Abstract
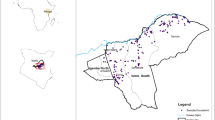
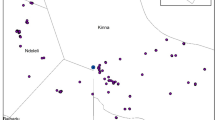
Rift Valley fever (RVF) is a zoonotic arbovirus, and livestock cases are often underreported in endemic countries due to reliance on passive clinical surveillance. During the 2023–2024 El Niño event, Kenya experienced widespread flooding, but no RVF outbreaks were reported in the southern regions. We implemented slaughterhouse-based surveillance in southern Kenya from May 2023—June 2024, using five consecutive cross-sectional surveys. Cattle, sheep, and goats were tested for anti-RVFV IgG and IgM antibodies, with concurrent recording of post-mortem lesions. Using age estimates from dentition, catalytic models estimated the force of infection (FOI) over time and spatial analysis assessed the Loitokitok sub-county for hotspots. Among 955 animals, 10.2% were IgG-positive, with seroprevalence and FOI increasing after El Niño rains, reaching 22.6% by May 2024. Six animals (0.6%) were IgM-positive, indicating recent infection, with cases detected in 3/5 sampling periods, including before the rains. All recently infected IgM-positive animals were deemed fit for slaughter and none had lesions. Adult animals in endemic areas, void of clinical and pathological signs, may therefore play a role in the silent spread and maintenance of RVFV. Slaughterhouse-based surveillance offers a practical and scalable platform for improving RVF detection and monitoring livestock in endemic regions.
Similar content being viewed by others
Data availability
The dataset used to generate this analysis is available at https://doi.org/10.5281/zenodo.17078167. Other associated data, including the georeferenced locations, is available on reasonable request by writing to the corresponding author.
References
Anyangu, A. S. et al. Risk factors for severe Rift Valley fever infection in Kenya, 2007. Am. J. Trop. Med. Hyg. 83, 14–21 (2010).
Pepin, M., Bouloy, M., Bird, B. H., Kemp, A. & Paweska, J. Rift Valley fever virus (Bunyaviridae: Phlebovirus): An update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. Vet. Res. 41, 61. https://doi.org/10.1051/vetres/2010033 (2010).
O’Neill, L., Gubbins, S., Reynolds, C., Limon, G. & Giorgakoudi, K. The socioeconomic impacts of Rift Valley fever: A rapid review. PLoS Negl. Trop. Dis. 18, e0012347. https://doi.org/10.1371/journal.pntd.0012347 (2024).
Golnar, A. J., Turell, M. J., LaBeaud, A. D., Kading, R. C. & Hamer, G. L. Predicting the mosquito species and vertebrate species involved in the theoretical transmission of Rift Valley fever virus in the United States. PLoS Negl. Trop. Dis. 8, 17–19 (2014).
Bird, B. H. & Nichol, S. T. Breaking the chain: Rift Valley fever virus control via livestock vaccination. Curr. Opin. Virol. 2, 315–323 (2012).
WHO. Rift Valley fever Fact Sheet. https://www.who.int/news-room/fact-sheets/detail/rift-valley-fever (2018).
Lichoti, J. K. et al. Detection of Rift Valley fever virus interepidemic activity in some hotspot areas of Kenya by sentinel animal surveillance, 2009–2012. Vet. Med. Int. 2014, 379010 (2014).
LaBeaud, A. D. et al. Interepidemic Rift Valley fever virus seropositivity, northeastern Kenya. Emerg. Infect. Dis. 14, 1240–1246 (2008).
Mbotha, D. et al. Inter-epidemic Rift Valley fever virus seroconversions in an irrigation scheme in Bura, south-east Kenya. Transbound. Emerg. Dis. 65, e55–e62 (2017).
Situma, S. et al. Widening geographic range of Rift Valley fever disease clusters associated with climate change in East Africa. BMJ Glob. Health https://doi.org/10.1136/bmjgh-2023-014737 (2024).
