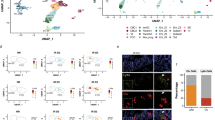
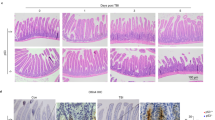
Abstract
Radiotherapy is constricted by collateral normal tissue injury during treatment, particularly the gastrointestinal tracts, which is usually referred as radiation-induced gastrointestinal syndrome (RIGS). Currently, there is no FDA-approved agent for the prevention or treatment of RIGS. By using a mice model of RIGS, we demonstrated that 1,2-propanediol (1,2-PD) prevents radiation-induced fatal intestinal injury and significantly increases mice survival following lethal doses of radiation. 1,2-PD pretreatment also enhanced the survival of Lgr5+ISCs and improved crypts regeneration after radiation. Moreover, we confirmed 1,2-PD induces dormant cell cycle arrest in enterocytes and ameliorates DNA damage both in vitro and in vivo. Although we did have observed 1,2-PD pretreatment inhibiting P53-PUMA signal pathway, but fail to prove its relation with radiation resistance. In RNA sequencing, we have observed 1,2-PD pretreatment significantly upregulates the Hif-2α, Hif-3α and PPARα target gene ACOX2, whilst downregulating the cell cycle drivers E2f3 and Cyclin D2. These results demonstrate that ISCs play a key role in radiation-induced intestinal regeneration and that 1,2-PD acts as a potent intestinal radioprotector
Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. The raw RNA-seq data reported in this study have been deposited in the NCBI Sequence Read Archive (SRA) under BioProject accession number PRJNA1358381.
References
Hauer-Jensen, M., Denham, J. W. & Andreyev, H. J. Radiation enteropathy–pathogenesis, treatment and prevention. Nat. Rev. Gastroenterol. Hepatol. 11, 470–479 (2014).
Kirsch, D. G. et al. p53 controls radiation-induced gastrointestinal syndrome in mice independent of apoptosis. Science 327, 593–596 (2010).
Takeda, N. et al. Interconversion between intestinal stem cell populations in distinct niches. Science 334, 1420–1424 (2011).
Li, L. & Clevers, H. Coexistence of quiescent and active adult stem cells in mammals. Science 327, 542–545 (2010).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007).
Schuijers, J., van der Flier, L. G., van Es, J. & Clevers, H. Robust cre-mediated recombination in small intestinal stem cells utilizing the olfm4 locus. Stem Cell Reports 3, 234–241 (2014).
Sangiorgi, E. & Capecchi, M. R. Bmi1 is expressed in vivo in intestinal stem cells. Nat. Genet. 40, 915–920 (2008).
Munoz, J. et al. The Lgr5 intestinal stem cell signature: Robust expression of proposed quiescent “+4” cell markers. EMBO J. 31, 3079–3091 (2012).
Barker, N., van Oudenaarden, A. & Clevers, H. Identifying the stem cell of the intestinal crypt: Strategies and pitfalls. Cell Stem Cell 11, 452–460 (2012).
Tian, H. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478, 255–259 (2011).
Metcalfe, C., Kljavin, N. M., Ybarra, R. & de Sauvage, F. J. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell 14, 149–159 (2014).
Leibowitz, B. J. et al. Uncoupling p53 functions in radiation-induced intestinal damage via PUMA and p21. Mol. Cancer Res. 9, 616–625 (2011).
Sullivan, J. M. et al. p21 protects “Super p53” mice from the radiation-induced gastrointestinal syndrome. Radiat. Res. 177, 307–310 (2012).
Qiu, W. et al. PUMA regulates intestinal progenitor cell radiosensitivity and gastrointestinal syndrome. Cell Stem Cell 2, 576–583 (2008).
Ruddick, J. A. Toxicology, metabolism, and biochemistry of 1,2-propanediol. Toxicol. Appl. Pharmacol. 21, 102–111 (1972).
Yi, L. et al. The protective effects of 1,2-propanediol against radiation-induced hematopoietic injury in mice. Biomed. Pharmacother. 114, 108806 (2019).
Whaley, D. et al. Cryopreservation: An overview of principles and cell-specific considerations. Cell Transplant. 30, 963689721999617 (2021).
Weeden, C. E. & Asselin-Labat, M. L. Mechanisms of DNA damage repair in adult stem cells and implications for cancer formation. Biochim. Biophys. Acta Mol. Basis Dis. 1864, 89–101 (2018).
Wei, L. et al. Inhibition of CDK4/6 protects against radiation-induced intestinal injury in mice. J. Clin. Invest. 126, 4076–4087 (2016).
Yuan, Q. et al. Disulfiram protects against radiation-induced intestinal injury in mice. Front. Pharmacol. 13, 852669 (2022).
Rogakou, E. P., Pilch, D. R., Orr, A. H., Ivanova, V. S. & Bonner, W. M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 273, 5858–5868 (1998).
Prabhu, K. S. et al. H2AX: A key player in DNA damage response and a promising target for cancer therapy. Biomed. Pharmacother. 175, 116663 (2024).
