Abstract
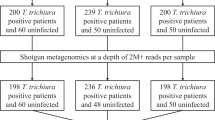
The mountain hare (Lepus timidus) is an arctic-alpine species with relictual populations in the Italian Alps, typically occurring at elevations above 2000 m a.s.l. This species is threatened by habitat loss and fragmentation, and declining snow cover due to climate warming. Moreover, as treelines shift upward, the European brown hare (L. europaeus) is expanding its distribution into areas previously dominated by the mountain hare, potentially leading to resource competition, and loss of local adaptation through hybridization and inter-specific gene flow. In particular, the consequences of sympatry on diversity and composition of prokaryote and fungal communities of the gut microbiota, which are critical to individual health, are currently unknown. Here, we compared the gut microbiota of these two hare species in an area of overlap in the central Alps by analysing fresh faecal pellets collected from Val Mazia/Matschertal, Italy along an elevational gradient (1000 to 2500 m a.s.l.). For the first time, we describe the prokaryote diversity and composition of L. timidus, and the fungal gut communities (mycobiota) of both Lepus species. Species identity was confirmed for 95 samples via mtDNA barcoding, while gut microbiota richness and composition were investigated using amplicon sequencing, targeting the V3-V4 region of the prokaryote 16S rRNA gene and fungal ITS2 regions. Distinct prokaryote and fungal communities were observed for each species, even where their distributions overlap, indicating differences in their functional diversity. Interestingly, for both Lepus species, elevation influenced fungal but not prokaryote diversity. Therefore, sympatry appears to have had minimal impact on gut microbiota composition of either species thus far. Given the expected upward range shift of L. europaeus under climate warming and its continued restocking for hunting, our findings provide an important baseline for assessing the health and adaptability of L. timidus as well as the effectiveness of conservation efforts aimed at protecting this species. However, expanding this research to other areas of sympatry will be essential to understand if gut microbiota composition is indicative of L. timidus conservation status across its range.
Similar content being viewed by others
Data availability
Sanger sequences have been deposited at NCBI GenBank with accession numbers PX122685—PX122779. Sanger sequences of the mitochondrial D-loop generated in our study were made available to the editor and reviewers with the uploaded file: Submission2992130.txt.gz. The raw amplicon-sequencing data has been deposited at NCBI Sequence Read Archive (SRA) under the BioProject ID PRJNA1304890. Reviewers can access BioProject and associated SRA metadata at [https://dataview.ncbi.nlm.nih.gov/object/PRJNA1304890?reviewer=rsuoanlfj6uo274kij1dik96rn].
References
Lenoir, J., Gégout, J. C., Marquet, P. A., de Ruffray, P. & Brisse, H. A significant upward shift in plant species optimum elevation during the 20th century. Science 320, 1768–1771 (2008).
Harsch, M. A., Hulme, P. E., McGlone, M. S. & Duncan, R. P. Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol. Lett. 12, 1040–1049 (2009).
Noce, S., Cipriano, C. & Santini, M. Altitudinal shifting of major forest tree species in Italian mountains under climate change. Front. For. Glob. Change 6, 1250651. https://doi.org/10.3389/ffgc.2023.1250651 (2023).
Hickling, R., Roy, D. B., Hill, J. K., Fox, R. & Thomas, C. D. The distributions of a wide range of taxonomic groups are expanding polewards. Glob. Change Biol. 12, 450–455 (2006).
Popy, S., Bordignon, L. & Prodon, R. A weak upward elevational shift in the distributions of breeding birds in the Italian Alps. J. Biogeogr. 37, 57–67 (2010).
Büntgen, U. et al. Elevational range shifts in four mountain ungulate species from the Swiss Alps. Ecosphere 8, e01761. https://doi.org/10.1002/ecs2.1761 (2017).
Schai-Braun, S. C., Jenny, H., Ruf, T. & Hackländer, K. Temperature increase and frost decrease driving upslope elevational range shifts in Alpine grouse and hares. Glob. Change Biol. 27, 6602–6614 (2021).
