Abstract
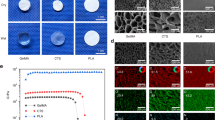
Bone defects resulting from trauma, tumors, or infection continue to pose significant clinical challenges, particularly when the defect size exceeds the capacity for spontaneous healing. In this study, we fabricated two types of composite scaffolds—calcium carbonate/mesoporous silica/poly(lactic-co-glycolic acid) (CMP) and mesoporous silica/poly(lactic-co-glycolic acid) (MP)—using a single-emulsion solvent evaporation method. Both scaffolds exhibited interconnected porous structures and favorable morphology. In vitro assays demonstrated that CMP scaffolds more effectively promoted the proliferation and osteogenic differentiation of mesenchymal stem cells (MSCs) compared to MP scaffolds. Furthermore, a rabbit femoral condylar defect model was established to assess the in vivo bone regeneration efficacy and biocompatibility. Micro-CT imaging, along with hematoxylin-eosin (HE), Masson’s trichrome, and Movat’s pentachrome staining, as well as serum biochemical analyses, consistently indicated that CMP scaffolds significantly enhanced new bone formation and defect repair relative to MP scaffolds. Both scaffold types showed excellent tissue compatibility and elicited no adverse systemic effects. These findings suggest that calcium-enriched mesoporous silica/PLGA scaffolds hold strong potential as clinical biomaterials for the treatment of bone defects.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are include in this published article.
References
Qi, J. et al. Development and characterization of cannabidiol-loaded alginate copper hydrogel for repairing open bone defects in vitro[J]. Colloids Surf. B Biointerfaces. 212, 112339 (2022).
Yang, J. et al. Flavonoid-Loaded Biomaterials in Bone Defect Repair. Molecules, 28(19), 6888 (2023).
Liu, Y. et al. Application of dental pulp stem cells for bone regeneration[J]. Front. Med. (Lausanne). 11, 1339573 (2024).
Sadek, A. A. et al. Enhancement of critical-sized bone defect regeneration using UiO-66 nanomaterial in rabbit femurs[J]. BMC Vet. Res. 18 (1), 260 (2022).
Liu, F. et al. The potential therapeutic role of extracellular vesicles in critical-size bone defects: Spring of cell-free regenerative medicine is coming[J]. Front. Bioeng. Biotechnol. 11, 1050916 (2023).
Aldaadaa, A., Owji, N. & Knowles, J. Three-dimensional Printing in Maxillofacial Surgery: Hype versus Reality[J]. J. Tissue Eng. 9, 1544466979 (2018).
Wang, Z. & Yang, Y. Application of 3D Printing in Implantable Medical Devices[J]. Biomed. Res. Int., 2021:6653967. (2021).
Zhang, Y. et al. Bilayer Membrane Composed of Mineralized Collagen and Chitosan Cast Film Coated With Berberine-Loaded PCL/PVP Electrospun Nanofiber Promotes Bone Regeneration[J]. Front. Bioeng. Biotechnol. 9, 684335 (2021).
Xu, W. et al. Biodegradable calcium carbonate/mesoporous silica/poly(lactic-glycolic acid) microspheres scaffolds with osteogenesis ability for bone regeneration[J]. RSC Adv. 11 (9), 5055–5064 (2021).
Xu, W. et al. Storage and release of rare earth elements in microsphere-based scaffolds for enhancing osteogenesis[J]. Sci. Rep. 12 (1), 6383 (2022).
Song, R. et al. Current development of biodegradable polymeric materials for biomedical applications[J]. Drug Des. Devel Ther. 12, 3117–3145 (2018).
Shiroud Heidari, B. et al. Natural, synthetic and commercially-available biopolymers used to regenerate tendons and ligaments[J]. Bioact Mater. 19, 179–197 (2023).
Lin, Z. et al. Precisely controlled delivery of magnesium ions thru sponge-like monodisperse PLGA/nano-MgO-alginate core-shell microsphere device to enable in-situ bone regeneration[J]. Biomaterials 174, 1–16 (2018).
Bittner-Frank, M. et al. Cortical and trabecular mechanical properties in the femoral neck vary differently with changes in bone mineral density[J]. JBMR Plus. 8 (6), e49 (2024).
Smith, A. N. et al. Characterization of degradation kinetics of additively manufactured PLGA under variable mechanical loading paradigms[J]. J. Mech. Behav. Biomed. Mater. 153, 106457 (2024).
Xu, D. et al. Improvement of the mechanical properties and osteogenic activity of 3D-printed polylactic acid porous scaffolds by nano-hydroxyapatite and nano-magnesium oxide[J]. Heliyon 8 (6), e9748 (2022).
