Abstract
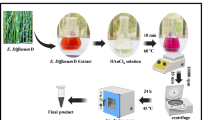
Cataract represents one of the leading causes of blindness worldwide and is primarily attributed to protein glycation, oxidative stress, and aggregation of lens crystallins. Surgical lens extraction remains the standard treatment, which, despite its effectiveness, carries potential postoperative risks and economic burdens, thereby underscoring the need for alternative, non-surgical therapeutic approaches. This study aimed to develop a green-synthesized nanosystem based on gold nanoparticles (AuNPs) using Alchemilla vulgaris L. (Lady’s mantle) extract, aiming to achieve efficient antioxidant delivery and sustained therapeutic retention within ocular tissues. Gold nanoparticles were synthesized via the reduction of chloroauric acid (HAuCl·3H2O) using the plant extract under ultrasonic agitation and dispersion, yielding AuNPs formulations 1%, 4%, and 7% (w/v). The synthesized nanocomposites were characterized using a UV–Vis spectroscopy, FTIR, XRD, FE-SEM, AFM, and zeta potential analysis. Bio activity was performed using DPPH radical scavenging, MTT assays for cytotoxicity testing, in vitro drug-release studies, and ex-vivo lens transparency assessments, all supported by computational molecular docking and ADMET (absorption, distribution, metabolism, and excretion) analyses. The results demonstrated that NAC-loaded gold nanoparticles (NAC–AuNPs) exhibited enhanced antioxidant activity, achieving 72–89% DPPH inhibition, and a high encapsulation efficiency of 86.1%. The drug-release profile exhibited a higher release rate observed under mildly acidic conditions (pH 6). Ex-vivo experiments using human cataractous lenses revealed a dose-dependent improvement in lens optical clarity at NAC concentrations of 0.05, 0.1, and 0.3 mM, loaded onto 7% (w/v) AuNPs formulations. Molecular docking studies suggested potential hydrogen-bond interactions between NAC and key lens crystallin proteins, providing mechanistic insight into possible structural stabilization of lens crystallin proteins. The green-synthesized AuNP–NAC nanosystem may represents a promising and biocompatible nanocarrier strategy that warrants further in vivo validation.
Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information file.
References
Chen, S. P., Woreta, F. & Chang, D. F. Cataracts: A review. JAMA 333, 2093–2103 (2025).
Study VLEGotGBoD. Global estimates on the number of people blind or visually impaired by cataract: A meta-analysis from 2000 to 2020. Eye 38(11), 2156 (2024).
Loh, D. & Reiter, R. J. β/γ-Crystallins, redox chemistry, and melatonin: The regulation of phase separation in cataractogenesis (2025).
Zhang, M. et al. The role of oxidative stress in the pathogenesis of ocular diseases: An overview. Mol. Biol. Rep. 51(1), 454 (2024).
Nadeem, S. Cataract Surgery: Historical Devices, Modern Innovations, and Future Perspectives 991–994 (Taylor & Francis, 2024).
Durgapal, S. et al. Cataract management in the modern era: Therapeutic advances and unmet needs. Curr. Pharm. Des. 32, 869–883 (2025).
Díez-Pascual, A. M. Surface engineering of nanomaterials with polymers, biomolecules, and small ligands for nanomedicine. Materials 15(9), 3251 (2022).
Nasraty, N. Investigating the Role of The Antioxidant N-Acetylcarnosine in Attenuating Oxidation-induced Retinal Damage in Retinitis Pigmentosa (Harvard University, 2021).
Dammak, A. et al. Oxidative stress in the anterior ocular diseases: Diagnostic and treatment. Biomedicines 11(2), 292 (2023).
Wang, Y., Cao, K., Guo, Z.-X. & Wan, X.-H. Effect of lens crystallins aggregation on cataract formation. Exp. Eye Res. https://doi.org/10.1016/j.exer.2025.110288 (2025).
Butt, A. & Bach, H. Advancements in nanotechnology for diagnostics: A literature review, Part II: Advanced techniques in nuclear and optical imaging. Nanomedicine 20(2), 183–206 (2025).