Rehnus, M. Der Schneehase in den Alpen: Ein Überlebenskünstler mit ungewisser Zukunft. Bristol-Stiftung, Zürich; Haupt, Bern (2013).
Stokes, A. W. et al. Altitude, latitude and climate zone as determinants of mountain hare (Lepus timidus) coat colour change. Ecol. Evol. 13, e10548. https://doi.org/10.1002/ece3.10548 (2023).
Sliwinski, K. et al. Habitat requirements of the European brown hare (Lepus europaeus Pallas 1778) in an intensively used agriculture region (Lower Saxony, Germany). BMC Ecol. 19, 31. https://doi.org/10.1186/s12898-019-0247-7 (2019).
Levänen, R., Thulin, C.-G., Spong, G. & Pohjoismäki, J. L. O. Widespread introgression of L. timidus genes into Fennoscandian brown hare populations. PLoS ONE 13, e0191790. https://doi.org/10.1371/journal.pone.0191790 (2018).
Jansson, G. & Pehrson, Å. The recent expansion of the brown hare (Lepus europaeus) in Sweden with possible implications to the mountain hare (L. timidus). Eur. J. Wildl. Res. 53, 125–130 (2007).
La Morgia, V. et al. Global warming is promoting the rapid invasion of the L. timidus range by the European hare in the Alps. Biodivers. Conserv. 32, 3875–3891 (2023).
Edwards, J. E. et al. Domesticated equine species and their derived hybrids differ in their fecal microbiota. Anim. Microbiome 2, 27. https://doi.org/10.1186/s42523-020-00027-7 (2020).
Wei, L. et al. Hybridization altered the gut microbiota of pigs. Front. Microbiol. 14, 1177947. https://doi.org/10.3389/fmicb.2023.1177947 (2023).
Rowland, I. et al. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 57, 1–24 (2018).
Sittipo, P. et al. The function of gut microbiota in immune-related neurological disorders: A review. J. Neuroinflammation 19, 154. https://doi.org/10.1186/s12974-022-02510-1 (2022).
Kasarello, K., Cudnoch-Jędrzejewska, A. & Czarzasta, K. Communication of gut microbiota and brain via immune and neuroendocrine signaling. Front. Microbiol. 14, 1118529. https://doi.org/10.3389/fmicb.2023.1118529 (2023).
Moeller, A. H. et al. Experimental evidence for adaptation to species-specific gut microbiota in house mice. mSphere 4, e00387-19. https://doi.org/10.1128/mSphere.00387-19 (2019).
Hauffe, H. C. & Barelli, C. Conserve the germs: The gut microbiota and adaptive potential. Conserv. Genet. 20, 19–27 (2019).
Rojas, C. A., Ramírez-Barahona, S., Holekamp, K. E. & Theis, K. R. Host phylogeny and host ecology structure the mammalian gut microbiota at different taxonomic scales. Anim. Microbiome 3, 33. https://doi.org/10.1186/s42523-021-00094-4 (2021).
Neha, S. A., Hanson, J. D., Wilkinson, J. E., Bradley, R. D. & Phillips, C. D. Impacts of host phylogeny, diet, and geography on the gut microbiome of rodents. PLoS ONE 20, e0316101. https://doi.org/10.1371/journal.pone.0316101 (2025).
Barelli, C. et al. The gut microbiota communities of wild arboreal and ground-feeding tropical primates are affected differently by habitat disturbance. mSystems 5, e00061-20. https://doi.org/10.1128/mSystems.00061-20 (2020).
Amato, K. R. et al. The gut microbiota appears to compensate for seasonal diet variation in the wild black howler monkey (Alouatta pigra). Microb. Ecol. 69, 434–443 (2015).
Stalder, G. L. et al. Gut microbiota of the European brown hare (Lepus europaeus). Sci. Rep. 9, 2738. https://doi.org/10.1038/s41598-019-39638-9 (2019).
Zhang, L., Zhan, H., Xu, W., Yan, S. & Ng, S. C. The role of gut mycobiome in health and diseases. Therap. Adv. Gastroenterol. 14, 17562848211047130. https://doi.org/10.1177/17562848211047130 (2021).