Manore, C. A. & Beechler, B. R. Inter-epidemic and between-season persistence of Rift Valley fever: Vertical transmission or cryptic cycling?. Transbound. Emerg. Dis. 62, 13–23 (2015).
Lumley, S. et al. Rift valley fever virus: Strategies for maintenance, survival and vertical transmission in mosquitoes. J. Gen. Virol. 98, 875–887. https://doi.org/10.1099/jgv.0.000765 (2017).
Oyas, H. et al. Enhanced surveillance for Rift Valley Fever in livestock during El Niño rains and threat of RVF outbreak, Kenya, 2015-2016. PLoS Negl. Trop. Dis. https://doi.org/10.1371/journal.pntd.0006353 (2018).
Kivaria, F. et al. Has the epidemiologic conundrum of Rift Valley fever changed?. Trop. Anim. Health Prod. https://doi.org/10.1007/s11250-025-04531-3 (2025).
Hassan, A. et al. Epidemiological investigation of a Rift Valley fever outbreak in humans and livestock in Kenya, 2018. Am. J. Trop. Med. Hyg. 103, 1649–1655 (2020).
Gerken, K. N. et al. Paving the way for human vaccination against Rift Valley Fever Virus: A systematic literature review of RVFV epidemiology from 1999 to 2021. PLoS Negl. Trop. Dis. https://doi.org/10.1371/journal.pntd.0009852 (2022).
Grossi-Soyster, E. N. & LaBeaud, A. D. Rift Valley fever: Important considerations for risk mitigation and future outbreaks. Trop. Med. Infect. Dis. 5, 25–31 (2020).
Sharan, M., Vijay, D., Yadav, J. P., Bedi, J. S. & Dhaka, P. Surveillance and response strategies for zoonotic diseases: a comprehensive review. Sci. One Health 2, 100050. https://doi.org/10.1016/j.soh.2023.100050 (2023).
Majiwa, H., Bukachi, S. A., Omia, D. & Fèvre, E. M. Knowledge, perceptions, and practices around zoonotic diseases among actors in the livestock trade in the Lake Victoria crescent ecosystem in East Africa. Front. Public Health https://doi.org/10.3389/fpubh.2023.1199664 (2023).
Nyariki, D. M. & Amwata, D. A. The value of pastoralism in Kenya: Application of total economic value approach. Pastoralism 9, 9 (2019).
FAO. Guidelines for the Enumeration of Nomadic and Semi-Nomadic (Transhumant) Livestock. https://www.developmentaid.org/api/frontend/cms/file/2021/08/ca6397en.pdf (2016).
de Glanville, W. A. et al. Inter-epidemic Rift Valley fever virus infection incidence and risks for zoonotic spillover in northern Tanzania. PLoS Negl. Trop. Dis. https://doi.org/10.1371/journal.pntd.0010871 (2022).
Kenyalaw. Meat Control Act. Kenya Law Report (2012).
Fèvre, E. M. et al. Slaughter facilities in East Africa as a focus for one health. One Health Cases https://doi.org/10.1079/onehealthcases.2023.0020 (2023).
Affroze, S. et al. Risk factors and gross pathology of bovine liver fluke infection at Netrokona District, Bangladesh. J. Anim. Sci. Adv. 3, 83 (2013).
Kithuka, J. M., Maingi, N., Njeruh, F. M. & Ombui, J. N. The prevalence and economic importance of bovine fasciolosis in Kenya–an analysis of abattoir data. Onderstepoort J. Vet. Res. 69, 255–262 (2002).
Swai, E. S. & Ulicky, E. An evaluation of the economic losses resulting from condemnation of cattle livers and loss of carcass weight due to Fasciolosis: A case study from Hai town abattoir, Kilimanjaro region, Tanzania. Livest. Res. Rural Dev. 21, 186 (2009).