Gupta, P. C., Sharma, N., Mishra, P., Rai, S. & Verma, T. Role of gold nanoparticles for targeted drug delivery. In Metal and Metal–Oxide Based Nanomaterials: Synthesis, Agricultural, Biomedical and Environmental Interventions 243–269 (Springer, 2024).
Hang, Y., Wang, A. & Wu, N. Plasmonic silver and gold nanoparticles: Shape-and structure-modulated plasmonic functionality for point-of-caring sensing, bio-imaging and medical therapy. Chem. Soc. Rev. 53(6), 2932–2971 (2024).
Babizhayev, M. A. Ocular drug metabolism of the bioactivating antioxidant N-acetylcarnosine for vision in ophthalmic prodrug and codrug design and delivery. Drug Dev. Ind. Pharm. 34(10), 1071–1089 (2008).
Georgeous, J., AlSawaftah, N., Abuwatfa, W. H. & Husseini, G. A. Review of gold nanoparticles: Synthesis, properties, shapes, cellular uptake, targeting, release mechanisms and applications in drug delivery and therapy. Pharmaceutics 16(10), 1332 (2024).
Gul, M., Kashif, M., Muhammad, S., Azizi, S. & Sun, H. Various methods of synthesis and applications of gold-based nanomaterials: A detailed review. Cryst. Growth Des. 25(7), 2227–2266 (2025).
Rosero, W. A. A., Barbezan, A. B., de Souza, C. D. & Rostelato, M. E. C. M. Review of advances in coating and functionalization of gold nanoparticles: From theory to biomedical application. Pharmaceutics 16(2), 255 (2024).
Zhang, W. et al. Nanoscale bioconjugates: A review of the structural attributes of drug-loaded nanocarrier conjugates for selective cancer therapy. Heliyon https://doi.org/10.1016/j.heliyon.2022.e09577 (2022).
Giri, B. R., Jakka, D., Sandoval, M. A., Kulkarni, V. R. & Bao, Q. Advancements in ocular therapy: A review of emerging drug delivery approaches and pharmaceutical technologies. Pharmaceutics 16(10), 1325 (2024).
Lanier, O. L. et al. Review of approaches for increasing ophthalmic bioavailability for eye drop formulations. AAPS PharmSciTech 22(3), 107 (2021).
Račić, A. & Krajišnik, D. Biopolymers in mucoadhesive eye drops for treatment of dry eye and allergic conditions: Application and perspectives. Pharmaceutics 15(2), 470 (2023).
Gabai, A., Zeppieri, M., Finocchio, L. & Salati, C. Innovative strategies for drug delivery to the ocular posterior segment. Pharmaceutics 15(7), 1862 (2023).
Rykowska, I., Nowak, I. & Nowak, R. Soft contact lenses as drug delivery systems: A review. Molecules 26(18), 5577 (2021).
Liu, Y. & Wu, N. Progress of nanotechnology in diabetic retinopathy treatment. Int. J. Nanomed. https://doi.org/10.2147/IJN.S294807 (2021).
Ko, W.-C. et al. Pharmacological role of functionalized gold nanoparticles in disease applications. Molecules 27(5), 1551 (2022).
Watchorn, J. et al. Untangling mucosal drug delivery: Engineering, designing, and testing nanoparticles to overcome the mucus barrier. ACS Biomater. Sci. Eng. 8(4), 1396–1426 (2022).
Budnar, P., Tangirala, R., Bakthisaran, R. & Rao, C. M. Protein aggregation and cataract: Role of age-related modifications and mutations in α-crystallins. Biochem. Mosc. 87(3), 225–241 (2022).
Anwar, S. et al. A review on mechanism of inhibition of advanced glycation end products formation by plant derived polyphenolic compounds. Mol. Biol. Rep. 48(1), 787–805 (2021).
Rodella, U., Honisch, C., Gatto, C., Ruzza, P. & D’Amato, T. J. Antioxidant nutraceutical strategies in the prevention of oxidative stress related eye diseases. Nutrients 15(10), 2283 (2023).
