Abstract
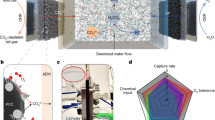
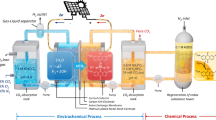
The electrochemical reduction of CO2 to oxalate has emerged as a promising pathway for both carbon utilization and negative-emission strategies, as it couples renewable electricity with the production of a high-value platform chemical. In this work, we investigated the electroreduction of CO2 in a novel designed-flow reactor employing stainless steel cathode in an acetonitrile medium. The reactor design was evaluated by varying electrode spacing (0.5, 1, and 2 mm) and scaling electrode area (from 10 mm2 to 656 mm²), aiming to enhance mass transport and reduce ohmic losses. Faradaic efficiencies up to 72% and current densities above 130 mA cm−2 were achieved, which surpass previously reported results for flow systems. Notably, scaling up to 656 mm² electrodes maintained competitive efficiency while significantly improving oxalate production rates. These results demonstrate one of the few successful demonstrations of CO2-to-oxalate conversion in a continuous-flow configuration, highlighting the potential of reactor engineering approaches for advancing scalable and environmentally benign CO2 electroreduction technologies.
Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Lin, Q., Zhang, X., Wang, T., Zheng, C. & Gao, X. Technical perspective of carbon capture, utilization, and storage. Engineering 14, 27–32. https://doi.org/10.1016/j.eng.2021.12.013 (2022).
Detz, R. J. et al. Electrochemical CO 2 conversion technologies: State-of-the-art and future perspectives. Sustain. Energy Fuels. 7, 5445–5472. https://doi.org/10.1039/D3SE00775H (2023).
Riemenschneider, W. & Tanifuji, M. Oxalic Acid. In: Ullmann’s Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany (2011).
Schuler, E., Demetriou, M., Shiju, N. R. & Gruter, G. M. Towards sustainable oxalic acid from CO 2 and biomass. Chem. Sus. Chem. 14, 3636–3664. https://doi.org/10.1002/cssc.202101272 (2021).
Popescu, I. et al. Eco-friendly synthesized PVA/Chitosan/Oxalic acid nanocomposite hydrogels embedding silver nanoparticles as antibacterial materials. Gels 8, 268. https://doi.org/10.3390/gels8050268 (2022).
Scarpa de Souza, E. L., Neumann, H., Roque Duarte Correia, C. & Beller, M. Proposing oxalic acid as chemical storage of carbon dioxide to achieve carbon neutrality. ChemSusChem 18. https://doi.org/10.1002/cssc.202401199 (2025).
Nogueira, J. A. et al. Development of electrochemical reactors for CO2 electroreduction—the viability of an electrochemical CO2 plant in Brazil. Progress Energy. 4, 043003. https://doi.org/10.1088/2516-1083/ac8865 (2022).
Hori, Y. Electrochemical CO2 Reduction on Metal Electrodes. In: Modern Aspects of Electrochemistry. Springer New York, New York, NY, pp 89–189
Ikeda, S., Takagi, T. & Ito, K. Selective formation of formic acid, oxalic acid, and carbon monoxide by electrochemical reduction of carbon dioxide. Bull. Chem. Soc. Jpn. 60, 2517–2522. https://doi.org/10.1246/bcsj.60.2517 (1987).
Ito, K., Ikeda, S., Yamauchi, N., Iida, T. & Takagi, T. Electrochemical reduction products of carbon dioxide at some metallic electrodes in nonaqueous electrolytes. Bull. Chem. Soc. Jpn. 58, 3027–3028. https://doi.org/10.1246/bcsj.58.3027 (1985).
Fischer, J., Lehmann, T. & Heitz, E. The production of oxalic acid from CO2 and H2O. J. Appl. Electrochem. 11, 743–750. https://doi.org/10.1007/BF00615179 (1981).
Amatore, C. & Saveant, J. M. Mechanism and kinetic characteristics of the electrochemical reduction of carbon dioxide in media of low proton availability. J. Am. Chem. Soc. 103, 5021–5023. https://doi.org/10.1021/ja00407a008 (1981).
Gennaro, A. et al. Mechanism of the electrochemical reduction of carbon dioxide at inert electrodes in media of low proton availability. J. Chem. Soc. Faraday Trans. 92, 3963–3968. https://doi.org/10.1039/FT9969203963 (1996).