Zandonai, A. et al. Six years of high-resolution climatic data collected along an elevation gradient in the Italian Alps. Sci. Data 11, 751. https://doi.org/10.1038/s41597-024-03580-x (2024).
Hilpold, A. et al. Decline of rare and specialist species across multiple taxonomic groups after grassland intensification and abandonment. Biodivers. Conserv. 27, 3729–3744 (2018).
Praeg, N., Pauli, H. & Illmer, P. Microbial diversity in bulk and rhizosphere soil of Ranunculus glacialis along a high-alpine altitudinal gradient. Front. Microbiol. 10, 1429. https://doi.org/10.3389/fmicb.2019.01429 (2019).
Galla, G. et al. Mock community as an in situ positive control for amplicon sequencing of microbiotas from the same ecosystem. Sci. Rep. 13, 4056. https://doi.org/10.1038/s41598-023-30916-1 (2023).
Wilkinson, G. S. & Chapman, A. M. Length and sequence variation in evening bat D-loop mtDNA. Genetics 128, 607–617 (1991).
Melo-Ferreira, J. et al. The rise and fall of Lepus timidus during Pleistocene glaciations: Expansion and retreat with hybridization in the Iberian Peninsula. Mol. Ecol. 16, 605–618 (2007).
Pecchioli, E. Variabilità e struttura genetica in Lepus timidus: aspetti micro e macrogeografici, filogenetici e adattativi (Unpublished PhD thesis, Università degli Studi di Ferrara) (2010).
Zachos, F. E., Ben Slimen, H., Hackländer, K., Giacometti, M. & Suchentrunk, F. Regional genetic in situ differentiation despite phylogenetic heterogeneity in Alpine Lepus timidus. J. Zool. 282, 47–53 (2010).
Kumar, S., Stecher, G., Li, M., Knyaz, C. & Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547–1549 (2018).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 41, e1 (2013).
Walters, W. et al. Improved prokaryotic 16S rRNA gene (V4 and V4-5) and fungal internal transcribed spacer marker gene primers for microbial community surveys. mSystems 1, e00009-15. https://doi.org/10.1128/mSystems.00009-15 (2015).
Ihrmark, K. et al. New primers to amplify the fungal ITS2 region – Evaluation by 454-sequencing of artificial and natural communities. FEMS Microbiol. Ecol. 82, 666–677 (2012).
White, T. J., Bruns, T., Lee, S. & Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications (eds Innis, M. A. et al.) 315–322 (Academic Press, 1990).
Callahan, B. J. et al. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 13, 581–583 (2016).
Quast, C. et al. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 41, D590–D596 (2012).
Nilsson, R. H. et al. The UNITE database for molecular identification of fungi: Handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res. 47, D259–D264 (2019).
Bodenhofer, U., Bonatesta, E., Horejs-Kainrath, C. & Hochreiter, S. Msa: An R package for multiple sequence alignment. Bioinformatics 31, 3997–3999 (2015).
Schliep, K. P. Phangorn: Phylogenetic analysis in R. Bioinformatics 27, 592–593 (2011).
Davis, N. M., Proctor, D. M., Holmes, S. P., Relman, D. A. & Callahan, B. J. Simple statistical identification and removal of contaminant sequences in marker-gene and metagenomics data. Microbiome 6, 226. https://doi.org/10.1186/s40168-018-0605-2 (2018).
McMurdie, P. J. & Holmes, S. Phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8, e61217. https://doi.org/10.1371/journal.pone.0061217 (2013).
Liu, C., Cui, Y., Li, X. & Yao, M. microeco: An R package for data mining in microbial community ecology. FEMS Microbiol. Ecol. 97, fiaa255 (2021).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag, 2016).
Neuwirth, E. RColorBrewer: ColorBrewer Palettes. R package version 1.1-3, Available at https://CRAN.R-project.org/package=RColorBrewer (2022).
Garnier, S. et al. viridis(Lite)—Colorblind-Friendly Color Maps for R. R package version 0.6.5. Available at: https://sjmgarnier.github.io/viridis/ (2024).