Li, S., Chen, L. & Fu, Y. Nanotechnology-based ocular drug delivery systems: recent advances and future prospects. J. Nanobiotechnol. 21(1), 232 (2023).
Santos, G., Delgado, E., Silva, B., Braz, B. S. & Gonçalves, L. Topical ocular drug delivery: The impact of permeation enhancers. Pharmaceutics 17(4), 447 (2025).
Raîche-Marcoux, G. et al. Localization of fluorescent gold nanoparticles throughout the eye after topical administration. Front. Med. 12, 1557611 (2025).
Rai, A. & Ferreira, L. Biomedical applications of the peptide decorated gold nanoparticles. Crit. Rev. Biotechnol. 41(2), 186–215 (2021).
Chen, Y., Ye, Z., Chen, H. & Li, Z. Breaking barriers: Nanomedicine-based drug delivery for cataract treatment. Int. J. Nanomed. 19, 4021–4040 (2024).
Qi, M. & Tay, A. Phage–nanomaterial platforms for precision antimicrobial therapy: From design to therapeutic application. Nanoscale https://doi.org/10.1039/d5nr02249e (2025).
Bilušić, T., Šola, I. & Čikeš Čulić, V. Identification of flavonoids, antioxidant and antiproliferative activity of aqueous infusions of Calendula officinalis L., Chelidonium majus L., Teucrium chamaedrys L. and Alchemilla vulgaris L.. Food Technol. Biotechnol. 62(1), 49–58 (2024).
Abdulsada, T. N., Rheima, A. M. & Al-Rubae’i, S. H. Sustainable new biosynthesis of zero valence gold nanoparticles via healthy Alchemilla vulgaris L. extract as capping agent. Case Stud. Chem. Environ. Eng. 10, 100888 (2024).
Parramon, J. S., Švarc, T., Majerič, P., Jelen, Ž & Rudolf, R. Optical characteristics of directly deposited gold nanoparticle films. Surfaces 7(2), 369–379 (2024).
Mradu, G., Saumyakanti, S., Sohini, M. & Arup, M. HPLC profiles of standard phenolic compounds present in medicinal plants. Int. J. Pharmacogn. Phytochem. Res. 4(3), 162–167 (2012).
Pasieczna-Patkowska, S., Cichy, M. & Flieger, J. Application of Fourier transform infrared (FTIR) spectroscopy in characterization of green synthesized nanoparticles. Molecules 30(3), 684 (2025).
Pechyen, C., Ponsanti, K., Tangnorawich, B. & Ngernyuang, N. Biogenic synthesis of gold nanoparticles mediated by Spondias dulcis (Anacardiaceae) peel extract and its cytotoxic activity in human breast cancer cell. Toxicol. Rep. 9, 1092–1098 (2022).
Thongwattana, T. et al. Synthesis of silver nanoparticles and gold nanoparticles used as biosensors for the detection of human serum albumin-diagnosed kidney disease. Pharmaceuticals. 17(11), 1421 (2024).
Farkas, S. et al. Reaction–diffusion assisted synthesis of gold nanoparticles: Route from the spherical nano-sized particles to micrometer-sized plates. J. Phys. Chem. C 125(47), 26116–26124 (2021).
Gorohovs, M. & Dekhtyar, Y. Surface functionalization of nanoparticles for enhanced electrostatic adsorption of biomolecules. Molecules 30(15), 3206 (2025).
Lopes, J. et al. Safety of gold nanoparticles: From in vitro to in vivo testing array checklist. Pharmaceutics 15, 1120 (2023).
Raiche-Marcoux, G., Loiseau, A., Maranda, C., Poliquin, A. & Boisselier, E. Parametric drug release optimization of anti-inflammatory drugs by gold nanoparticles for topically applied ocular therapy. Int. J. Mol. Sci. 23(24), 16191 (2022).
Wang, J. et al. Multi-environment and multi-parameter screening of stability and coating efficiency of gold nanoparticle bioconjugates in application media. Sci. Rep. 14(1), 31568 (2024).
de Diego-García, L., Rejas-González, R., Latre, I. C. & Guzman-Aranguez, A. Pharmacological strategies for cataract management: From molecular targets to clinical translation. Int. J. Mol. Sci. 26(12), 5658 (2025).
