Abstract
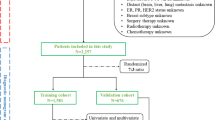
With increasing opportunities for patients with bone metastasis (BM) to benefit from local surgical intervention, accurate survival analysis across different primary cancers remains challenging. Current analytical frameworks commonly rely on single-center, pan-cancer cohorts and provide insufficient integration of cancer-specific characteristics. In this retrospective, multicenter, registry-based cohort study, baseline demographic and clinical characteristics of 13,742 patients with AJCC stage IV or TNM stage M1 metastatic cancer were collected from 42 studies registered in the cBioPortal for Cancer Genomics database. Overall survival (OS) after metastatic diagnosis was the primary outcome. Univariate analyses were performed using Kaplan–Meier methods, log-rank tests, and non-parametric tests. Variables with p < 0.20 were included in multivariable Cox proportional hazards models to examine independent associations with survival. Multiple imputation was applied to address missing data. Among the 25 primary cancers analyzed, approximately half showed observable survival differences between BM and other-site metastasis, with 6 cancers reaching statistical significance. Based on median survival, all cancers could be stratified into 3 distinct survival tiers, ranging from prolonged survival exceeding 15 months to markedly shorter survival of 3–10 months, with multivariable analyses further demonstrating that primary cancer type was the strongest factor associated with survival heterogeneity among BM patients (HR = 1.422–1.758, p < 0.001). Moreover, poorly differentiated or undifferentiated histology was independently associated with worse OS (HR = 1.249, p < 0.001), and age > 60 years was also associated with shorter survival (p < 0.001). No single metastatic site demonstrated a consistent adverse association with survival across cancer types. Overall, BM demonstrates cancer-specific and heterogeneous associations with survival compared with other metastatic sites. All primary cancers could be stratified into 3 groups, representing the most important factor associated with survival differences. Moreover, pathological differentiation was significantly associated with survival among BM patients. Notably, no metastatic site functions as a universal prognostic factor across cancers. Large-scale, multicenter, registry-based analyses provide a valuable framework for cancer-specific survival analysis and for identifying clinically relevant factors that may serve as a reference for risk stratification in surgical decision-making.
Similar content being viewed by others
Data availability
The datasets supporting the conclusions of this study are available from the corresponding author on reasonable request or can be accessed from the cBioPortal for Cancer Genomics (https://www.cbioportal.org/).
Abbreviations
- AJCC:
-
American Joint Committee on Cancer
- BM:
-
Bone metastasis
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- CV:
-
Coefficient of variation
- EGFR:
-
Epidermal growth factor receptor
- EMT:
-
Epithelial–mesenchymal transition
- ECOG:
-
Eastern Cooperative Oncology Group
- HF:
-
Hazard ratio
- IHC:
-
Immunohistochemistry
- IQR:
-
Interquartile range
- K–S:
-
Kolmogorov–Smirnov
- MAXSTAT:
-
Maximally selected rank statistics
- NSCLC:
-
Non-small cell lung cancer
- OBM:
-
Only bone metastasis
- OS:
-
Overall survival
- OSM:
-
Other-site metastasis
- PH:
-
Proportional hazards
- PS:
-
Probability of superiority
- RECORD:
-
Reporting of studies conducted using observational routinely-collected health data
- STROBE:
-
Strengthening the reporting of observational studies in epidemiology
- TNM:
-
Tumor–node–metastasis
- VIF:
-
Variance inflation factor
- WFNS:
-
World federation of neurosurgical societies
References
Challapalli, A. et al. Spine and non-spine bone metastases—Current controversies and future direction. Clin. Oncol. (R Coll Radiol.) 32(11), 728–744. https://doi.org/10.1016/j.clon.2020.07.010 (2020).
Sciubba, D. M. et al. Diagnosis and management of metastatic spine disease. A review. J. Neurosurg. Spine 13(1), 94–108. https://doi.org/10.3171/2010.3.SPINE09202 (2010).
Cole, J. S. & Patchell, R. A. Metastatic epidural spinal cord compression. Lancet Neurol. 7(5), 459–466. https://doi.org/10.1016/S1474-4422(08)70089-9 (2008).
