Abstract
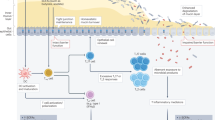
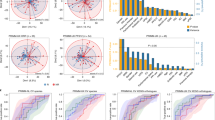
The gut microbiota influences immune checkpoint inhibitors (ICIs) efficacy. Microbiome-modulating strategies (MMSs), including probiotics, synbiotics, and faecal microbiota transplantation (FMT), have emerged as promising adjuncts, but their clinical impact remains uncertain. We systematically reviewed PubMed, Embase, and CENTRAL to February 2025 for clinical cohorts evaluating MMS in cancer patients receiving ICIs. Thirty-six studies (25 trials/cohorts; n = 2,746) were included. Meta-analyses, and subgroup analyses were performed for efficacy along with microbiome shifts and safety. MMS plus ICIs achieved a pooled objective response rate (ORR) of 40% (95% CI: 31%–49%; I² = 63.4%; p = 0.0003; 95% PI: 15%–72%). Descriptive proportions showed ORR of 45% (95% CI: 32%–58%; I² = 72.5%; p = 0.0058) for probiotics and 33% (95% CI: 22%–48%; I² = 60.7%; p = 0.0064) for FMT; however, these findings are non-comparative and confounded by study differences. Exploratory subgroup signals were noted for probiotics in NSCLC (ORR 55%; 95%CI: 45%–64%; I² = 0%; p = 0.3683) and FMT in melanoma (ORR 39%; 95% CI: 15%–69%; I² = 72.5%; p = 0.0262). Dual ICI regimens showed the highest point estimate for ORR (43%; 95% CI: 17%–73%; I² = 68.5%; p = 0.0747) but increased toxicity. Microbiome analyses revealed enrichment of short-chain fatty acid-producing taxa and Bifidobacterium spp. among responders. Based on a limited pooled sample size (n = 143), MMS-related adverse events were mostly grade 1–2 (42%; 95% CI: 14%–77%, I² = 53.8%, p = 0.0210), with rare severe events (1%). Overall, MMS show promising, though preliminary, hypothesis-generating signals for modulating ICI response. Given high heterogeneity and reliance on early-phase, single-arm trials, the findings underscore urgent need for large, biomarker-driven randomized controlled trials to define optimal interventions and cautiously integrate microbiome modulation into immuno-oncology care.
Similar content being viewed by others
Data availability
The data are available as supplementary files.
Abbreviations
- CENTRAL:
-
Cochrane central register of controlled trials
- CIs:
-
Confidence intervals
- PRISMA-P:
-
Preferred reporting items for systematic review and meta-analysis protocols
- RCTs:
-
Randomized controlled trials
- US:
-
United States
References
Siegel, R. L., Miller, K. D., Fuchs, H. E. & Jemal, A. Cancer statistics, 2022. CA. Cancer J. Clin. https://doi.org/10.3322/caac.21708 (2022).
Bagchi, S., Yuan, R. & Engleman, E. G. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Annu. Rev. Pathol. 16 (1), 223–249. https://doi.org/10.1146/annurev-pathol-042020-042741 (2021).
Lao, Y., Shen, D., Zhang, W., He, R. & Jiang, M. Immune checkpoint inhibitors in cancer therapy-how to overcome drug resistance?. Cancers (Basel) https://doi.org/10.3390/cancers14153575 (2022).
Mamdani, H., Matosevic, S., Khalid, A. B., Durm, G. & Jalal, S. I. Immunotherapy in lung cancer: current landscape and future directions. Front. Immunol. 13, 823618. https://doi.org/10.3389/fimmu.2022.823618 (2022).
Dobosz, P., Stępień, M., Golke, A. & Dzieciątkowski, T. Challenges of the immunotherapy: perspectives and limitations of the immune checkpoint inhibitor treatment. Int. J. Mol. Sci. 23 (5), 2847. https://doi.org/10.3390/ijms23052847 (2022).