Kim, H.-M., Park, J. H., Choi, Y. J., Oh, J.-M. & Park, J. Hyaluronic acid-coated gold nanoparticles as a controlled drug delivery system for poorly water-soluble drugs. RSC Adv. 13(8), 5529–5537 (2023).
Seegobin, N. et al. Dual action tofacitinib-loaded PLGA nanoparticles alleviate colitis in an IBD mouse model. Drug Deliv. Transl. Res. 15(7), 2372–2389 (2025).
Budama-Kilinc, Y. et al. Novel NAC-loaded poly (lactide-co-glycolide acid) nanoparticles for cataract treatment: Preparation, characterization, evaluation of structure, cytotoxicity, and molecular docking studies. PeerJ 6, e4270 (2018).
Sunseri, J. & Koes, D. R. Pharmit: Interactive exploration of chemical space. Nucleic Acids Res. 44(W1), W442–W448 (2016).
Daina, A., Michielin, O. & Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 7(1), 42717 (2017).
Banerjee, P., Kemmler, E., Dunkel, M. & Preissner, R. ProTox 3.0: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 52(W1), W513–W520 (2024).
Trott, O. & Olson, A. J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31(2), 455–461 (2010).
Du, J., Singh, H., Dong, W., Bai, Y. & Yi, T.-H. Colorimetric detection of Listeria monocytogenes using one-pot biosynthesized flower-shaped gold nanoparticles. Sens. Actuators B Chem. 265, 285–292 (2018).
Bharadwaj, K. K. et al. Green synthesis of gold nanoparticles using plant extracts as beneficial prospect for cancer theranostics. Molecules 26(21), 6389 (2021).
Fathalla, Z. et al. Hybrid thermosensitive-mucoadhesive in situ forming gels for enhanced corneal wound healing effect of l-carnosine. Drug Deliv. 29(1), 374–385 (2022).
Wang, Y., Xia, R., Hu, H. & Peng, T. Biosynthesis, characterization and cytotoxicity of gold nanoparticles and their loading with N-acetylcarnosine for cataract treatment. J. Photochem. Photobiol. B 187, 180–183 (2018).
Botteon, C. E. A. et al. Biosynthesis and characterization of gold nanoparticles using Brazilian red propolis and evaluation of its antimicrobial and anticancer activities. Sci. Rep. 11(1), 1974 (2021).
Wang, L., Liu, W. & Huang, X. An approach to revolutionize cataract treatment by enhancing drug probing through intraocular cell line. Libyan J. Med. https://doi.org/10.1080/19932820.2018.1500347 (2018).
Muddapur, U. M. et al. Plant-based synthesis of gold nanoparticles and theranostic applications: A review. Molecules 27(4), 1391 (2022).
Sharma, R., Yadav, A., Arora, S. & Kishore, N. To study the structural properties of cobalt doped tin oxide nanostructured by using Williamson–Hall and size-strain plot methodology. Mater. Today Proc. 44, 4651–4656 (2021).
Adnan R. Gold-based Nanomaterials: Spectroscopy, Microscopy and Applications in Catalysis and Sensing (2015).
Lin, Z. et al. Biosynthesis, optimization, and multifunctional biomedical applications of gold nanoparticles mediated by Streptomyces sp. YJD18. Front. Microbiol. 16, 1667928 (2025).
Das, B. et al. Peroxidase-mimicking activity of biogenic gold nanoparticles produced from Prunus nepalensis fruit extract: Characterizations and application for the detection of Mycobacterium bovis. ACS Appl. Bio Mater. 5(6), 2712–2725 (2022).
Kusuma, S. A. F., Harmonis, J. A., Pratiwi, R. & Hasanah, A. N. Gold nanoparticle-based colorimetric sensors: Properties and application in detection of heavy metals and biological molecules. Sensors 23(19), 8172 (2023).
