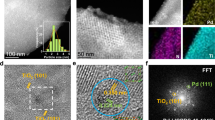
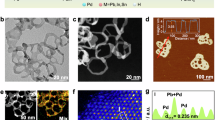
Abstract
The oxidative addition of a bromoporphyrin onto in situ formed highly active Pd(0) complex allowed us to characterize the Pd(II) intermediate by X-ray crystallography. In addition, after the heterobidentate coordination of Xantphos hemioxide (Xantphos: 4,5-bis(diphenylphosphino)-9,9-dimethyl-xanthene), the oxidized product responsible for Pd(II)-Pd(0) reduction, was also proved. The species isolated by us is the product of oxidative addition, the first step of the suggested catalytic cycle. Earlier only the structure of the dichloro complex of palladium with (P-P(O)) ligand was known. The isolated Pd(P-P(O))X(porphyrinyl) complex can be considered as a new example of catalytic intermediates of important cross-coupling and carbonylation reactions giving further strong proof for their mechanism.
Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available in the Crystallographic Data Centre repository, http://www.ccdc.cam.ac.uk/data_request/cif using reference deposition numbers: 2512487 for 2 and 2,512,488 for 3, as well as 2,512,489 for DPP-Bn, 2,512,490 for DPP-Gly, 2,385,880 for DPP-Gly2 and 2,512,491 for DPP-Ala2. In addition to analytical data (1H and 13C NMR, MS) discussed in Supplementary Information, further details are available from the authors upon request.
References
Beller, M., Bolm, C. Transition Metals for Organic Synthesis: Building Blocks and Fine Chemicals, Wiley-VCH, (2004)
Beller, M., Wu, X.-F. Transition Metal Catalyzed Carbonylation Reactions: Carbonylative Activation of C-X Bonds, Springer-Verlag (2013)
Cornils, B., Herrmann, W. A., Beller, M., Paciello, R. Applied Homogeneous Catalysis with Organometallic Compounds: A Comprehensive Handbook in Four Volumes, Third edition, Wiley‐VCH (2017).
Miessler, G. L., Tarr, D. A. Inorganic Chemistry, Third edition, Pearson Prentice Hall (2004)
Shriver, D. F., Atkins, P. W. Inorganic Chemistry, Tthird edition, Oxford University Press (1999)
Crabtree, R. The Organometallic Chemistry of The Transition Metals (Wiley-Interscience, 2005).
Hartwig, J. F. Organotransition Metal Chemistry: from Bonding to Catalysis. University Science Books (2010)
Labinger, J. A. Tutorial on oxidative addition. Organometallics 34, 4784–4795 (2015).
Elschenbroich, C., Salzer, A. Organometallics. A Concise Introduction, Second edition, Wily-VCH (1992).
Tsou, T. T. & Kochi, J. K. Mechanism of oxidative addition. Reaction of nickel(0) complexes with aromatic halides. J. Am. Chem. Soc. 101, 6319–6332 (1979).
Lau, K. S. Y., Wong, P. K. & Stille, J. K. Oxidative addition of benzyl halides to zero-valent palladium complexes. Inversion of configuration at carbon. J. Am. Chem. Soc. 98, 5832–5840 (1976).
Heck, R. F. & Nolley, J. P. Jr. Palladium-catalyzed vinylic hydrogen substitution reactions with aryl, benzyl, and styryl halides. J. Org. Chem. 37, 2320–2322 (1972).
Mizoroki, T., Mori, K. & Ozaki, A. Arylation of olefin with aryl iodide catalyzed by palladium. Bull. Chem. Soc. Jpn. 44, 581–581 (2006).
Knowles, J. P. & Whiting, A. The Heck–Mizoroki cross-coupling reaction: A mechanistic perspective. Org. Biomol. Chem. 5, 31–44 (2007).
O'Connor, C., Wilkinson, G. Selective homogeneous hydrogenation of alk-1-enes using hydridocarbonyltris(triphenylphosphine)rhodium(I) as catalyst. Journal of the Chemical Society A: Inorganic, Physical, Theoretical 2665–2671 (1968)
Yagupsky, G., Brown, C., Wikinson, G. Intermediates or their analogues in hydroformylation of alkenes catalysed by hydridocarbonyltris(triphenylphosphine)rhodium(I). Journal of the Chemical Society D: Chemical Communications 1244–1245 (1969)
Halpern, J. Mechanistic aspects of homogeneous catalytic hydrogenation and related processes. Inorg. Chim. Acta 50, 11–19 (1981).
Schrock, R. R., Osborn, J. A. Catalytic hydrogenation using cationic rhodium complexes. I. Evolution of the catalytic system and the hydrogenation of olefins. Journal of the American Chemical Society 98, 2134–2143 (1976).