Hiltbrunner, S. et al. Acquired resistance to anti-PD1 therapy in patients with NSCLC associates with immunosuppressive T cell phenotype. Nat. Commun. 14 (1), 5154. https://doi.org/10.1038/s41467-023-40745-5 (2023).
Mariniello, A. et al. Primary and Acquired Resistance to Immunotherapy with Checkpoint Inhibitors in NSCLC: From Bedside to Bench and Back. BioDrugs 39 (2), 215–235. https://doi.org/10.1007/s40259-024-00700-2 (2025).
He, R. et al. Dysbiosis and extraintestinal cancers. J. Experimental Clin. Cancer Res. 44 (1), 44. https://doi.org/10.1186/s13046-025-03313-x (2025).
Kumari, S., Srilatha, M. & Nagaraju, G. P. Effect of Gut Dysbiosis on Onset of GI Cancers. Cancers 17 (1), 90 (2025). https://www.mdpi.com/2072-6694/17/1/90
Sarkar, J. et al. Fluctuations in gut microbiome composition during immune checkpoint inhibitor therapy. World J. Oncol. 14 (3), 178. https://doi.org/10.14740/wjon1587 (2023).
Kuziel, G. A. & Rakoff-Nahoum, S. The gut microbiome. Curr. Biol. 32(6), R257–R64. https://doi.org/10.1016/j.cub.2022.02.023 (2022).
Li, Z. et al. Critical role of the gut microbiota in immune responses and cancer immunotherapy. J. Hematol. Oncol. 17(1), 33. https://doi.org/10.1186/s13045-024-01541-w (2024).
Bretto, E., Urpì-Ferreruela, M., Casanova, G. R. & González-Suárez, B. The role of gut microbiota in gastrointestinal immune homeostasis and inflammation: Implications for inflammatory bowel disease. Biomedicines 13(8), 1807 (2025).
Di Vincenzo, F., Del Gaudio, A., Petito, V., Lopetuso, L. R. & Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: A narrative review. Intern. Emerg. Med. 19(2), 275–93. https://doi.org/10.1007/s11739-023-03374-w (2024).
Hagihara, M. et al. Clostridium butyricum modulates the microbiome to protect intestinal barrier function in mice with antibiotic-induced dysbiosis. Iscience https://doi.org/10.1016/j.isci.2019.100772 (2020).
Soto Chervin, C. & Gajewski, T. F. Microbiome-based interventions: Therapeutic strategies in cancer immunotherapy. Immuno-Oncol. Technol. 8, 12–20. https://doi.org/10.1016/j.iotech.2020.11.001 (2020).
Ghosh, S., Whitley, C. S., Haribabu, B. & Jala, V. R. Regulation of intestinal barrier function by microbial metabolites. Cell. Mol. Gastroenterol. Hepatol. 11(5), 1463–82. https://doi.org/10.1016/j.jcmgh.2021.02.007 (2021).
Hou, S., Yu, J., Li, Y., Zhao, D. & Zhang, Z. Advances in fecal microbiota transplantation for gut dysbiosis-related diseases. Adv. Sci. 12(13), 2413197. https://doi.org/10.1002/advs.202413197 (2025).
Yang, R., Chen, Z. & Cai, J. Fecal microbiota transplantation: Emerging applications in autoimmune diseases. J. Autoimmun. 141, 103038. https://doi.org/10.1016/j.jaut.2023.103038 (2023).
Zhang, M., Liu, J. & Xia, Q. Role of gut microbiome in cancer immunotherapy: from predictive biomarker to therapeutic target. Experimental Hematol. Oncol. 12 (1), 84. https://doi.org/10.1186/s40164-023-00442-x (2023).
Reunanen, J. et al. Akkermansia muciniphila Adheres to Enterocytes and Strengthens the Integrity of the Epithelial Cell Layer. Appl. Environ. Microbiol. 81 (11), 3655–3662. https://doi.org/10.1128/AEM.04050-14 (2015).