Segata, N. et al. Metagenomic biomarker discovery and explanation. Genome Biol. 12, R60. https://doi.org/10.1186/gb-2011-12-6-r60 (2011).
Oksanen, J. et al. vegan: Community Ecology Package. R package version 2.6–4, Available at https://CRAN.R-project.org/package=vegan (2022).
Douglas, G. M. et al. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 38, 685–688 (2020).
Metsalu, T. & Vilo, J. ClustVis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic Acids Res. 43, W566–W570 (2015).
The GIMP Development Team. GIMP (Version 2.10.18) [Computer software]. https://www.gimp.org (2020).
Tuddenham, S. & Sears, C. L. The intestinal microbiome and health. Curr. Opin. Infect. Dis. 28, 464–470 (2015).
Zhan, G. et al. Abnormal gut microbiota composition contributes to cognitive dysfunction in SAMP8 mice. Aging 10, 1257–1267. https://doi.org/10.18632/aging.101464 (2018).
Martínez-Haro, M., Triadó-Margarit, X., Mateo, R., Viñuela, J. A. & Casamayor, E. O. Taxonomic and functional fingerprints in the gut microbiota of Iberian hare populations (Lepus granatensis) inhabiting areas with organic and conventional farming. Environ. Pollut. 369, 125862 (2025).
Stothart, M. R. et al. Population dynamics and the microbiome in a wild boreal mammal: The snowshoe hare cycle and impacts of diet, season and predation risk. Mol. Ecol. 34, e17629. https://doi.org/10.1111/mec.17629 (2025).
Cao, Z. et al. Comparison and association of winter diets and gut microbiota using trnL and 16S rRNA gene sequencing for three herbivores in Taohongling, China. Glob. Ecol. Conserv. 53, e03041. https://doi.org/10.1016/j.gecco.2024.e03041 (2024).
Koskella, B. & Bergelson, J. The study of host–microbiome (co)evolution across levels of selection. Philos. Trans. R. Soc. B Biol. Sci. 375, 20190604 (2020).
Bock, A. Lepus europaeus (Lagomorpha: Leporidae). Mamm. Species 52, 125–142. https://doi.org/10.1093/mspecies/seaa010 (2020).
Angerbjörn, A. & Flux, J. E. C. Lepus timidus. Mamm. Species 495, 1–11 (1995).
Ley, R. E. et al. Evolution of mammals and their gut microbes. Science 320, 1647–1651 (2008).
Knowles, S. C. L., Eccles, R. M. & Baltrūnaitė, L. Species identity dominates over environment in shaping the microbiota of small mammals. Ecol. Lett. 22, 826–837 (2019).
Anders, J. L. et al. Comparing the gut microbiome along the gastrointestinal tract of three sympatric species of wild rodents. Sci. Rep. 11, 19929. https://doi.org/10.1038/s41598-021-99379-6 (2021).
Reichlin, T., Klansek, E. & Hackländer, K. Diet selection by hares (Lepus europaeus) in arable land and its implications for habitat management. Eur. J. Wildl. Res. 52, 109–118 (2006).
Schai-Braun, S. C. et al. The European hare (Lepus europaeus): A picky herbivore searching for plant parts rich in fat. PLoS ONE 10, e0134278. https://doi.org/10.1371/journal.pone.0134278 (2015).
Schai-Braun, S. C. et al. Diet niches of the Alpine Lepus timidus (Lepus timidus varronis) and European hare (Lepus europaeus) living in coexistence in the Alps. Mamm. Biol. 105, 139–145 (2025).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Carmody, R. N. et al. Diet dominates host genotype in shaping the murine gut microbiota. Cell Host Microbe 17, 72–84 (2015).
Alessandri, G., Rizzo, S. M., Ossiprandi, M. C., van Sinderen, D. & Ventura, M. Creating an atlas to visualize the biodiversity of the mammalian gut microbiota. Curr. Opin. Biotechnol. 73, 28–33 (2022).
Zoetendal, E. G. & de Vos, W. M. Effect of diet on the intestinal microbiota and its activity. Curr. Opin. Gastroenterol. 30, 189–195 (2014).