Lv, W., Zhang, R., Gao, P., Gong, C. & Lei, L. Electrochemical reduction of carbon dioxide with lead cathode and zinc anode in dry acetonitrile solution. J. Solid State Electrochem. 17, 2789–2794. https://doi.org/10.1007/s10008-013-2186-0 (2013).
König, M., Lin, S. H., Vaes, J., Pant, D. & Klemm, E. Integration of aprotic CO2reduction to oxalate at a Pb catalyst into a GDE flow cell configuration. Faraday Discuss. 230, 360–374. https://doi.org/10.1039/d0fd00141d (2021).
Subramanian, S., Athira, K. R., Anbu Kulandainathan, M., Senthil Kumar, S. & Barik, R. C. New insights into the electrochemical conversion of CO2 to oxalate at stainless steel 304L cathode. J. CO2 Utilization. 36, 105–115. https://doi.org/10.1016/j.jcou.2019.10.011 (2020).
Marx, M., Frauendorf, H., Spannenberg, A., Neumann, H. & Beller, M. Revisiting reduction of CO 2 to oxalate with first-row transition metals: Irreproducibility, ambiguous analysis, and conflicting reactivity. JACS Au. 2, 731–744. https://doi.org/10.1021/jacsau.2c00005 (2022).
Zhou, Y., Wu, X., Zhu, P. & Zhang, W. Single-atom catalysts for CO 2 reduction to oxalate: Theoretical design and reaction condition prediction. ACS Appl. Mater. Interfaces. 17, 49564–49572. https://doi.org/10.1021/acsami.5c11771 (2025).
Sale, H., Ubbara, G. R. & Symes, M. D. Optimising the electrochemical reduction of CO 2 to oxalic acid in propylene carbonate. Sustain. Energy Fuels. 7, 5093–5100. https://doi.org/10.1039/D3SE00652B (2023).
Cheng, Y., Hou, P., Pan, H., Shi, H. & Kang, P. Selective electrocatalytic reduction of carbon dioxide to oxalate by lead tin oxides with low overpotential. Appl. Catal. B. 272, 118954. https://doi.org/10.1016/j.apcatb.2020.118954 (2020).
Brower, R. S. et al. Selective electrochemical reduction of CO 2 to metal oxalates in nonaqueous solutions using trace metal Pb on carbon supports enhanced by a tailored microenvironment. Adv. Energy Mater. 15 https://doi.org/10.1002/aenm.202501286 (2025).
Jia, J. et al. Investigating the electrochemical reduction of CO 2 to oxalate in aprotic solvents with Pb. Chem. Phys. Chem 26. https://doi.org/10.1002/cphc.202500136 (2025).
Masel, R. I. et al. An industrial perspective on catalysts for low-temperature CO2 electrolysis. Nat. Nanotechnol. 16, 118–128. https://doi.org/10.1038/s41565-020-00823-x (2021).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO 2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177. https://doi.org/10.1021/acs.iecr.7b03514 (2018).
Compton, R. G. & Sanders, G. H. W. Electrode Potentials 1st edn (Oxford University Press, 1996).
Wan, T. H., Saccoccio, M., Chen, C. & Ciucci, F. Influence of the discretization methods on the distribution of relaxation times deconvolution: Implementing radial basis functions with DRTtools. Electrochim. Acta. 184, 483–499. https://doi.org/10.1016/j.electacta.2015.09.097 (2015).
Leonide, A., Sonn, V., Weber, A. & Ivers-Tiffée, E. Evaluation and modeling of the cell resistance in anode-supported solid oxide fuel cells. J. Electrochem. Soc. 155, B36. https://doi.org/10.1149/1.2801372 (2008).
Dodson, L. G., Thompson, M. C. & Weber, J. M. Characterization of intermediate oxidation states in CO 2 activation. Annu. Rev. Phys. Chem. 69, 231–252. https://doi.org/10.1146/annurev-physchem-050317-021122 (2018).
Eggins, B. R., Ennis, C., Mcconnell, R. & Spence, M. Improved yields of oxalate, glyoxylate and glycolate from the electrochemical reduction of carbon dioxide in methanol.
Amaral, P. H. R. et al. A machine learning model for adsorption energies of chemical species applied to CO 2 electroreduction. J. Electrochem. Soc. 169, 116505. https://doi.org/10.1149/1945-7111/ac9f7a (2022).
Bard, A. J., Inzelt, G. & Scholz, F. Electrochemical dictionary 1st edn (Springer Berlin Heidelberg, 2008).