Ouyang, J. et al. The bacterium Akkermansia muciniphila: A sentinel for gut permeability and its relevance to HIV-related inflammation. Front. Immunol. 11-2020. https://doi.org/10.3389/fimmu.2020.00645 (2020).
Routy, B. et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359 (6371), 91–97. https://doi.org/10.1126/science.aan3706 (2018).
Baruch, E. N. et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science 371 (6529), 602–609. https://doi.org/10.1126/science.abb5920 (2021).
Davar, D. et al. Fecal microbiota transplant overcomes resistance to anti-PD-1 therapy in melanoma patients. Science 371 (6529), 595–602. https://doi.org/10.1126/science.abf3363 (2021).
Gu, C. et al. Therapeutic potential of fecal microbiota transplantation in colorectal cancer based on gut microbiota regulation: From pathogenesis to efficacy. Therap Adv Gastroenterol https://doi.org/10.1177/17562848251327167 (2025).
Jiang, S. et al. An emerging strategy: probiotics enhance the effectiveness of tumor immunotherapy via mediating the gut microbiome. Gut Microbes. 16 (1), 2341717. https://doi.org/10.1080/19490976.2024.2341717 (2024).
Chandrasekaran, P., Weiskirchen, S. & Weiskirchen, R. Effects of Probiotics on Gut Microbiota: An Overview. Int. J. Mol. Sci. 25 (11), 6022 (2024). https://www.mdpi.com/1422-0067/25/11/6022
Rowaiye, A. et al. Gut microbiota alteration - Cancer relationships and synbiotic roles in cancer therapies. Microbe 4, 100096. https://doi.org/10.1016/j.microb.2024.100096 (2024).
Scott, A. J., Merrifield, C. A., Younes, J. A. & Pekelharing, E. P. Pre-, pro- and synbiotics in cancer prevention and treatment-a review of basic and clinical research. Ecancermedicalscience 12, 869. https://doi.org/10.3332/ecancer.2018.869 (2018).
Singh, N. K. et al. Synbiotics as Supplemental Therapy for the Alleviation of Chemotherapy-Associated Symptoms in Patients with Solid Tumours. Nutrients 15 (7), 1759 (2023). https://www.mdpi.com/2072-6643/15/7/1759
Huang, C. H., Cheng, J. Y., Deng, M. C., Chou, C. H. & Jan, T. R. Prebiotic effect of diosgenin, an immunoactive steroidal sapogenin of the Chinese yam. Food Chem. 132 (1), 428–432. https://doi.org/10.1016/j.foodchem.2011.11.016 (2012).
Dong, M. et al. Diosgenin promotes antitumor immunity and PD-1 antibody efficacy against melanoma by regulating intestinal microbiota. Cell. Death Dis. 9 (10), 1039. https://doi.org/10.1038/s41419-018-1099-3 (2018).
Zhang, J., Ma, X., Du, W., Mu, G. & Wu, X. Immune restoration mechanism analysis of Akkermansia PROBIO in mice treated with cyclophosphamide through Re-balancing microbiota composition. Food Bioscience. 68, 106450. https://doi.org/10.1016/j.fbio.2025.106450 (2025).
Derosa, L. & Zitvogel, L. A probiotic supplement boosts response to cancer immunotherapy. Nat. Med. 28 (4), 633–634. https://doi.org/10.1038/s41591-022-01723-4 (2022).
Gao, G. et al. Lacticaseibacillus rhamnosus Probio-M9 enhanced the antitumor response to anti-PD-1 therapy by modulating intestinal metabolites. EBioMedicine 91, 104533. https://doi.org/10.1016/j.ebiom.2023.104533 (2023).
Bhatt, A., Haslam, A. & Prasad, V. The effect of gastrointestinal microbiome supplementation on immune checkpoint inhibitor immunotherapy: a systematic review. J. Cancer Res. Clin. Oncol. 149 (10), 7355–7362. https://doi.org/10.1007/s00432-023-04656-8 (2023).
Zhao, S. et al. Assessing the impact of probiotics on immunotherapy effectiveness and antibiotic-mediated resistance in cancer: A systematic review and meta-analysis. Front. Immunol. 16, 2025. https://doi.org/10.3389/fimmu.2025.1538969 (2025).