Hong, S. H., Chang, B. S., Kim, H., Kang, D. H. & Chang, S. Y. An updated review on the treatment strategy for spinal metastasis from the spine surgeon’s perspective. Asian Spine J. 16(5), 799–811. https://doi.org/10.31616/asj.2022.0367 (2022).
Laufer, I. & Bilsky, M. H. Advances in the treatment of metastatic spine tumors: The future is not what it used to be. J. Neurosurg. Spine 30(3), 299–307. https://doi.org/10.3171/2018.11.SPINE18709 (2019).
Orenday-Barraza, J. et al. 10-Year trends in the surgical management of patients with spinal metastases: a scoping review. World Neurosurg. 157(170–186), e173 (2022).
Bristow, R. G. et al. Combining precision radiotherapy with molecular targeting and immunomodulatory agents: A guideline by the American Society for Radiation Oncology. Lancet Oncol. 19(5), e240–e251. https://doi.org/10.1016/S1470-2045(18)30096-2 (2018).
Luo, Q., Zhang, L., Luo, C. & Jiang, M. Emerging strategies in cancer therapy combining chemotherapy with immunotherapy. Cancer Lett. 454, 191–203. https://doi.org/10.1016/j.canlet.2019.04.017 (2019).
Li, F., Zhao, C. & Wang, L. Molecular-targeted agents combination therapy for cancer: Developments and potentials. Int. J. Cancer 134(6), 1257–1269. https://doi.org/10.1002/ijc.28261 (2014).
Zileli, M. et al. Surgical options for metastatic spine tumors: WFNS spine committee recommendations. Neurosurg. Rev. 47(1), 821. https://doi.org/10.1007/s10143-024-02949-1 (2024).
Tokuhashi, Y., Uei, H., Oshima, M. & Ajiro, Y. Scoring system for prediction of metastatic spine tumor prognosis. World J. Orthop. 5(3), 262–271. https://doi.org/10.5312/wjo.v5.i3.262 (2014).
Park, S. J., Park, J. S., Kang, D. H. & Lee, C. S. Which prognostic model best predicts poor prognosis in patients with spinal metastases? A comparative analysis of 8 scoring systems. World Neurosurg. 193, 553–566. https://doi.org/10.1016/j.wneu.2024.09.123 (2025).
Yan, Y. et al. Comparing the accuracy of seven scoring systems in predicting survival of lung cancer patients with spinal metastases: An external validation from two centers. Spine (Phila Pa 1976) 48(14), 1009–1016. https://doi.org/10.1097/BRS.0000000000004576 (2023).
Chanplakorn, P. et al. Validation of traditional prognosis scoring systems and Skeletal Oncology Research Group nomogram for predicting survival of spinal metastasis patients undergoing surgery. Clin. Orthop. Surg. 14(4), 548–556. https://doi.org/10.4055/cios22014 (2022).
Nater, A. et al. Predictive factors of survival in a surgical series of metastatic epidural spinal cord compression and complete external validation of 8 multivariate models of survival in a prospective North American multicenter study. Cancer 124(17), 3536–3550. https://doi.org/10.1002/cncr.31585 (2018).
Park, S.-J., Park, J.-S., Kang, D.-H. & Lee, C.-S. Which prognostic model best predicts poor prognosis in patients with spinal metastases? A comparative analysis of 8 scoring systems. World Neurosurg. 193, 553–566 (2025).
Ahmed, A. K. et al. Predicting survival for metastatic spine disease: A comparison of nine scoring systems. Spine J. 18(10), 1804–1814. https://doi.org/10.1016/j.spinee.2018.03.011 (2018).
Yang, M. et al. Prediction of survival prognosis for spinal metastasis from cancer of unknown primary: Derivation and validation of a nomogram model. Global Spine J. 14(1), 283–294. https://doi.org/10.1177/21925682221103833 (2024).
Hu, C., Wu, J., Duan, Z., Qian, J. & Zhu, J. Risk factor analysis and predictive model construction for bone metastasis in newly diagnosed malignant tumor patients. Am. J. Transl. Res. 16(10), 5890–5899. https://doi.org/10.62347/MPEV9272 (2024).