Al-Radadi, N. S., Al-Bishri, W. M., Salem, N. A. & ElShebiney, S. A. Plant-mediated green synthesis of gold nanoparticles using an aqueous extract of Passiflora ligularis, optimization, characterizations, and their neuroprotective effect on propionic acid-induced autism in Wistar rats. Saudi Pharm. J. 32(2), 101921 (2024).
Huang, X., Devi, S., Bordiga, M., Brennan, C. S. & Xu, B. Phenolic compounds mediated biosynthesis of gold nanoparticles and evaluation of their bioactivities: A review. Int. J. Food Sci. Technol. 58(4), 1673–1694 (2023).
Kahraman, H. T. Synthesis of silver nanoparticles using Alchemilla vulgaris and Helichrysum arenarium for methylene blue and 4-nitrophenol degradation and antibacterial applications. Biomass Convers. Biorefin. 14(12), 13479–13490 (2024).
Usman, M. et al. Evaporation-induced self-assembly of gold nanorods on a hydrophobic substrate for surface enhanced Raman spectroscopy applications. Front. Mater. 9, 1048011 (2023).
Panchal, P. et al. A review on biomedical applications of plant extract-mediated metallic Ag, Au, and ZnO nanoparticles and future prospects for their combination with graphitic carbon nitride. Pharmaceuticals 18(6), 820 (2025).
Holišová, V. et al. Colloidal stability of phytosynthesised gold nanoparticles and their catalytic effects for nerve agent degradation. Sci. Rep. 11, 1–9 (2021).
Nisha, et al. Plant-mediated gold nanoparticles in cancer therapy: Exploring anti-cancer mechanisms, drug delivery applications, and future prospects. Front. Nanotechnol. 6, 1490980 (2024).
Chonanant, C. et al. Green synthesis of biocompatible gold nanoparticles using Andrographis paniculata leaves extract for targeted detection of human epidermal growth factor receptor 2-positive breast cancer cells. ACS Org. Inorg. Au 5(5), 328–337 (2025).
Bilardo, R., Traldi, F., Vdovchenko, A. & Resmini, M. Influence of surface chemistry and morphology of nanoparticles on protein corona formation. WIREs Nanomed. Nanobiotechnol. 14(4), e1788 (2022).
Uritu, C. M. et al. Radiolabeled multi-layered coated gold nanoparticles as potential biocompatible PET/SPECT tracers. J. Mater. Chem. B 12(15), 3659–3675 (2024).
Hussain, M. H. et al. Synthesis of various size gold nanoparticles by chemical reduction method with different solvent polarity. Nanoscale Res. Lett. 15(1), 140 (2020).
Leach, R. Characterisation of Areal Surface Texture (Springer, 2013).
Lipa, Z. & Tomanícková, D. Utilisation of Abbott–Firestone curves characteristics for the determination of turned surface properties. Ann. Fac. Eng. Hunedoara 9(3), 223 (2011).
Tukova, A., Tavakkoli Yaraki, M., Rodger, A. & Wang, Y. Shape-induced variations in aromatic thiols adsorption on gold nanoparticle: A novel method for accurate evaluation of adsorbed molecules. Langmuir 39(44), 15828–15836 (2023).
Yang, Y. et al. Protein adsorption at nanorough titanium oxide surfaces: The importance of surface statistical parameters beyond surface roughness. Nanomaterials 11(2), 357 (2021).
Adamczyk, Z., Sadowska, M. & Nattich-Rak, M. Quantifying nanoparticle layer topography: Theoretical modeling and atomic force microscopy investigations. Langmuir 39(42), 15067–15077 (2023).
Mizzi, L., Chatzitzika, C., Gatt, R. & Valdramidis, V. HPLC analysis of phenolic compounds and flavonoids with overlapping peaks. Food Technol. Biotechnol. 58(1), 12–19 (2020).
Irakli, M., Skendi, A., Bouloumpasi, E., Chatzopoulou, P. & Biliaderis, C. G. LC–MS identification and quantification of phenolic compounds in solid residues from the essential oil industry. Antioxidants 10(12), 2016 (2021).