Chang, P. Y., Qu, Y. Q., Wang, J. & Dong, L. H. The potential of mesenchymal stem cells in the management of radiation enteropathy. Cell Death Dis. 6, e1840 (2015).
Achison, M. & Hupp, T. R. Hypoxia attenuates the p53 response to cellular damage. Oncogene 22, 3431–3440 (2003).
Li, L. et al. Hypoxic preconditioning accelerates the healing of ischemic intestinal injury by activating HIF-1α/PPARα pathway-mediated fatty acid oxidation. Cell Death Discov. 10, 164 (2024).
Leone, G. et al. E2F3 activity is regulated during the cell cycle and is required for the induction of S phase. Genes Dev. 12, 2120–2130 (1998).
Nan, Y. M., Wang, R. Q. & Fu, N. Peroxisome proliferator-activated receptor α, a potential therapeutic target for alcoholic liver disease. World J. Gastroenterol. 20, 8055–8060 (2014).
Yue, R. et al. Activation of PPARα-catalase pathway reverses alcoholic liver injury via upregulating NAD synthesis and accelerating alcohol clearance. Free Radic. Biol. Med. 174, 249–263 (2021).
Sherr, C. J. D-type cyclins. Trends Biochem. Sci. 20, 187–190 (1995).
Yan, K. S. et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations. Proc. Natl. Acad. Sci. U. S. A. 109, 466–471 (2012).
Koo, B. K. & Clevers, H. Stem cells marked by the R-spondin receptor LGR5. Gastroenterology 147, 289–302 (2014).
Zhu, Y., Huang, Y. F., Kek, C. & Bulavin, D. V. Apoptosis differently affects lineage tracing of Lgr5 and Bmi1 intestinal stem cell populations. Cell Stem Cell 12, 298–303 (2013).
Merritt, A. J. et al. The role of p53 in spontaneous and radiation-induced apoptosis in the gastrointestinal tract of normal and p53-deficient mice. Cancer Res. 54, 614–617 (1994).
Wang, J. et al. p53 suppresses MHC class II presentation by intestinal epithelium to protect against radiation-induced gastrointestinal syndrome. Nat. Commun. https://doi.org/10.1038/s41467-023-44390-w (2024).
Diao, X. et al. Identification of oleoylethanolamide as an endogenous ligand for HIF-3α. Nat. Commun. https://doi.org/10.1038/s41467-022-30338-z (2022).
Taniguchi, C. M. et al. PHD inhibition mitigates and protects against radiation-induced gastrointestinal toxicity via HIF2. Sci. Transl. Med. 6, 236ra264 (2014).
Ravi, R. et al. Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1alpha. Genes Dev. 14, 34–44 (2000).
Chen, D., Li, M., Luo, J. & Gu, W. Direct interactions between HIF-1α and Mdm2 modulate p53 function*. J. Biol. Chem. 278, 13595–13598 (2003).
Zhao, Y., Chen, X. Q. & Du, J. Z. Cellular adaptation to hypoxia and p53 transcription regulation. J. Zhejiang Univ. Sci. B 10, 404–410 (2009).
Mahe, M. M. et al. Establishment of gastrointestinal epithelial organoids. Curr. Protoc. Mouse Biol. 3, 217–240 (2013).
Qu, M. et al. Establishment of intestinal organoid cultures modeling injury-associated epithelial regeneration. Cell Res. 31, 259–271 (2021).
Li, M. et al. Vanillin derivative VND3207 activates DNA-PKcs conferring protection against radiation-induced intestinal epithelial cells injury in vitro and in vivo. Toxicol. Appl. Pharmacol. 387, 114855 (2020).
Acknowledgements
We thank Professor Yuwen Cong for his generous gift of Lgr5-EGFP-IRES-CreERT2 mice. We also thank Goulin Xiong and Yin Xu for their technical assistance in experiments. The authors declare no use of Artificial Intelligence in this study.
Funding
This research received no specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Ying Jiang and Xing Shen designed this study and conducted the main analysis. Jiwei Zhao, Chunan Zhao, Ying Jiang, Xing Shen, Aoqiang Ji and Xun Wang performed the experiments. Chunan Zhao wrote and edited the manuscript. Zuyin Yu, Gang Sun, Shuang Xing and Yuwen Cong provided additional expertise. Xing Shen, He Xiao, Zuyin Yu reviewed and edited the article. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
There are no ethical/legal conflicts involved in the article.
Ethics approval
The research was carried out in accordance with the approval of the ethics committee. Studies involving animal experiment were approved by the Institutional Animal Care and Use Committee of the Beijing Institute of Radiation Medicine. The approval project title is "Mechanism Study of Propylene Glycol in the Prevention and Treatment of Intestinal Acute Radiation Syndrome”. The permit number is “Ethics Review Approval no. IACUC-DWZX-2020–599″, which issued at April 15, 2025. All protocols followed are compliant with specific ethical regulations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, J., Zhao, C., Shen, X. et al. 1,2-propanediol ameliorated radiation-induced intestinal injury in mice. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43614-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43614-5