater. 5(7), 539–51 (2020).
Li, K., He, Y., Jin, X., Jin, K. & Qian, J. Reproducible extracellular matrices for tumor organoid culture: Challenges and opportunities. J. Transl. Med. 23(1), 497. (2025).
Kozlowski, M. T., Crook, C. J. & Ku, H. T. Towards organoid culture without Matrigel. Commun. Biol. 4(1), 1387. (2021).
Brassard, J. A. & Lutolf, M. P. Engineering stem cell self-organization to build better organoids. Cell Stem Cell 24(6), 860–76 (2019).
Schutgens, F. & Clevers, H. Human organoids: Tools for understanding biology and treating diseases. Annu. Rev. Pathol. 15, 211–234 (2020).
Soto-Gamez, A., Gunawan, J. P., Barazzuol, L., Pringle, S. & Coppes, R. P. Organoid-based personalized medicine: From tumor outcome prediction to autologous transplantation. Stem Cells 42(6), 499–508 (2024).
Naderi-Meshkin, H. et al. Unveiling impaired vascular function and cellular heterogeneity in diabetic donor-derived vascular organoids. Stem Cells 42(9), 791–808 (2024).
Ahn, Y. et al. Blood vessel organoids generated by base editing and harboring single nucleotide variation in Notch3 effectively recapitulate CADASIL-related pathogenesis. Mol. Neurobiol. 61(11), 9171–83 (2024).
Gjorevski, N. et al. Designer matrices for intestinal stem cell and organoid culture. Nature 539(7630), 560–4 (2016).
Giles, R. et al. Animal-free alternatives for Matrigel in human iPSC-derived blood vessel organoid culture. Sci. Rep. 15(1), 36042 (2025).
Brandenberg, N. et al. High-throughput automated organoid culture via stem-cell aggregation in microcavity arrays. Nat. Biomed. Eng. 4 (9), 863–874 (2020).
Quintard, C. et al. A microfluidic platform integrating functional vascularized organoids-on-chip. Nat. Commun. 15 (1), 1452 (2024).
Tubbs, E. et al. Human assembloid of human blood vessel organoids with pancreatic islets improves insulin secretion over time ex vivo. Cell Rep. 44(10), 116378 (2025).
Heo, J. H., Kang, D., Seo, S. J. & Jin, Y. Engineering the extracellular matrix for organoid culture. Int. J. Stem Cells 15(1), 60–9 (2022).
Moss, S. P., Bakirci, E. & Feinberg, A. W. Engineering the 3D structure of organoids. Stem Cell Reports 20(1), 102379 (2025).
Nwokoye, P. N. & Abilez, O. J. Bioengineering methods for vascularizing organoids. Cell Rep. Methods 4(6), 100779 (2024).
Acknowledgements
The authors would like to thank Sophia Wedel and Maria Dumitrascuta for their excellent preliminary work that formed the basis of this manuscript. We further acknowledge the Tyrolean Cancer Research Institute (TKFI) for their generous support and for providing access to their facilities and equipment.
Funding
Standortagentur Tirol, Health Hub grant, TAM: TM-12514133. Austrian Research Promotion Agency (FFG), Project Number: FO999925858. European HORIZON, Grant agreement ID: 101135053.
Author information
Authors and Affiliations
Contributions
A.H as first author designed and performed experiments, carried out the statistical analyses and wrote the manuscript. D.S. and J.T. designed and performed experiments and contributed to the writing of the manuscript. Y.H.T. performed image analysis. J.F.B. contributed to the mouse experiment. T.E.Y. initiated and supervised the project, designed experiments and wrote the manuscript as corresponding author.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hoffmann, A., Schorn, D., Thönig, J. et al. Animal-origin-free method for generating blood vessel organoids. Sci Rep (2026). https://doi.org/10.1038/s41598-026-42977-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-42977-z