Corbel, M. J. Brucellosis in Humans and Animals. (World Health Organization, 2006).
Odendaal, L., Davis, A. S., Fosgate, G. T. & Clift, S. J. Lesions and cellular tropism of natural Rift Valley fever virus infection in young lambs. Vet. Pathol. 57, 66–81 (2020).
van den Bergh, C., Venter, E. H., Swanepoel, R. & Thompson, P. N. High seroconversion rate to Rift Valley fever virus in cattle and goats in far northern KwaZulu-Natal, South Africa, in the absence of reported outbreaks. PLoS Negl. Trop. Dis. 13, 1–19 (2019).
Fao Igad. FAO AND IGAD ALERT FOR EASTERN AFRICA TO INCREASE VIGILANCE FOR RIFT VALLEY FEVER. (2023).
Maurice Owiny, K. F. E. and L. T. P. (FELTP). Investigation of Rift Valley Fever (RVF) outbreak in Wajir and Marsabit Counties. FELTP (2024).
Gerken, K. N. et al. Leveraging livestock movements to urban slaughterhouses for wide-spread Rift Valley fever virus surveillance in Western Kenya. One Health 15, 100457 (2022).
Gerken, K. N. et al. Expanding understanding of urban Rift Valley fever risk and associated vector ecology at slaughterhouses in Kisumu, Kenya. Pathogens https://doi.org/10.3390/pathogens13060488 (2024).
Muturi, M. et al. Ecological and subject-level drivers of interepidemic Rift Valley fever virus exposure in humans and livestock in Northern Kenya. Sci. Rep. https://doi.org/10.1038/s41598-023-42596-y (2023).
Souza, T. M. L. et al. Emergence of the East-Central-South-African genotype of Chikungunya virus in Brazil and the city of Rio de Janeiro may have occurred years before surveillance detection. Sci. Rep. https://doi.org/10.1038/s41598-019-39406-9 (2019).
ten Bosch, Q. A. et al. Contributions from the silent majority dominate dengue virus transmission. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1006965 (2018).
de Glanville, W. A. et al. An outbreak of Rift Valley fever among peri-urban dairy cattle in northern Tanzania. Trans. R. Soc. Trop. Med. Hyg. 116, 1082–1090 (2022).
Thomas, K. M. et al. Prospective cohort study reveals unexpected aetiologies of livestock abortion in northern Tanzania. Sci. Rep. https://doi.org/10.1038/s41598-022-15517-8 (2022).
Alarcon, P. et al. Mapping of beef, sheep and goat food systems in Nairobi — A framework for policy making and the identification of structural vulnerabilities and deficiencies. Agric. Syst. 152, 1–17 (2017).
Wichgers Schreur, P. J. et al. Co-housing of Rift Valley fever virus infected lambs with immunocompetent or immunosuppressed lambs does not result in virus transmission. Front. Microbiol. 7, 1–9 (2016).
Bird, B. H., Ksiazek, T. G., Nichol, S. T. & Maclachlan, N. J. Rift Valley Fever Virus. JAVMA 234, 883–893 (2009).
World Health Organization, F. and A. O. of the U. N. W. O. for A. H. Rift Valley Fever: A Manual for Veterinary and Public Health Workers. (2010).
Rodarte, K. A. et al. A scoping review of zoonotic parasites and pathogens associated with abattoirs in Eastern Africa and recommendations for abattoirs as disease surveillance sites. Front. Public Health 11, 1194964. https://doi.org/10.3389/fpubh.2023.1194964 (2023).
Hill, A. et al. A risk and benefit assessment for visual-only meat inspection of UK Indoor and Outdoor Pigs. Food Control 30, 255–264 (2013).
Kahariri, S. et al. The evolution of Kenya’s animal health surveillance system and its potential for efficient detection of zoonoses. Front. Vet. Sci. 11, 1379907 (2024).
de Glanville, W. A. et al. An outbreak of Rift Valley fever among peri-urban dairy cattle in northern Tanzania. Trans. R. Soc. Trop. Med. Hyg. 116, 1082–1090 (2022).