Page, M. J. et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ 372, n160. https://doi.org/10.1136/bmj.n160 (2021).
Higgins, J. P. T. et al. (eds) C,. Cochrane Handbook for Systematic Reviews of Interventions. version 5.2.0 (updated June (2017). https://training.cochrane.org/handbook: Cochrane; 2017.
Slim, K. et al. Methodological index for non-randomized studies (minors): Development and validation of a new instrument. ANZ J. Surg. 73(9), 712–6. https://doi.org/10.1046/j.1445-2197.2003.02748.x (2003).
Barker, T. H. et al. Conducting proportional meta-analysis in different types of systematic reviews: A guide for synthesisers of evidence. BMC Med. Res. Methodol. 21(1), 189. https://doi.org/10.1186/s12874-021-01381-z (2021).
Jackson, D., Bowden, J. & Baker, R. How does the DerSimonian and Laird procedure for random effects meta-analysis compare with its more efficient but harder to compute counterparts?. J. Stat. Plan. Inference 140(4), 961–70. https://doi.org/10.1016/j.jspi.2009.09.017 (2010).
Balduzzi, S., Rücker, G. & Schwarzer, G. How to perform a meta-analysis with R: A practical tutorial. Evid. Based Ment. Health. 22(4), 153–60. https://doi.org/10.1136/ebmental-2019-300117 (2019).
Al Amer, F. M. & Lin, L. Empirical assessment of prediction intervals in Cochrane meta-analyses. Eur. J. Clin. Invest. 51(7), e13524. https://doi.org/10.1111/eci.13524 (2021).
Baruch, E. N. et al. Preliminary results from a microbiome-based phase i clinical trial - Fecal microbiota transplantation in metastatic melanoma patients who failed immunotherapy (NCT03353402). Pigment Cell. Melanoma Res. 32 (1), 97–98. https://doi.org/10.1111/pcmr.12738 (2019).
Baruch, E. N. et al. The gut microbiome enhances anti-PD-1 efficacy in a tumor-agnostic manner: results from a phase II trial of fecal microbiota transplantation and anti-PD-1 re-induction in MSI-H refractory cancers. J. Immunother. Cancer. 12, A1422. https://doi.org/10.1136/jitc-2024-SITC2024.1266 (2024).
Baruch, E. N. et al. Fecal microbiota transplantation (FMT) and re-induction of anti-PD-1 therapy in refractory metastatic melanoma patients - Preliminary results from a phase I clinical trial (NCT03353402). Cancer Res. 79 (13). https://doi.org/10.1158/1538-7445.SABCS18-CT042 (2019).
Bergerot, P. et al. Pilot study to evaluate the biologic effect of the probiotic CBM588 in combination with nivolumab/ ipilimumab for patients with mRCC. Kidney Cancer. 4, S45. https://doi.org/10.3233/KCA-200001 (2020).
Ciccarese, C. et al. Fecal microbiota transplantation (FMT) versus placebo in patients receiving pembrolizumab plus axitinib for metastatic renal cell carcinoma: Preliminary results of the randomized phase II TACITO trial. Ann. Oncol. https://doi.org/10.1016/j.annonc.2024.08.2320 (2024).
Dizman, N. et al. Randomized prospective trial assessing Bifidobacteriumcontaining probiotic supplementation in metastatic renal cell carcinoma (mRCC) patients receiving vascular endothelial growth factor-tyrosine kinase inhibitors (VEGF-TKIs). J. Clin. Oncol. https://doi.org/10.1200/JCO.2020.38.15_suppl.5078 (2020).
Duttagupta, S. et al. Phase II trial of fecal microbiota transplantation in combination with ipilimumab and nivolumab in patients with advanced cutaneous melanoma (FMT-LUMINate trial). Cancer Res. 84 (7). https://doi.org/10.1158/1538-7445.AM2024-CT258 (2024).