Delacourt, C., Ridgway, P. L., Kerr, J. B. & Newman, J. Design of an electrochemical cell making syngas (CO + H[sub 2]) from CO[sub 2] and H[sub 2]O Reduction at room temperature. J. Electrochem. Soc. 155, B42. https://doi.org/10.1149/1.2801871 (2008).
Ma, L. et al. Covalent triazine framework confined copper catalysts for selective electrochemical CO 2 reduction: Operando diagnosis of active sites. ACS Catal. 10, 4534–4542. https://doi.org/10.1021/acscatal.0c00243 (2020).
Sun, L., Ramesha, G. K., Kamat, P. V. & Brennecke, J. F. Switching the reaction course of electrochemical CO 2 reduction with ionic Liquids. Langmuir 30, 6302–6308. https://doi.org/10.1021/la5009076 (2014).
Yang, Y. et al. Aromatic ester-functionalized ionic liquid for highly efficient CO 2 electrochemical reduction to oxalic acid. ChemSusChem 13, 4900–4905. https://doi.org/10.1002/cssc.202001194 (2020).
Lv, W. X., Zhang, R., Gao, P. R., Gong, C. X. & Lei, L. X. Electrochemical reduction of carbon dioxide on stainless steel electrode in acetonitrile. Adv. Mat. Res. 807–809, 1322–1325. https://doi.org/10.4028/www.scientific.net/AMR.807-809.1322 (2013).
Boor, V. et al. Electrochemical reduction of CO 2 to oxalic acid: Experiments, process modeling, and economics. Ind. Eng. Chem. Res. 61, 14837–14846. https://doi.org/10.1021/acs.iecr.2c02647 (2022).
Moia, D. et al. The reorganization energy of intermolecular hole hopping between dyes anchored to surfaces. Chem. Sci. 5, 281–290. https://doi.org/10.1039/C3SC52359D (2014).
Abdelmohsen, A. M., Abdo, L. A., Khedr, G. E. & Allam, N. K. Breaking barriers in nitrate electroreduction: Robust Cu–Zn catalysts for selective ammonia production with ultra-high rate in neutral medium. J. Mater. Chem. Mater. 13, 39970–39981. https://doi.org/10.1039/D5TA07667F (2025).
Ashour, A. A. et al. Tuning C–C Coupling and selectivity in CO 2 electrochemical reduction reaction via pyramidal dilute Sn–Cu alloy. ACS Appl. Mater. Interfaces. 17, 64687–64698. https://doi.org/10.1021/acsami.5c20454 (2025).
Kibria Nabil, S., McCoy, S. & Kibria, M. G. Comparative life cycle assessment of electrochemical upgrading of CO 2 to fuels and feedstocks. Green Chem. 23, 867–880. https://doi.org/10.1039/D0GC02831B (2021).
Liu, J. et al. Experimental investigation of PEM water electrolyser stack performance under dynamic operation conditions. J. Electrochem. Soc. 171, 054521. https://doi.org/10.1149/1945-7111/ad4d1f (2024).
Fragiacomo, P. & Genovese, M. Numerical simulations of the energy performance of a PEM water electrolysis based high-pressure hydrogen refueling station. Int. J. Hydrogen Energy. 45, 27457–27470. https://doi.org/10.1016/j.ijhydene.2020.07.007 (2020).
Funding
The authors gratefully acknowledge support of the RCGI – Research Centre for Greenhouse Gas Innovation (23.1.8493.1.9), hosted at the University of São Paulo (USP) and sponsored by FAPESP – São Paulo Research Foundation (2020/15230-5) and Shell Brasil, and the strategic importance of the support given by ANP (Brazil’s National Oil, Natural Gas and Biofuels Agency) through the R&DI levy regulation. We also thank FAPESP for the support through grants 2023/09823-1, 2017/22130-6, 2024/08727-1, 2022/14049-0, and 2020/01177-5. ECNS would like to thank CNPq for the support under project No. 304508/2023-3.
Author information
Authors and Affiliations
Contributions
DD carried out the experiments, data analysis, and interpretation, experimental design, and wrote the main manuscript text. BNR assisted in the experiments, data analysis, and interpretation and elaborated on Figure 01. LNBSR contributed to reactor designs and to the elaboration of Figure 01. ECNS contributed to manuscript elaboration. JRM contributed to fundraising and project management. TL conceived the project, supervised the work, contributed to fundraising, designed experiments, conducted data analysis and interpretation, and managed the project. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dionisio, D., Narváez-Romo, B., Ribeiro, L.N.B.S. et al. Scalable electrochemical CO2 reduction to oxalate in a continuous flow reactor. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43540-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43540-6