Riss, T. L., Moravec, R. A., Niles, A. L., Duellman, S., Benink, H. A., Worzella, T. J., et al. Cell Viability Assays. Assay Guidance Manual (2016).
Gruber, S. & Nickel, A. Toxic or not toxic? Interlaboratory comparison reveals almost fifty-fifty chance in the cytotoxicity assessment (ISO 10993-5) of an identical medical device. MedRxiv. 2023.03. 28.23287847 (2023).
Jablonska, E., Kubásek, J., Vojtěch, D., Ruml, T. & Lipov, J. Test conditions can significantly affect the results of in vitro cytotoxicity testing of degradable metallic biomaterials. Sci. Rep. 11(1), 6628 (2021).
Pfaff, A., Chernatynskaya, A., Vineyard, H. & Ercal, N. Thiol antioxidants protect human lens epithelial (HLE B-3) cells against tert-butyl hydroperoxide-induced oxidative damage and cytotoxicity. Biochem. Biophys. Rep. 29, 101213 (2022).
Kanak, S., Krzemińska, B., Celiński, R., Bakalczuk, M. & Dos Santos, S. K. Phenolic composition and antioxidant activity of Alchemilla species. Plants 11(20), 2709 (2022).
Lee, H.-L. et al. Gold nanodandelions as nanozymes and reactive oxygen species scavengers in tumor microenvironment components. ACS Appl. Nano Mater. 6(22), 21359–21370 (2023).
Dong, Y. et al. Self-assembly of amphiphilic phospholipid peptide dendrimer-based nanovectors for effective delivery of siRNA therapeutics in prostate cancer therapy. J. Control Release 322, 416–425 (2020).
Kanniyappan, H., Jose, J., Chakraborty, S., Ramasamy, M. & Muthuvijayan, V. pH-responsive drug release from positively charged mesoporous silica nanoparticles and their potential for anticancer drug delivery. J. Aust. Ceram. Soc. 59(1), 207–220 (2023).
Sandaruwan, H. B. et al. Next-generation methods for precise pH detection in ocular chemical burns: A review of recent analytical advancements. Anal. Methods 17(3), 408–431 (2025).
Amrutkar, C. S. & Patil, S. B. Nanocarriers for ocular drug delivery: Recent advances and future opportunities. Indian J. Ophthalmol. 71(6), 2355–2366 (2023).
Islam, S. et al. α-Crystallin chaperone mimetic drugs inhibit lens γ-crystallin aggregation: Potential role for cataract prevention. J. Biol. Chem. 298(10), 102417 (2022).
Hill, J. A. et al. An ultraviolet-driven rescue pathway for oxidative stress to eye lens protein human gamma-D crystallin. Commun. Chem. 7(1), 81 (2024).
Basak, A. et al. High-resolution X-ray crystal structures of human γD crystallin (1.25 Å) and the R58H mutant (1.15 Å) associated with aculeiform cataract. J. Mol. Biol. 328(5), 1137–1147 (2003).
Van Montfort, R. L., Bateman, O. A., Lubsen, N. H. & Slingsby, C. Crystal structure of truncated human βB1‐crystallin. Protein Sci. 12(11), 2606–2612 (2003).
Andley, U. P. Crystallins in the eye: Function and pathology. Prog. Retin. Eye Res. 26(1), 78–98 (2007).
Li, H. et al. The mechanism for thermal-enhanced chaperone-like activity of α-crystallin against UV irradiation-induced aggregation of γD-crystallin. Biophys. J. 121(12), 2233–2250 (2022).
Kolodziejczyk, R. et al. Crystal structure of human cystatin C stabilized against amyloid formation. FEBS J. 277(7), 1726–1737 (2010).
Moreau, K. L. & King, J. A. Protein misfolding and aggregation in cataract disease and prospects for prevention. Trends Mol. Med. 18(5), 273–282 (2012).
Wang, L., Li, X., Men, X., Liu, X. & Luo, J. Research progress on antioxidants and protein aggregation inhibitors in cataract prevention and therapy. Mol. Med. Rep. 31(1), 22–22 (2024).