Krishnan, M. et al. Predicting life expectancy in patients with advanced incurable cancer: A review. J. Support Oncol. 11(2), 68–74. https://doi.org/10.12788/j.suponc.0004 (2013).
Mezei, T. et al. A novel prognostication system for spinal metastasis patients based on network science and correlation analysis. Clin. Oncol. (R Coll Radiol.) 35(1), e20–e29. https://doi.org/10.1016/j.clon.2022.09.054 (2023).
Bjerregaard-Michelsen, S., Poulsen, L. O., Bjerrum, A., Bogsted, M. & Vesteghem, C. Machine learning for prediction of 30-day mortality in patients with advanced cancer: Comparing pan-cancer and single-cancer models. ESMO Real World Data Digit. Oncol. 8, 100146. https://doi.org/10.1016/j.esmorw.2025.100146 (2025).
Oh, E. J., Parikh, R. B., Chivers, C. & Chen, J. Two-stage approaches to accounting for patient heterogeneity in machine learning risk prediction models in oncology. JCO Clin. Cancer Inform. 5, 1015–1023. https://doi.org/10.1200/CCI.21.00077 (2021).
Bindels, B. et al. External validation of twelve existing survival prediction models for patients with spinal metastases. Spine J. 25, 1347–1359 (2025).
Fung, M. Y., Wong, Y. L., Cheung, K. M., Bao, K. H. K. & Sung, W. W. Y. Prognostic models for survival predictions in advanced cancer patients: A systematic review and meta-analysis. BMC Palliat. Care 24(1), 54. https://doi.org/10.1186/s12904-025-01696-4 (2025).
Morris, L. G. et al. Pan-cancer analysis of intratumor heterogeneity as a prognostic determinant of survival. Oncotarget 7(9), 10051–10063. https://doi.org/10.18632/oncotarget.7067 (2016).
Karhade, A. V., Shin, J. H. & Schwab, J. H. Prognostic models for spinal metastatic disease: Evolution of methodologies, limitations, and future opportunities. Ann. Transl. Med. 7(10), 219. https://doi.org/10.21037/atm.2019.04.87 (2019).
Phung, M. T., Tin Tin, S. & Elwood, J. M. Prognostic models for breast cancer: A systematic review. BMC Cancer 19(1), 230. https://doi.org/10.1186/s12885-019-5442-6 (2019).
Goh, V. et al. Evaluation of prognostic models to improve prediction of metastasis in patients following potentially curative treatment for primary colorectal cancer: The PROSPECT trial. Health Technol. Assess. 29(8), 1–91. https://doi.org/10.3310/BTMT7049 (2025).
Ben Gal, O. et al. The prediction of survival after surgical management of bone metastases of the extremities-A comparison of prognostic models. Curr. Oncol. 29(7), 4703–4716. https://doi.org/10.3390/curroncol29070373 (2022).
Gan, C. L., Dudani, S. & Heng, D. Y. C. Prognostic and predictive factors in metastatic renal cell carcinoma: Current perspective and a look into the future. Cancer J. 26(5), 365–375. https://doi.org/10.1097/PPO.0000000000000468 (2020).
Luyendijk, M. et al. Changes in survival in de novo metastatic cancer in an era of new medicines. J. Natl. Cancer Inst. 115(6), 628–635. https://doi.org/10.1093/jnci/djad020 (2023).
Hudock, N. L. et al. Future trends in incidence and long-term survival of metastatic cancer in the United States. Commun. Med. 3(1), 76. https://doi.org/10.1038/s43856-023-00304-x (2023).
von Elm, E. et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 61(4), 344–349. https://doi.org/10.1016/j.jclinepi.2007.11.008 (2008).
Benchimol, E. I. et al. The REporting of studies conducted using observational routinely-collected health data (RECORD) statement. PLoS Med. 12(10), e1001885. https://doi.org/10.1371/journal.pmed.1001885 (2015).
The cBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data. https://www.cbioportal.org/.
Brierley, J. D., Van Eycken, E., Rous, B. & Giuliani, M. TNM Classification of Malignant Tumours (John Wiley & Sons, 2025).