Owji, N. et al. Inclusion of calcium phosphate does not further improve in vitro and in vivo osteogenesis in a novel, highly biocompatible, mechanically stable and 3D printable polymer[J]. Sci. Rep. 12 (1), 16977 (2022).
Zhao, D. et al. Poly(lactic-co-glycolic acid)-based composite bone-substitute materials[J]. Bioactive Mater. 6 (2), 346–360 (2021).
Shen, M. et al. bFGF-Loaded Mesoporous Silica Nanoparticles Promote Bone Regeneration Through the Wnt/beta-Catenin Signalling Pathway[J]. Int. J. Nanomed. 17, 2593–2608 (2022).
Gong, S. et al. pH-Responsive Mesoporous Silica Nanoparticles Loaded with Naringin for Targeted Osteoclast Inhibition and Bone Regeneration[J]. Int. J. Nanomed. 19, 6337–6358 (2024).
Liang, W. et al. Current status of nano-embedded growth factors and stem cells delivery to bone for targeted repair and regeneration[J]. J. Orthop. Translat. 50, 257–273 (2025).
Budiman, A. et al. The Application of Mesoporous Silica Nanoparticles in Enhancing the Efficacy of Anti-Atherosclerosis Therapies: A Review[J]. Int. J. Nanomed. 20, 9825–9856 (2025).
Petousis, M. et al. Biomedical Composites of Polycaprolactone/Hydroxyapatite for Bioplotting: Comprehensive Interpretation of the Reinforcement Course[J]. Polym. (Basel), 16(17). (2024)
Chen, Y. & Zhao, Q. Innovative modification strategies and emerging applications of natural hydrogel scaffolds for osteoporotic bone defect regeneration[J]. Front. Bioeng. Biotechnol. 13, 1591896 (2025).
Shi, X. et al. Novel mesoporous silica-based antibiotic releasing scaffold for bone repair[J]. Acta Biomater. 5 (5), 1697–1707 (2009).
Liu, H. et al. Unlocking the potential of amorphous calcium carbonate: A star ascending in the realm of biomedical application[J]. Acta Pharm. Sin B. 14 (2), 602–622 (2024).
Munyemana, J. C. et al. Recombinant Collagen-Templated Biomineralized Synthesis of Biocompatible pH-Responsive Porous Calcium Carbonate Nanospheres[J]. ACS Omega. 8 (34), 30879–30887 (2023).
Xiao, S. et al. In situ comparison of osteogenic effects of polymer-based scaffolds with different degradability by integrated scaffold model[J]. Colloids Surf. B Biointerfaces. 241, 114047 (2024).
Si, Y. et al. Curcumin-encapsulated exosomes in bisphosphonate-modified hydrogel microspheres promote bone repair through macrophage polarization and DNA damage mitigation[J]. Mater. Today Bio. 32, 101874 (2025).
Wang, G. et al. Surface Functionalization of Hydroxyapatite Scaffolds with MgAlEu-LDH Nanosheets for High-Performance Bone Regeneration[J]. Adv. Sci. (Weinh). 10 (1), e2204234 (2022).
Shineh, G. et al. Biomolecule-functionalized dental implant surfaces: Towards augmenting soft tissue integration[J]. Bioact Mater. 53, 540–590 (2025).
Chen, Q. et al. Promoting implant osseointegration via the osteoblast-selective beta-amino acid polymer strategy[J]. Nat. Commun. 16 (1), 7190 (2025).
Kuten Pella, O. et al. Albumin as a Biomaterial and Therapeutic Agent in Regenerative Medicine[J]. Int. J. Mol. Sci., 23(18). (2022)
Shams, F. et al. A study on the cellular adhesion properties of a hybrid scaffold for vascular tissue engineering through molecular dynamics simulation[J]. Sci. Rep. 15 (1), 16433 (2025).
Moghaddam, A. S. et al. Hydrogels with multiple RGD presentations increase cell adhesion and spreading[J]. Acta Biomater. 199, 142–153 (2025).
Kim, J. et al. Effects of ECM protein mimetics on adhesion and proliferation of chorion derived mesenchymal stem cells[J]. Int. J. Med. Sci. 11 (3), 298–308 (2014).
Yan, B. et al. Surface modification progress for PLGA-based cell scaffolds. Polymers (Basel), 16(1), 165 (2024).
de Carvalho, A. B. et al. Personalized bioceramic grafts for craniomaxillofacial bone regeneration[J]. Int. J. Oral Sci. 16 (1), 62 (2024).
Zhou, J. et al. Study on the influence of scaffold morphology and structure on osteogenic performance[J]. Front. Bioeng. Biotechnol. 11, 1127162 (2023).