Timsina, R. & Mainali, L. Association of alpha-crystallin with fiber cell plasma membrane of the eye lens accompanied by light scattering and cataract formation. Membranes 11(6), 447 (2021).
de Diego-García, L., Rejas-González, R., Latre, I. C. & Guzman-Aranguez, A. Pharmacological strategies for cataract management: From molecular targets to clinical translation. Int. J. Mol. Sci. 26(12), 5658 (2025).
Daina, A., Michielin, O. & Zoete, V. SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 7(1), 42717 (2017).
Daina, A. & Zoete, V. A boiled‐egg to predict gastrointestinal absorption and brain penetration of small molecules. ChemMedChem 11(11), 1117–1121 (2016).
Chitra, P. S. et al. Status of oxidative stress markers, advanced glycation index, and polyol pathway in age-related cataract subjects with and without diabetes. Exp. Eye Res. 200, 108230 (2020).
Ahmed, S., Amin, M. M. & Sayed, S. Ocular drug delivery: A comprehensive review. AAPS PharmSciTech 24(2), 66 (2023).
Masse, F., Ouellette, M., Lamoureux, G. & Boisselier, E. Gold nanoparticles in ophthalmology. Med. Res. Rev. 39(1), 302–327 (2019).
Guo, Z., Ma, X., Zhang, R. X. & Yan, H. Oxidative stress, epigenetic regulation and pathological processes of lens epithelial cells underlying diabetic cataract. Adv. Ophthalmol. Pract. Res. 3(4), 180–186 (2023).
Souza, J. G., Dias, K., Pereira, T. A., Bernardi, D. S. & Lopez, R. F. Topical delivery of ocular therapeutics: Carrier systems and physical methods. J. Pharm. Pharmacol. 66(4), 507–530 (2014).
Fangueiro, F. J., Veiga, F., Silva, M. A. & Souto, B. E. Ocular drug delivery-new strategies for targeting anterior and posterior segments of the eye. Curr. Pharm. Des. 22(9), 1135–1146 (2016).
Acknowledgements
This work was a PhD project for Ahmed which issued in 2023. Deep appreciation is also extended to the staff of Ibn Al-Haitham Teaching Eye Hospital (Baghdad, Iraq) for their generous cooperation in providing human cataractous lens samples and facilitating the ex vivo experimental procedures. The authors further extends his profound thanks to the Department of Chemistry, College of Science, Mustansiriyah University, for their technical support and assistance.
Author information
Authors and Affiliations
Contributions
Ahmed Salih Abid performed the lab work, interpreted the results, wrote the first draft, edited it, and authorized the final draft. Zahraa S. Al-Garawi designed the study, supervised the whole project, interpreted the results, participated in writing the first and final version, edited and authorized the final version. Füreya Elif Öztürkkan performed the computerized and theoretical study, participated in writing the first and final version, edited and authorized the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The Institutional Ethics Committee of Ibn Al-Haitham Teaching Eye Hospital – Minsitry of Health in Baghdad, Iraq, reviewed and approved all experimental protocols that used samples of human cataractous lenses. The study adhered to the ethical guidelines outlined in the Declaration of Helsinki, and written informed consent was secured from all participants prior to sample collection. The College of Science Ethics committee in Mustansiriyah University approved the study proposal. This Ethics committee is working in accordance to College of Science guidelines on biomedical research. Ref. BCSMU/0225/00020C. None of the Investigator and co-investigator participating in this study took part in the decision making and voting procedure for this study. The College of Science`s Scientific committee expects to be informed about the progress of the study, any serious adverse events occurring in the course of the study, and any revision in the protocol.
AI-assisted language editing statement
The authors enhanced the manuscript’s grammar, readability, and clarity by using AI-based language editing tools (ChatGPT, OpenAI).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Abid, A.S., Al-Garawi, Z.S. & Öztürkkan, F.E. Green-synthesized N-acetylcarnosine–loaded gold nanoparticles as a novel ocular nanocarrier for antioxidant therapy and cataract prevention. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43070-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43070-1