Diederich, F. & de Mejiere, A. Metal Catalysed Cross-Coupling Reactions Second edition. (Wiley-VCH, 2004).
Ananikov, V. P., Musaev, D. G. & Morokuma, K. Theoretical insight into the C−C coupling reactions of the vinyl, phenyl, ethynyl, and methyl complexes of palladium and platinum. Organometallics 24, 715–723 (2005).
Corbet, J.-P. & Mignani, G. Selected patented cross-coupling reaction technologies. Chem. Rev. 106, 2651–2710 (2006).
Johansson Seechurn, C. C. C., Kitching, M. O., Colacot, T. J. & Snieckus, V. Palladium-catalyzed cross-coupling: A historical contextual perspective to the 2010 Nobel Prize. Angew. Chem. Int. Ed. 51, 5062–5085 (2012).
Korch, K. M. & Watson, D. A. Cross-coupling of heteroatomic electrophiles. Chem. Rev. 119, 8192–8228 (2019).
Arnold, D. P.; Sakata, Y.; Sugiura, K.; Worthington, E. I. meso-η1-Metalloporphyrins: Preparation of Palladio- and Platinioporphyrins and the Crystal Structure of 5-[Bromo-1,2-Bis(diphenylphosphino)-ethanepalladio(II)-10,20-Diphenylporphyrin. Chem. Commun. 2331−2332 (1998)
Arnold, D. P.; Healy, P. C.; Hodgson, M. J.; Williams, M. L. Peripherally-Metalated Porphyrins: Synthesis and Spectra of meso-η1-Palladio- and Platinioporphyrins and the Crystal Structures of cis-{PtBr[10,20-Diphenylporphyrinatonickel(II)-5-yl](PPh3)2} and trans-{PtBr[10,20-Diphenylporphyrin-5-yl](PPh3)2}·0.25CH2Cl2. J. Organomet. Chem. 607, 41−50 (2000).
Hartnell, R. D.; Arnold, D. P. Peripherally Metallated Porphyrins: The First Examples of meso-η1-Palladio(II) and -Platinio-(II) Complexes with Chelating Diamine Ligands. Eur. J. Inorg. Chem. 1262−1269 (2004)
Hodgson, M. J.; Healy, P. C.; Williams, M. L.; Arnold, D. P. Peripherally-Metalated Porphyrins: Preparations, Spectroscopic Properties and Structural Studies of trans-[PtBr(MDPP) (PPh3)2] (DPP = Dianion of 5,15-Diphenylporphyrin, M = MnCl, Co, Ni, Zn) and Related meso-η1-Organoplatinum Porphyrins. J. Chem. Soc., Dalton Trans. 4497−4504 (2002)
Hodgson, M. J., Borovkov, V. V., Inoue, Y. & Arnold, D. P. A new type of chiral porphyrin: Organopalladium porphyrins with chiral chelating diphosphine ligands. J. Organomet. Chem. 691, 2162–2170 (2006).
Hartnell, R. D., Edwards, A. J. & Arnold, D. P. Peripherally-metalated porphyrins: meso-η1-Porphyrinylplatinum(II) complexes of 5,15-diaryl- and 5,10,15-triarylporphyrins. J. Porphyrins Phthalocyanines 6, 695–707 (2002).
Hartnell, R. D. & Arnold, D. P. Peripherally η1-platinated organometallic porphyrins as building blocks for multiporphyrin arrays. Organometallics 23, 391–399 (2004).
Amatore, C., Jutand, A. & M’Barki, M. A. Evidence of the formation of zerovalent palladium from Pd(OAc)2 and triphenylphosphine. Organometallics 11, 3009–3013 (1992).
Amatore, C., Carre, E., Jutand, A., M’Barki, M. A. & Meyer, G. Evidence for the ligation of palladium(0) complexes by acetate ions: Consequences on the mechanism of their oxidative addition with phenyl iodide and PhPd(OAc)(PPh3)2 as intermediate in the Heck reaction. Organometallics 14, 5605–5614 (1995).
Jover, J. et al. Expansion of the Ligand Knowledge Base for Monodentate P-Donor Ligands (LKB-P). Organometallics 29, 6245–6258 (2010).
Jover, J. et al. Expansion of the Ligand Knowledge Base for Chelating P,P-Donor Ligands (LKB-PP). Organometallics 31, 5302–5306 (2012).
Csákai, Z., Skoda-Földes, R. & Kollár, L. NMR investigation of Pd(II)–Pd(0) reduction in the presence of mono- and ditertiary phosphines. Inorg. Chim. Acta 286, 93–97 (1999).
Amatore, C., Jutand, A. & Thuilliez, A. Formation of palladium(0) complexes from Pd(OAc)2 and a bidentate phosphine ligand (dppp) and their reactivity in oxidative addition. Organometallics 20, 3241–3249 (2001).