Huang, D. et al. Biomimetic structural design in 3D-printed scaffolds for bone tissue engineering[J]. Mater. Today Bio. 32, 101664 (2025).
Aydin, M. S. et al. Impact of Porosity and Stiffness of 3D Printed Polycaprolactone Scaffolds on Osteogenic Differentiation of Human Mesenchymal Stromal Cells and Activation of Dendritic Cells[J]. ACS Biomater. Sci. Eng. 10 (12), 7539–7554 (2024).
Zebaze, R. et al. Increased Cortical Porosity and Reduced Trabecular Density Are Not Necessarily Synonymous With Bone Loss and Microstructural Deterioration[J]. JBMR Plus. 3 (4), e10078 (2019).
Coburn, B. & Salary, R. R. Mechanical characterization of porous bone-like scaffolds with complex microstructures for bone regeneration. Bioengineering (Basel), 12(4), 416 (2025).
Zhang, B. et al. 3D-printed porous zinc scaffold combined with bioactive serum exosomes promotes bone defect repair in rabbit radius[J]. Aging (Albany NY). 16 (11), 9625–9648 (2024).
Matsushita, A. et al. Frequency-regulated repeated micro-vibration promotes osteoblast differentiation through BMP signaling in MC3T3-E1 cells. Life (Basel), 15(4), 588 (2025)
Sun, Y., Jia, X. & Meng, Q. Characteristic Evaluation of Recombinant MiSp/Poly(lactic-co-glycolic) Acid (PLGA) Nanofiber Scaffolds as Potential Scaffolds for Bone Tissue Engineering[J]. Int. J. Mol. Sci., 24(2). (2023).
Contreras-Caceres, R. et al. Electrospun nanofibers: Recent applications in drug delivery and cancer therapy. Nanomaterials (Basel), 9(4), 656 (2019)
Lian, M. et al. Bi-layered electrospun nanofibrous membrane with osteogenic and antibacterial properties for guided bone regeneration[J]. Colloids Surf. B Biointerfaces. 176, 219–229 (2019).
Gentili, C. et al. Sustainably cultured coral scaffold supports human bone marrow mesenchymal stromal cell osteogenesis[J]. Regen Ther. 26, 366–381 (2024).
Lun, D. et al. Limitations and modifications in the clinical application of calcium sulfate[J]. Front. Surg. 11, 1278421 (2024).
Viti, F. et al. Osteogenic Differentiation of MSC through Calcium Signaling Activation: Transcriptomics and Functional Analysis[J]. PloS one. 11 (2), e148173 (2016).
Cheng, L. et al. Black phosphorus-based 2D materials for bone therapy[J]. Bioactive Mater. 5 (4), 1026–1043 (2020).
Liu, T. et al. Customized Design 3D Printed PLGA/Calcium Sulfate Scaffold Enhances Mechanical and Biological Properties for Bone Regeneration[J]. Front. Bioeng. Biotechnol. 10, 874931 (2022).
Chen, X. et al. Mesoporous silica promotes osteogenesis of human adipose-derived stem cells identified by a high-throughput microfluidic chip assay. Pharmaceutics, 14(12), 2730 (2022)
Nii, T. & Katayama, Y. Biomaterial-assisted regenerative medicine. Int. J. Mol. Sci., 22(16), 8657 (2021).
Min, K. H. et al. Biomimetic scaffolds of calcium-based materials for bone regeneration. Biomimetics (Basel), 9(9), 511 (2024)
Funding
This study was supported by The National Natural Science Foundation of China (82160577); The Zunyi City Science & Technology Innovation Talent Project (No. [2024] 04).
Author information
Authors and Affiliations
Contributions
**Honghan Wu: ** Writing–original draft, Software, Methodology, Investigation.**Junxi Wu: ** Resources, Methodology, Formal analysis. **Hao Tang: ** Validation, Methodology. **Qian Zhong: ** Visualization, Data curation. **Guangquan Zhao: ** Visualization, Conceptualization. **Qianyu Xie: ** Software, Validation. **Weikang Xu: ** review & editing. **Qingde Wa: ** Writing - review & editing, Funding acquisition, Supervision, Conceptualization.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All experimental protocols of this study have been approved by the Medical Ethics Committee of Zunyi Medical University, and all methods were carried out in accordance with ARRIVE guidelines and regulations (the anesthesia method used in this study was intramuscular injection of xylazine hydrochloride).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, H., Wu, J., Tang, H. et al. Calcium-enriched mesoporous silica/PLGA scaffolds enhance bone repair in a rabbit femoral condylar defect model. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44490-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44490-9