World Meteorological organization. El Niño weakens but impacts continue. (2024).
Campbell, D. J., Lusch, D. P., Smucker, T. Wangui, E. E. LUCID’s Land Use Change Analysis as an Approach for Investigating Biodiversity Loss and Land Degradation Project Root Causes of Land Use Change in the Loitokitok Area, Kajiado D Strict, Kenya LUCID Working Pa Series Number: 19 i Per. www.lucideastafrica.org (2003).
Kenya Markets Trust. The Economics of Modernising the Meat Industry in Kenya: Untapped Potential of Meat Industry In. www.kenyamarkets.org (2020).
Kenya Law Reports. Meat Control Act, Revised Edition. www.kenyalaw.org (2012).
Clark, M. H. A., Warimwe, G. M., Di Nardo, A., Lyons, N. A. & Gubbins, S. Systematic literature review of Rift Valley fever virus seroprevalence in livestock, wildlife and humans in Africa from 1968 to 2016. PLoS Negl. Trop. Dis. 12, 1–19 (2018).
Bird, B. H., Bawiec, D. A., Ksiazek, T. G., Shoemaker, T. R. & Nichol, S. T. Highly sensitive and broadly reactive quantitative reverse transcription-PCR assay for high-throughput detection of Rift Valley fever virus. J. Clin. Microbiol. 45, 3506–3513 (2007).
Juma, J. et al. Using multiplex amplicon PCR technology to efficiently and timely generate Rift Valley Fever virus sequence data for genomic surveillance. Viruses https://doi.org/10.3390/v15020477 (2023).
Onwongá, A. A. et al. Genome characterization of Rift Valley fever virus isolated from cattle, goats and sheep during interepidemic periods in Kenya. BMC Vet. Res. 20, 376 (2024).
Heisey, D. M., Joly, D. O. & Messier, F. The fitting of general force-of-infection models to wildlife disease prevalence data. Ecology 87, 2356–2365 (2006).
Acknowledgements
We thank the slaughterhouse employees for accommodating our sampling within their routines. We also appreciate the owners of the livestock for allowing us to sample their animals and time providing the origin information. We are grateful to Mr Issac Nayayio, Mr. James Kayiaa, Ms. Cecelia Parsae, and Ms. Terry (Ivey) Lekanayi for their contributions to sampling and laboratory work. We also thank Ms. Elly Wallis, Programme Manager of the Liverpool-Wellcome Clinical PhD Fellowship, for her invaluable logistical support.
Funding
This study has been funded by KNG’s Liverpool—Wellcome Trust Clinical PhD fellowship [223502/Z/21/Z]. Field activities also benefited from support of the “Loitoktiok One Health Initiative”, which was part of the CGIAR Initiative on One Health, supported by contributors to the CGIAR Trust Fund (https://www.cgiar.org/funders).
Author information
Authors and Affiliations
Contributions
K.N.G, E.M.F, M.B, and AS conceived the study. Methodology was developed by K.N.G, V.M, M.B, E.M.F, A.S, E.A.J.C, and F.S. V.M, A.R, and R.R.O curated the data. Formal analysis, including laboratory work, was carried out by K.N.G, A.K, and R.M. K.N.G developed the software and prepared the visualizations. K.N.G wrote the original draft of the manuscript, and all authors reviewed and edited the manuscript. E.M.F, M.B, and A.S supervised the work. Funding was acquired by K.N.G. Project administration was managed by K.N.G, R.R.O, and A.R. Resources were provided by E.M.F. M.B was responsible for validation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gerken, K.N., Rereu, A., Mutai, V. et al. Unreported Rift Valley fever virus circulation during 2023–2024 El Niño event detected by slaughterhouse-based surveillance in southern Kenya. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44706-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44706-y