Amatore, C., Broeker, G., Jutand, A., Khalil, F. Identification of the Effective Palladium(0) Catalytic Species Generated in Situ from Mixtures of Pd(dba)2 and Bidentate Phosphine Ligands. Determination of Their Rates and Mechanism in Oxidative Addition. Journal of the American Chemical Society 119: 5176–5185 (1997).
van Leeuwen, P. W. N. M. v., Kamer, P. C. J., Reek, J. N. H. The bite angle makes the catalyst. Pure and Applied Chemistry 71, 1443–1452 (1999)
van Leeuwen, P. W. N. M., Kamer, P. C. J., Reek, J. N. H. & Dierkes, P. Ligand bite angle effects in metal-catalyzed C−C bond formation. Chem. Rev. 100, 2741–2770 (2000).
Kamer, P. C. J., van Leeuwen, P. W. N. M. & Reek, J. N. H. Wide bite angle diphosphines: Xantphos ligands in transition metal complexes and catalysis. Acc. Chem. Res. 34, 895–904 (2001).
van der Veen, L. A., Keeven, P. K., Kamer, P. C. J., van Leeuwen, P. W. N. M. Wide bite angle amine, arsine and phosphine ligands in rhodium- and platinum/tin-catalysed hydroformylation. Journal of the Chemical Society, Dalton Transactions, 2105–2112 (2000).
Yin, J. & Buchwald, S. L. Pd-catalyzed intermolecular amidation of aryl halides: The discovery that Xantphos can be trans-chelating in a palladium complex. J. Am. Chem. Soc. 124, 6043–6048 (2002).
Martinelli, J. R., Watson, D. A., Freckmann, D. M. M., Barder, T. E. & Buchwald, S. L. Palladium-catalyzed carbonylation reactions of aryl bromides at atmospheric pressure: A general system based on Xantphos. J. Org. Chem. 73, 7102–7107 (2008).
Kollár, L., Ince, N. U., Zugó, A., Dörnyei, Á. & Takács, A. Selective synthesis of Isoquinoline-1-Carboxamides via palladium-catalyzed aminocarbonylation in DMF and biomass-derived solvents. Catalysts 15, 78 (2025).
Uzunlu Ince, N. et al. Alkyl 4-Alkoxyvalerates: Characterization and application in Pd-catalyzed aminocarbonylation of iodo(hetero)arene compounds. ChemPlusChem 90, e202400713 (2025).
Schoenberg, A. & Heck, R. F. Palladium-catalyzed amidation of aryl, heterocyclic, and vinylic halides. J. Org. Chem. 39, 3327–3331 (1974).
Schoenberg, A., Bartoletti, I. & Heck, R. F. Palladium-catalyzed carboalkoxylation of aryl, benzyl, and vinylic halides. J. Org. Chem. 39, 3318–3326 (1974).
Schoenberg, A. & Heck, R. F. Palladium-catalyzed formylation of aryl, heterocyclic, and vinylic halides. J. Am. Chem. Soc. 96, 7761–7764 (1974).
Szuroczki, P. et al. Unlocking new porphyrin aminoacid bioconjugates with a Pd-catalyzed carboxamide synthesis. ChemPlusChem 90, e202500193 (2025).
Klingensmith, L. M., Strieter, E. R., Barder, T. E. & Buchwald, S. L. New insights into Xantphos/Pd-catalyzed C−N bond forming reactions: A structural and kinetic study. Organometallics 25, 82–91 (2006).
Groom, C. R., Bruno, I. J., Lightfoot, M. P. & Ward, S. C. The Cambridge structural database. Acta Crystallogr. B 72, 171–179 (2016).
Ding, Y., Wu, J. & Huang, H. Carbonylative formal cycloaddition between alkylarenes and aldimines enabled by palladium-catalyzed double C–H bond activation. J. Am. Chem. Soc. 145, 4982–4988 (2023).
Zhao, L., Dong, Y., Xia, Q., Bai, J. & Li, Y. Zn-catalyzed cyanation of aryl iodides. J. Org. Chem. 85, 6471–6477 (2020).
Gladiali, S., Medici, S., Kégl, T. & Kollàr, L. Synthesis, characterization, and catalytic activity of Rh(I) complexes with (S)-BINAPO, an axially chiral inducer capable of hemilabile P,O-heterobidentate coordination. Monatsh. Chem. 131, 1351–1361 (2000).
Gladiali, S., Alberico, E., Pulacchini, S. & Kollàr, L. Synthesis, characterization and use in enantioselective hydroformylation of (BINAPO)PtCl2 (BINAPO=2-diphenylphosphino-2′-diphenylphosphinyl-1,1′-binaphthalene), the first chiral catalyst with an atropisomeric hemilabile P,O-heterodonor ligand. J. Mol. Catal. A Chem. 143, 155–162 (1999).