Sommer, F. et al. The gut microbiota modulates energy metabolism in the hibernating brown bear Ursus arctos. Cell Rep. 14, 1655–1661 (2016).
Xu, Z. et al. Cold-induced lipid dynamics and transcriptional programs in white adipose tissue. BMC Biol. 17, 74. https://doi.org/10.1186/s12915-019-0693-x (2019).
Vázquez-Baeza, Y. et al. Dog and human inflammatory bowel disease rely on overlapping yet distinct dysbiosis networks. Nat. Microbiol. 1, 16177 (2016).
Lupp, C. et al. Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe 2, 119–129 (2007).
Shin, N. R., Whon, T. W. & Bae, J.-W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 33, 496–503 (2015).
Praeg, N. et al. The role of land management and elevation in shaping soil microbial communities: Insights from the Central European Alps. Soil Biol. Biochem. 150, 107951 (2020).
Mouhamadou, B. et al. Differences in fungal communities associated to Festuca paniculata roots in subalpine grasslands. Fungal Divers. 47, 55–63 (2011).
Irwin, N. A., Twynstra, C. S., Mathur, V. & Keeling, P. J. The molecular phylogeny of Chionaster nivalis reveals a novel order of psychrophilic and globally distributed Tremellomycetes (Fungi, Basidiomycota). PLoS ONE 16, e0247594. https://doi.org/10.1371/journal.pone.0247594 (2021).
Stoppiello, G. A. et al. Seasonality is the main determinant of microbial diversity associated to snow/ice around Concordia station on the Antarctic polar plateau. Biology 12, 1193. https://doi.org/10.3390/biology12091193 (2023).
Kurtzman, C. P., Fell, J. W. & Boekhout, T. The Yeasts: A Taxonomic Study. Fifth Edition (2011).
Olsen, L., Thum, E. & Rohner, N. Lipid metabolism in adaptation to extreme nutritional challenges. Dev. Cell https://doi.org/10.1016/j.devcel.2021.02.024 (2021).
Bauer, R., Begerow, D. & Oberwinkler, F. Ustilaginomycotina: The true smut fungi. Tree of Life Web Project Available at: http://tolweb.org/Ustilaginomycotina/20530/2008.01.23 (2006).
Murphy, M. A., Waits, L. E. & Kendall, K. C. Quantitative evaluation of fecal drying methods for brown bear DNA analysis. Wildl. Soc. Bull. 28, 951–957 (2000).
Elyasigorji, Z., Izadpanah, M., Hadi, F. & Zare, M. Mitochondrial genes as strong molecular markers for species identification. Nucleus 66, 81–93. https://doi.org/10.1007/s13237-022-00393-4 (2022).
Thulin, C.-G., Stone, J., Tegelström, H. & Walker, C. W. Species assignment and hybrid identification among Scandinavian hares Lepus europaeus and L. timidus. Wildl. Biol. 12, 29–38 (2006).
Schai-Braun, S. C., Schwienbacher, S., Smith, S. & Hackländer, K. Coexistence of European hares and Alpine mountain hares in the Alps: What drives the occurrence and frequency of their hybrids?. J. Zool. 320(1), 15–25 (2023).
Wang, J. et al. Analysis of intestinal microbiota in hybrid house mice reveals evolutionary divergence in a vertebrate hologenome. Nat. Commun. 6, 6440. https://doi.org/10.1038/ncomms7440 (2015).
Huggett, M. J. et al. Gut microbial communities of hybridising pygmy angelfishes reflect species boundaries. Commun. Biol. 6, 542 (2023).
Wei, L. et al. Hybridization alters red deer gut microbiome and metabolites. Front. Microbiol. 15, 1387957 (2024).
Suzuki, T. A., Martins, F. M. & Nachman, M. W. Altitudinal variation of the gut microbiota in wild house mice. Mol. Ecol. 28, 2378 (2019).
Li, H., Zhou, R., Zhu, J., Huang, X. & Qu, J. Environmental filtering increases with elevation for the assembly of gut microbiota in wild pikas. Microb. Biotechnol. https://doi.org/10.1111/1751-7915.13450 (2019).