Elkrief, A. et al. Phase II trial of fecal microbiota transplantation (FMT) plus immune checkpoint inhibition (ICI) in advanced non-small cell lung cancer and cutaneous melanoma (FMT-LUMINate). Ann. Oncol. https://doi.org/10.1016/j.annonc.2024.08.1126 (2024).
Espinoza, I. R. G. et al. Effect of probiotics on quality of life for patients with cancer undergoing immunotherapy. J. Clin. Oncol. https://doi.org/10.1200/JCO.2024.42.16_suppl.e24148 (2024).
Fernandes, R. et al. Preventing adverse events in patients with renal cell carcinoma treated with doublet immunotherapy using fecal microbiota transplantation (FMT): Initial results from perform a phase I study. J. Clin. Oncol. https://doi.org/10.1200/JCO.2022.40.16_suppl.4553 (2022).
Galizia, D. et al. 863P Microbiota and cytokines profile in patients (pts) affected by recurrent metastatic head and neck squamous cell carcinoma (R/M HNSCC) treated with immune checkpoint inhibitors (ICIs) +/- chemotherapy (CT) and prebiotic inulin in the PRINCESS study. Ann. Oncol. 34, S560. https://doi.org/10.1016/j.annonc.2023.09.2009 (2023).
Huang, M. J. et al. Fecal Microbiota Transplantation Plus Rechallenging Immunotherapy in Patients with Advanced Non Small Cell Lung Cancer: An Exploratory Study. pp. S262–S3. (2024).
Kim, Y. et al. Fecal microbiota transplantation improves anti-PD-1 inhibitor efficacy in unresectable or metastatic solid cancers refractory to anti-PD-1 inhibitor. Cell Host Microbe 32(8), 1380–93.e9. https://doi.org/10.1016/j.chom.2024.06.010 (2024).
Lenehan, J. G. et al. Combining Fecal Microbiota Transplantation with Immunotherapy in Treatment-Naive Patients with Advanced Melanoma. Pigment Cell. Melanoma Res. 35 (1), 139–140. https://doi.org/10.1111/pcmr.13018 (2022).
Meza, L. A. et al. Intestinal microbiome associated with development of grade 3/4 adverse in patients with metastatic renal cell carcinoma (mRCC) treated with nivolumab plus ipilimumab (N/I) and probiotic support: Results from a phase Ib study. J. Clin. Oncol. https://doi.org/10.1200/JCO.2022.40.6_suppl.374 (2022).
Morita, A. et al. Impacts of probiotics on the efficacies of immune checkpoint inhibitors with or without chemotherapy for patients with advanced non-small-cell lung cancer. Int. J. Cancer. 154(9), 1607–15. https://doi.org/10.1002/ijc.34842 (2024).
Nakatsukasa, H. et al. Clinical impact of concomitant BIO-three use in advanced or recurrent non-small cell lung cancer treated with immune-checkpoint inhibitor. Int. J. Clin. Oncol. 29(12), 1840–9. https://doi.org/10.1007/s10147-024-02622-z (2024).
Park, S. R. et al. Fecal microbiota transplantation combined with anti-PD-1 inhibitor for unresectable or metastatic solid cancers refractory to anti-PD-1 inhibitor. J. Clin. Oncol. 41(16), 105. https://doi.org/10.1200/jco.2023.41.16_suppl.105 (2023).
Pomej, K. et al. Fecal microbiota transplant combined with atezolizumab plus bevacizumab in patients with hepatocellular carcinoma who progressed on atezolizumab plus bevacizumab - interim analysis of the FAB-HCC phase II pilot study. Z. Gastroenterol. 62 (5), e479–e80. https://doi.org/10.1055/s-0044-1786886 (2024).
Routy, B. et al. Microbiome modification with fecal microbiota transplant from healthy donors before anti-PD1 therapy reduces primary resistance to immunotherapy in advanced and metastatic melanoma patients. J. Immunother. Cancer. 10, A645. https://doi.org/10.1136/jitc-2022-SITC2022.0614 (2022).