Wu, X.-F., Neumann, H. & Beller, M. Synthesis of heterocycles via palladium-catalyzed carbonylations. Chem. Rev. 113, 1–35 (2013).
Roy, S., Roy, S. & Gribble, G. W. Metal-catalyzed amidation. Tetrahedron 68, 9867–9923 (2012).
Wu, X.-F., Neumann, H. & Beller, M. Palladium-catalyzed carbonylative coupling reactions between Ar–X and carbon nucleophiles. Chem. Soc. Rev. 40, 4986–5009 (2011).
Magano, J. & Dunetz, J. R. Large-scale applications of transition metal-catalyzed couplings for the synthesis of pharmaceuticals. Chem. Rev. 111, 2177–2250 (2011).
Grigg, R. & Mutton, S. P. Pd-catalysed carbonylations: Versatile technology for discovery and process chemists. Tetrahedron 66, 5515–5548 (2010).
Brennführer, A., Neumann, H. & Beller, M. Palladium-catalyzed carbonylation reactions of aryl halides and related compounds. Angew. Chem. Int. Ed. 48, 4114–4133 (2009).
Barnard, C. F. J. Palladium-catalyzed carbonylation—A reaction come of age. Organometallics 27, 5402–5422 (2008).
Skoda-Foldes, R. & Kollar, L. Synthetic applications of palladium catalysed carbonylation of organic halides. Curr. Org. Chem. 6, 1097–1119 (2002).
Wu, X.-F., Neumann, H. & Beller, M. Selective palladium-catalyzed aminocarbonylation of aryl halides with CO and ammonia. Chem. Eur. J. 16, 9750–9753 (2010).
Gadge, S. T. & Bhanage, B. M. Recent developments in palladium catalysed carbonylation reactions. RSC Adv. 4, 10367–10389 (2014).
Wu, X.-F. Palladium-catalyzed carbonylative transformation of aryl chlorides and aryl tosylates. RSC Adv. 6, 83831–83837 (2016).
Peng, J.-B., Qi, X. & Wu, X.-F. Recent achievements in carbonylation reactions: A personal account. Synlett 28, 175–194 (2017).
DiMagno, S. G., Lin, V. S. Y. & Therien, M. J. Facile elaboration of porphyrins via metal-mediated cross-coupling. J. Org. Chem. 58, 5983–5993 (1993).
Manka, J. S. & Lawrence, D. S. High yield synthesis of 5,15-diarylporphyrins. Tetrahedron Lett. 30, 6989–6992 (1989).
Sheldrick, G. A short history of SHELX. Acta Crystallogr. A 64, 112–122 (2008).
Westrip, S. publCIF: Software for editing, validating and formatting crystallographic information files. J. Appl. Crystallogr. 43, 920–925 (2010).
Macrae, C. F. et al. Mercury 4.0: From visualization to analysis, design and prediction. J. Appl. Crystallogr. 53, 226–235 (2020).
Spek, A. L. PLATON, An integrated tool for the analysis of the results of a single crystal structure determination. Acta Crystallogr. A 46, 34–34 (1990).
Acknowledgements
The research reported in this paper is part of project no. TKP2021-EGA-17, implemented with the support provided by the Ministry of Innovation and Technology of Hungary from the National Research Development and Innovation Fund financed under the TKP2021 funding scheme. The research was supported by the EU and co-financed by the European Regional Development Fund under the project GINOP-2.3.2-15-2016-00008 and also under the project GINOP-2.3.3-15-2016-00004 to purchase the diffractometer at the University of Debrecen. The Coimbra Chemistry Centre-Institute of Molecular Sciences (CQC-IMS) is supported by national funds from Fundação para a Ciência e a Tecnologia (FCT) through projects UIDB/00313/2025 and UIDP/00313/2025 and LA/P/0056/2020 (https://doi.org/10.54499/LA/P/0056/2020). This research was also funded by project “ECOXOPLAS” n. 15161 (COMPETE2030-FEDER-00882400).
Funding
Open access funding provided by University of Pécs. The authors declare funding by the Ministry of Innovation and Technology of Hungary, by the European Regional Development Fund, and by Fundação para a Ciência e a Tecnologia (FCT, Portugal).
Author information
Authors and Affiliations
Contributions
P.S. carried out the catalytic experiments and isolated the complexes as crystals A.B. determined the crystal structures and wrote the discusssion on XRD R.A. prepared the porphyrins M.M.Pereira reviewed the manuscript L.K. wrote the main text.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szuroczki, P., Bényei, A., Aroso, R.T. et al. First time exploration and characterization of key-intermediates in palladium-catalysed coupling reactions. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43634-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43634-1