Wang, X. et al. High-altitude drives the convergent evolution of alpha diversity and indicator microbiota in the gut microbiomes of ungulates. Front. Microbiol. https://doi.org/10.3389/fmicb.2022.953234 (2022).
Zhao, J. et al. Diet and high altitude strongly drive convergent adaptation of gut microbiota in wild macaques, humans, and dogs to high altitude environments. Front. Microbiol. https://doi.org/10.3389/fmicb.2023.1067240 (2023).
Perrotti, A. G. & van Asperen, E. Dung fungi as a proxy for megaherbivores: Opportunities and limitations for archaeological applications. Veg. Hist. Archaeobot. 28, 93–104 (2019).
Tang, X. et al. Stochastic processes derive gut fungi community assembly of plateau pikas (Ochotona curzoniae) along altitudinal gradients across warm and cold seasons. J. Fungi. https://doi.org/10.3390/jof9101032 (2023).
Auchtung, T. A. et al. Investigating colonization of the healthy adult gastrointestinal tract by fungi. mSphere https://doi.org/10.1128/mSphere.00092-18 (2018).
Suhr, M. J. & Hallen-Adams, H. E. The human gut mycobiome: Pitfalls and potentials—A mycologist’s perspective. Mycologia 107, 1057–1073 (2015).
Li, J. et al. Pattern and drivers of soil fungal community along elevation gradient in the Abies georgei forests of Segila mountains, Southeast Tibet. Glob. Ecol. Conserv. 39, e02446. https://doi.org/10.1016/j.gecco.2022.e02291 (2022).
Vélez-Martínez, G. A. et al. Soil bacteria and fungi communities are shaped by elevation influences in Colombian forest and páramo natural ecosystems. Int. Microbiol. 27, 377–391 (2024).
Dean, R. et al. The top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 13, 414–430 (2012).
Rodriguez, R. J., White, J. F. Jr., Arnold, A. E. & Redman, R. S. Fungal endophytes: Diversity and functional roles. New Phytol. 182, 314–330 (2009).
Semchenko, M. et al. Fungal diversity regulates plant-soil feedbacks in temperate grassland. Sci. Adv. https://doi.org/10.1126/sciadv.aau4578 (2018).
Grieneisen, L. E. et al. Genes, geology and germs: Gut microbiota across a primate hybrid zone are explained by site soil properties, not host species. Proc. R. Soc. B Biol. Sci. 286, 20190431 (2019).
Banerjee, S. & van der Heijden, M. G. A. Soil microbiomes and one health. Nat. Rev. Microbiol. 21, 6–20 (2023).
Acknowledgements
The authors wish to thank the Fondazione E. Mach for access to facilities, and the staff of the Sequencing and Genotyping Platform for their outstanding support.
Funding
The EUREGIO project: MICROVALU—Evaluating microbiodiversity in alpine pastures (Project ID: IPN94), awarded to PI, JS and HCH, was funded by the “Euregio Tirolo-Alto Adige-Trentino” Interregional Project Network. This study was also partially carried out with funding to HCH at the Fondazione E. Mach (Project BIOALPEC) under the National Biodiversity Future Centre (NBFC) Project (code CN_00000033, Concession Decree No. 1034 of 17 June 2022 adopted by the Italian Ministry of University and Research, CUPD43C22001280006), funded under the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4—Call for tender No. 3138 of 16 December 2021, rectified by Decree n.3175 of 18 December 2021 of Italian Ministry of University and Research funded by the European Union—NextGenerationEU. This manuscript reflects only the authors’ views and opinions, neither the European Union nor the European Commission can be considered responsible for them.
Author information
Authors and Affiliations
Contributions
B.C., H.C.H., and G.G. conducted the sampling. L.M., B.C. and G.G. completed the laboratory analyses. Data analyses were provided by L.M. and G.G., with support from N.P., T.R., J.S., P.I. and F.N.M.; L.M., H.C.H. and G.G. drafted the manuscript. All authors edited and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Marinangeli, L., Crestanello, B., Praeg, N. et al. Sympatric Lepus spp. in the central Italian Alps host significantly different gut microbiotas. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44592-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44592-4