Amin, M. B. et al. AJCC cancer staging manual Vol. 1024 (Springer, 2017).
WHO Classification of Tumours, 5th Edition [https://tumourclassification.iarc.who.int/welcome/].
Jakobsen, J. C., Gluud, C., Wetterslev, J. & Winkel, P. When and how should multiple imputation be used for handling missing data in randomised clinical trials—A practical guide with flowcharts. BMC Med. Res. Methodol. 17(1), 162. https://doi.org/10.1186/s12874-017-0442-1 (2017).
Efthimiou, O. et al. Developing clinical prediction models: a step-by-step guide. BMJ 386, e078276. https://doi.org/10.1136/bmj-2023-078276 (2024).
Riley, R. D. et al. Evaluation of clinical prediction models (part 2): How to undertake an external validation study. BMJ 384, e074820. https://doi.org/10.1136/bmj-2023-074820 (2024).
Field, A. Discovering Statistics Using IBM SPSS Statistics (Sage Publications Limited, 2024).
Moore, D. S., McCabe, G. P. & Craig, B. A. introduction to the practice of statistics Vol. 4 (WH Freeman, 2009).
Concato, J., Feinstein, A. R. & Holford, T. R. The risk of determining risk with multivariable models. Ann. Intern. Med. 118(3), 201–210. https://doi.org/10.7326/0003-4819-118-3-199302010-00009 (1993).
Bursac, Z., Gauss, C. H., Williams, D. K. & Hosmer, D. W. Purposeful selection of variables in logistic regression. Source Code Biol. Med. 3(1), 17. https://doi.org/10.1186/1751-0473-3-17 (2008).
McLernon, D. J. et al. Assessing performance and clinical usefulness in prediction models with survival outcomes: practical guidance for cox proportional hazards models. Ann. Intern. Med. 176(1), 105–114. https://doi.org/10.7326/M22-0844 (2023).
Hothorn, T. & Lausen, B. On the exact distribution of maximally selected rank statistics. Comput. Stat. Data Anal. 43(2), 121–137 (2003).
Schoenfeld, A. J. et al. Assessing the utility of a clinical prediction score regarding 30-day morbidity and mortality following metastatic spinal surgery: The New England Spinal Metastasis Score (NESMS). Spine J. 16(4), 482–490. https://doi.org/10.1016/j.spinee.2015.09.043 (2016).
Balain, B. et al. The Oswestry Risk Index: An aid in the treatment of metastatic disease of the spine. Bone Joint J. 95-B(2), 210–216. https://doi.org/10.1302/0301-620X.95B2.29323 (2013).
Katagiri, H. et al. New prognostic factors and scoring system for patients with skeletal metastasis. Cancer Med. 3(5), 1359–1367. https://doi.org/10.1002/cam4.292 (2014).
Bollen, L. et al. Prognostic factors associated with survival in patients with symptomatic spinal bone metastases: A retrospective cohort study of 1,043 patients. Neuro Oncol. 16(7), 991–998. https://doi.org/10.1093/neuonc/not318 (2014).
van der Linden, Y. M. et al. Prediction of survival in patients with metastases in the spinal column: Results based on a randomized trial of radiotherapy. Cancer 103(2), 320–328. https://doi.org/10.1002/cncr.20756 (2005).
Katagiri, H. et al. Prognostic factors and a scoring system for patients with skeletal metastasis. J. Bone Joint Surg. Br. 87(5), 698–703. https://doi.org/10.1302/0301-620X.87B5.15185 (2005).
Tokuhashi, Y., Matsuzaki, H., Oda, H., Oshima, M. & Ryu, J. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine (Phila Pa 1976) 30(19), 2186–2191. https://doi.org/10.1097/01.brs.0000180401.06919.a5 (2005).
Bauer, H. C. Controversies in the surgical management of skeletal metastases. J. Bone Joint Surg. Br. 87(5), 608–617. https://doi.org/10.1302/0301-620X.87B5.16021 (2005).
Tomita, K. et al. Surgical strategy for spinal metastases. Spine (Phila Pa 1976) 26(3), 298–306. https://doi.org/10.1097/00007632-200102010-00016 (2001).