Routy, B. et al. Fecal microbiota transplantation plus anti-PD-1 immunotherapy in advanced melanoma: A phase I trial. Nat. Med. 29(8), 2121–32. https://doi.org/10.1038/s41591-023-02453-x (2023).
Spencer, C. N. et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science 374(6575), 1632–40. https://doi.org/10.1126/science.aaz7015 (2021).
Spreafico, A. et al. First-in-class microbial ecosystem therapeutic 4 (MET4) in combination with immune checkpoint inhibitors in patients with advanced solid tumors (MET4-IO trial). Ann. Oncol. 34(6), 520–30. https://doi.org/10.1016/j.annonc.2023.02.011 (2023).
Tomita, Y. et al. Association of probiotic Clostridium butyricum therapy with survival and response to immune checkpoint blockade in patients with lung cancer. Cancer Immunol. Res. 8(10), 1236–42. https://doi.org/10.1158/2326-6066.CIR-20-0051 (2020).
Tong, L. et al. Evaluating Oral Probiotic Supplements as Complementary Treatment in Advanced Lung Cancer Patients Receiving ICIs: A Prospective Real-World Study. Cancer Control https://doi.org/10.1177/10732748241253959 (2024).
Ullern, A. et al. MITRIC: Microbiota transplant to cancer patients progressing on immunotherapy using feces from clinical responders. J. Immunother. Cancer 12, A767. https://doi.org/10.1136/jitc-2024-SITC2024.0669 (2024).
Wang, X. et al. Effect of probiotics combined with immune checkpoint suppressors and chemotherapeutic agents on digestive system function, intestinal immunity and prognosis in patients with metastatic colorectal carcinoma: A quasi-experimental study. BMC Gastroenterol. https://doi.org/10.1186/s12876-025-03604-9 (2025).
Wang, Y. et al. Concomitant medications alter clinical outcomes in patients with advanced digestive tract cancer receiving PD-1 checkpoint inhibitors combined with antiangiogenetic agents. J. Gastrointest. Cancer 55(3), 1388–400. https://doi.org/10.1007/s12029-024-01095-7 (2024).
Xue, Y. et al. Fecal microbiota transplantation combined with anti-PD-(L)1 inhibitors as first-line maintenance therapy for advanced gastric and non small cell lung cancer. J. Clin. Oncol. https://doi.org/10.1200/JCO.2024.42.16_suppl.2646 (2024).
Zhang, Y. et al. Fecal microbiota transplantation promotes immunotherapy sensitivity in refractory gastrointestinal cancer patients: open label, single-arm, single center, phase 1 study. (2024).
Zhao, W. & Chen, Y. Fecal microbiota transplantation in combination with fruquintinib and tislelizumab in refractory microsatellite stable metastatic colorectal cancer: A single center phase II trial. Immuno-Oncology Technol. 16 https://doi.org/10.1016/j.iotech.2022.100214 (2022).
Zhao, W. et al. Updated outcomes and exploratory analysis of RENMIN-215: Tislelizumab plus fruquintinib and fecal microbiota transplantation in refractory microsatellite stable metastatic colorectal cancer. Am. J. Cancer Res. 14(11), 5351–64. https://doi.org/10.62347/XKUJ3012 (2024).
Zhao, W. et al. Fecal microbiota transplantation plus tislelizumab and fruquintinib in refractory microsatellite stable metastatic colorectal cancer: An open-label, single-arm, phase II trial (RENMIN-215). eClinicalMedicine https://doi.org/10.1016/j.eclinm.2023.102315 (2023).
Zhu, W. et al. Safety and preliminary efficacy of different doses of fecal microbiota capsule transplantation combined with anti-PD-1 inhibitor for patients with advanced malignant solid tumors. J. Clin. Oncol. https://doi.org/10.1200/JCO.2024.42.16_suppl.e14622 (2024).
Dizman, N. et al. Nivolumab plus ipilimumab with or without live bacterial supplementation in metastatic renal cell carcinoma: A randomized phase 1 trial. Nat. Med. 28(4), 704–12. https://doi.org/10.1038/s41591-022-01694-6 (2022).