Tokuhashi, Y., Matsuzaki, H., Toriyama, S., Kawano, H. & Ohsaka, S. Scoring system for the preoperative evaluation of metastatic spine tumor prognosis. Spine (Phila Pa 1976) 15(11), 1110–1113. https://doi.org/10.1097/00007632-199011010-00005 (1990).
Yucel, B. et al. The negative prognostic impact of bone metastasis with a tumor mass. Clinics (Sao Paulo) 70(8), 535–540. https://doi.org/10.6061/clinics/2015(08)01 (2015).
Luksanapruksa, P. et al. Prognostic factors in patients with spinal metastasis: A systematic review and meta-analysis. Spine J. 17(5), 689–708. https://doi.org/10.1016/j.spinee.2016.12.003 (2017).
Bollen, L. et al. A systematic review of prognostic factors predicting survival in patients with spinal bone metastases. Eur. Spine J. 27(4), 799–805. https://doi.org/10.1007/s00586-017-5320-3 (2018).
Szendroi, M., Antal, I., Szendroi, A., Lazary, A. & Varga, P. P. Diagnostic algorithm, prognostic factors and surgical treatment of metastatic cancer diseases of the long bones and spine. EFORT Open Rev. 2(9), 372–381. https://doi.org/10.1302/2058-5241.2.170006 (2017).
Yun, J. H. et al. Clinical features and prognostic factors of spinal metastatic pancreatic cancer: A retrospective observational study. Medicine (Baltimore) 102(28), e34264. https://doi.org/10.1097/MD.0000000000034264 (2023).
Kawahara, N., Tomita, K., Murakami, H. & Demura, S. Total en bloc spondylectomy for spinal tumors: Surgical techniques and related basic background. Orthop. Clin. North Am. 40(1), 47–63. https://doi.org/10.1016/j.ocl.2008.09.004 (2009).
Romuald, K. et al. Prognostic factors influencing survival rate of patients operated for prostate-related metastatic spinal cord compression (MSCC). Indian J. Surg. Oncol. 11(3), 498–508. https://doi.org/10.1007/s13193-020-01155-x (2020).
Ren, Y. et al. Predicting survival of patients with bone metastasis of unknown origin. Front. Endocrinol. (Lausanne) 14, 1193318. https://doi.org/10.3389/fendo.2023.1193318 (2023).
Zheng, Z., Hu, Y., Ren, Y., Mo, G. & Wan, H. Correlation between metastatic patterns and age in patients with metastatic primary liver cancer: A population-based study. PLoS ONE 18(1), e0267809. https://doi.org/10.1371/journal.pone.0267809 (2023).
Dong, M. et al. Sex differences in cancer incidence and survival: A pan-cancer analysis. Cancer Epidemiol. Biomarkers Prev. 29(7), 1389–1397. https://doi.org/10.1158/1055-9965.EPI-20-0036 (2020).
Cook, M. B., McGlynn, K. A., Devesa, S. S., Freedman, N. D. & Anderson, W. F. Sex disparities in cancer mortality and survival. Cancer Epidemiol. Biomarkers Prev. 20(8), 1629–1637. https://doi.org/10.1158/1055-9965.EPI-11-0246 (2011).
Yang, D. et al. Survival of metastatic gastric cancer: Significance of age, sex and race/ethnicity. J. Gastrointest. Oncol. 2(2), 77–84. https://doi.org/10.3978/j.issn.2078-6891.2010.025 (2011).
Wang, Y. et al. Sex disparities in the clinical characteristics, synchronous distant metastasis occurrence and prognosis: A pan-cancer analysis. J. Cancer 12(2), 498–507. https://doi.org/10.7150/jca.50536 (2021).
Goodwin, C. R. et al. Gender and sex differences in health-related quality of life, clinical outcomes and survival after treatment of metastatic spine disease. Spine (Phila Pa 1976) https://doi.org/10.1097/BRS.0000000000004910 (2023).
Luo, X., E, Y., Wu, J., Lu, C. & Zhang, J. Analysis of prognostic factors in different grades of histologic differentiation in colorectal cancer patients receiving preoperative neoadjuvant chemotherapy and establishment of prognostic nomograms for moderately differentiated grade. Int. J. Colorectal Dis. 38(1), 237. https://doi.org/10.1007/s00384-023-04539-x (2023).