Zhang, Y. et al. Fecal microbiota transplantation promotes immunotherapy sensitivity in refractory gastrointestinal cancer patients: open label, single-arm, single center, phase 1 study. medRxiv https://doi.org/10.1101/2024.08.21.24312340 (2024).
Huang, M. J. et al. P2.11A.23 Fecal microbiota transplantation plus rechallenging immunotherapy in patients with advanced non small cell lung cancer: An exploratory study. J. Thorac. Oncol. 19(10), S262–S3. https://doi.org/10.1016/j.jtho.2024.09.472 (2024).
Wang, X. et al. Effect of probiotics combined with immune checkpoint suppressors and chemotherapeutic agents on digestive system function, intestinal immunity and prognosis in patients with metastatic colorectal carcinoma: A quasi-experimental study. BMC Gastroenterol. 25(1), 38. https://doi.org/10.1186/s12876-025-03604-9 (2025).
Kim, S., Kim, A., Shin, J.-Y. & Seo, J.-S. The tumor immune microenvironmental analysis of 2,033 transcriptomes across 7 cancer types. Sci. Rep. 10(1), 9536. https://doi.org/10.1038/s41598-020-66449-0 (2020).
Liu, Y., Wang, J. & Wu, C. Modulation of gut microbiota and immune system by probiotics, pre-biotics, and post-biotics. Front. Nutr. 8, 2021. https://doi.org/10.3389/fnut.2021.634897 (2022).
Chen, S. et al. Response efficacy of PD-1 and PD-L1 inhibitors in clinical trials: A systematic review and meta-analysis. Front. Oncol. 11, 562315. https://doi.org/10.3389/fonc.2021.562315 (2021).
Mangla, A. et al. Neoadjuvant Dual Checkpoint Inhibitors vs Anti-PD1 Therapy in High-Risk Resectable Melanoma: A Pooled Analysis. JAMA Oncol. 10 (5), 612–620. https://doi.org/10.1001/jamaoncol.2023.7333 (2024).
Mc Neil, V. & Lee, S. W. Advancing Cancer Treatment: A Review of Immune Checkpoint Inhibitors and Combination Strategies. Cancers 17 (9), 1408 (2025). https://www.mdpi.com/2072-6694/17/9/1408
Devaraji, M. & Varghese Cheriyan, B. Immune-based cancer therapies: mechanistic insights, clinical progress, and future directions. J. Egypt. Natl Cancer Inst. 37 (1), 62. https://doi.org/10.1186/s43046-025-00319-6 (2025).
Alsaafeen, B. H., Ali, B. R. & Elkord, E. Resistance mechanisms to immune checkpoint inhibitors: updated insights. Mol. Cancer. 24 (1), 20. https://doi.org/10.1186/s12943-024-02212-7 (2025).
Baruch, E. N., Gaglani, T. & Wargo, J. A. Fecal microbiota transplantation as a mean of overcoming immunotherapy-resistant cancers - hype or hope? Ther. Adv. Med. Oncol. 13, 17588359211045853. https://doi.org/10.1177/17588359211045853 (2021).
Yang, Y. et al. Fecal microbiota transplantation: no longer cinderella in tumour immunotherapy. eBioMedicine 100, 104967. https://doi.org/10.1016/j.ebiom.2024.104967 (2024).
De Palma, M. & Hanahan, D. The biology of personalized cancer medicine: Facing individual complexities underlying hallmark capabilities. Mol. Oncol. 6 (2), 111–127. https://doi.org/10.1016/j.molonc.2012.01.011 (2012).
Zhang, L. et al. The correlation between probiotic use and outcomes of cancer patients treated with immune checkpoint inhibitors. Front. Pharmacol. 13, 937874. https://doi.org/10.3389/fphar.2022.937874 (2022).