Karihtala, P., Jääskeläinen, A., Roininen, N. & Jukkola, A. Prognostic factors in metastatic breast cancer: A prospective single-centre cohort study in a Finnish university hospital. BMJ Open 10(10), e038798 (2020).
Hu, M., Shao, B., Ran, R. & Li, H. Prognostic factors for patients with metastatic breast cancer: A literature review. Transl. Cancer Res. 10(4), 1644–1655. https://doi.org/10.21037/tcr-20-2119 (2021).
Kim, H. et al. Prognosis of differentiated thyroid carcinoma with initial distant metastasis: A multicenter study in Korea. Endocrinol. Metab. (Seoul) 33(2), 287–295. https://doi.org/10.3803/EnM.2018.33.2.287 (2018).
Paulino Pereira, N. R. et al. The SORG nomogram accurately predicts 3‐and 12‐months survival for operable spine metastatic disease: External validation. J. Surg. Oncol. 115(8), 1019–1027. https://doi.org/10.1002/jso.24620 (2017).
Collins, G. S. et al. Evaluation of clinical prediction models (Part 1): From development to external validation. BMJ 384, e074819. https://doi.org/10.1136/bmj-2023-074819 (2024).
Riley, R. D. et al. Evaluation of clinical prediction models (Part 3): Calculating the sample size required for an external validation study. BMJ 384, e074821. https://doi.org/10.1136/bmj-2023-074821 (2024).
Jairam, V. & Park, H. S. Strengths and limitations of large databases in lung cancer radiation oncology research. Transl. Lung Cancer Res. 8(Suppl 2), S172–S183. https://doi.org/10.21037/tlcr.2019.05.06 (2019).
Plichta, J. K. et al. Implications of missing data on reported breast cancer mortality. Breast Cancer Res. Treat. 197(1), 177–187. https://doi.org/10.1007/s10549-022-06764-4 (2023).
Song, Q. et al. Nomogram prediction of overall survival in patients with bone metastases from hepatocellular carcinoma: A SEER-based retrospective cohort study. Sci. Rep. 15(1), 21767. https://doi.org/10.1038/s41598-025-06665-8 (2025).
Li, C. et al. Novel models by machine learning to predict prognosis of breast cancer brain metastases. J. Transl. Med. 21(1), 404. https://doi.org/10.1186/s12967-023-04277-2 (2023).
Xiong, F. et al. A machine learning-based model to predict early death among bone metastatic breast cancer patients: A large cohort of 16,189 patients. Front. Cell Dev. Biol. 10, 1059597. https://doi.org/10.3389/fcell.2022.1059597 (2022).
Acknowledgements
We sincerely thank Prof. Wei and Prof. Liu for their valuable guidance on integrating this project with clinical practice. The authors gratefully acknowledge Peking University Third Hospital and the National Natural Science Foundation of China for hardware support in data processing and financial funding for the overall implementation of this project.
Funding
This study was supported by the Peking University Third Hospital (Grant No. BYSYZD2023017) and the National Natural Science Foundation of China (Grant Nos. 82201644 and 82471505). The funding bodies had no role in the design of the study, data collection, analysis, interpretation of data, or in writing the manuscript.
Author information
Authors and Affiliations
Contributions
Z.L. Yun, Y.C. Tang, and J. Sun conceived and designed the study. Z.L. Yun, J.C. Lei, and G.Q. Zhang collected the data. Z.L. Yun and Y.C. Tang performed the statistical analysis. Z.L. Yun drafted the manuscript. F. Wei and X.G. Liu supervised the study and were responsible for manuscript revision and correspondence. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study is retrospective and registry-based without clinical or experimental intervention, and the data used were collected from a public database. For these reasons, informed consent was applied for exemption. The study was conducted with the support of Peking University Third Hospital. Ethical approval was granted by the Research Ethics Committee (approval number: LM2025366).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yun, Z., Tang, Y., Sun, J. et al. Cancer-specific survival patterns in patients with bone metastasis: a registry-based analysis of 13,742 patients. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43780-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43780-6