Batista, V. L. et al. Probiotics, prebiotics, synbiotics, and paraprobiotics as a therapeutic alternative for intestinal mucositis. Front. Microbiol. 11, 2020. https://doi.org/10.3389/fmicb.2020.544490 (2020).
Spencer, C. N. et al. Abstract 2838: The gut microbiome (GM) and immunotherapy response are influenced by host lifestyle factors. Cancer Res. 79 (13_Supplement), 2838. https://doi.org/10.1158/1538-7445.Am2019-2838 (2019).
Ajab, S. M. et al. Microbiota composition effect on immunotherapy outcomes in colorectal cancer patients: A systematic review. PLoS. One 19(7), e0307639. https://doi.org/10.1371/journal.pone.0307639 (2024).
Kang, X., Lau, H. C. & Yu, J. Modulating gut microbiome in cancer immunotherapy: Harnessing microbes to enhance treatment efficacy. Cell Rep. Med. 5(4), 101478. https://doi.org/10.1016/j.xcrm.2024.101478 (2024).
Lei, W., Zhou, K., Lei, Y., Li, Q. & Zhu, H. Gut microbiota shapes cancer immunotherapy responses. Npj. Biofilms Microbiomes 11(1), 143. https://doi.org/10.1038/s41522-025-00786-8 (2025).
Sun, J. et al. Gut microbiota as a new target for anticancer therapy: From mechanism to means of regulation. Npj. Biofilms Microbiomes 11(1), 43. https://doi.org/10.1038/s41522-025-00678-x (2025).
Xu, Q. et al. The oral-gut microbiota axis: A link in cardiometabolic diseases. Npj. Biofilms Microbiomes 11(1), 11. https://doi.org/10.1038/s41522-025-00646-5 (2025).
Blaut, M. & Clavel, T. Metabolic diversity of the intestinal microbiota: Implications for health and disease1,. J. Nutr. 137(3), 751S–5S. https://doi.org/10.1093/jn/137.3.751S (2007).
Jin, Y., Jie, Z. & Fan, X. Gut microbes and immunotherapy for non-small cell lung cancer: A systematic review. Front. Oncol. 15, 2025. https://doi.org/10.3389/fonc.2025.1518474 (2025).
Lim, M. Y., Hong, S. & Nam, Y.-D. Understanding the role of the gut microbiome in solid tumor responses to immune checkpoint inhibitors for personalized therapeutic strategies: A review. Front. Immunol. 15, 2024. https://doi.org/10.3389/fimmu.2024.1512683 (2025).
Jayathilaka, B., Mian, F., Franchini, F., Au-Yeung, G. & Ijzerman, M. Cancer and treatment specific incidence rates of immune-related adverse events induced by immune checkpoint inhibitors: A systematic review. Br. J. Cancer. 132(1), 51–57. https://doi.org/10.1038/s41416-024-02887-1 (2025).
Funding
This project is funded by the National Research Council of Thailand (NRCT): the High-Potential Research Team Grant Program (N42A680423) and the Rachadapisek Sompote Matching Fund (RA-MF-01/69). The academic endeavors of the Thailand Hub of Talents in Cancer Immunotherapy (TTCI) receive support from the National Research Council of Thailand. NH and MT were funded by the Second Century Fund (C2F), Chulalongkorn University. HL, VM, and ND were supported by the Graduate Scholarship Programme for ASEAN or Non-ASEAN Countries, Chulalongkorn University. The funder did not influence the results/outcomes of the study despite author affiliations with the funde
Author information
Authors and Affiliations
Contributions
MT and NH contributed to conceptualization, supervision, and critical revision of the manuscript. The MT took a role in project administration and introduced the required software and resources. MT, HL, VM, and ND contributed to the data curation, formal analysis, and methodology. All the authors contributed to the writing and editing of the manuscript. All the authors read and approved the final manuscript. NH is the guarantor of the review for the integrity of the work as a whole.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Thu, M.S., Le, H.B.C., Duc, N.P. et al. Impact of microbiome-modulating strategies in cancer patients receiving immunotherapy (MSIT): A systematic review and meta-analysis. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44743-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44743-7