Abstract
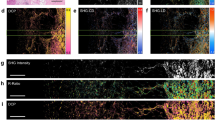
Histological analysis remains the gold standard for tissue diagnosis and evaluation, but it often relies on subjective visual interpretation and limited field of view areas. In this study, we present a macroscopic, wide-field Mueller polarimetric imaging approach for the quantitative analysis of histological sections of melanoma induced with the B16F10 cell line in mice. By using the depolarization index, our method enables objective tissue discrimination based on intrinsic tissue properties. A color-coded segmentation mask derived from the depolarization index values was implemented to classify tissue into strongly depolarizing regions, associated with blood vessels, and weakly depolarizing regions, which predominantly correspond to tumor cell–dominated tissue. This pixel-wise classification allowed for quantitative mapping and statistical analysis of spatial heterogeneity across several mm² of entire histological sections, significantly extending the field of view compared to conventional microscopy. As a potential application, the technique was applied to evaluate histological sections from a preclinical murine melanoma model subjected to a treatment regimen including antiangiogenic agents. The results demonstrated measurable differences in the amount of strongly depolarizing tissue, associated with blood vessels, in the treatment group, supporting the method’s sensitivity to antiangiogenic effects in the tumor microenvironment. These findings highlight the potential of Mueller polarimetry as a complementary tool for histological assessment, enabling reproducible, large-area tissue characterization beyond visual inspection.
Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Martínez-Jiménez, F. et al. A compendium of mutational cancer driver genes. Nat. Rev. Cancer. 20, 555–572. https://doi.org/10.1038/s41568-020-0290-x (2020).
Vitale, I., Shema, E., Loi, S. & Galluzzi, L. Intratumoral heterogeneity in cancer progression and response to immunotherapy. Nat. Med. 27 (2), 212–224. https://doi.org/10.1038/s41591-021-01233-9 (2021).
Kim, I. S. & Zhang, X. H. One microenvironment does not fit all: heterogeneity beyond cancer cells. Cancer Metastasis Rev. 35 (4), 601–629. https://doi.org/10.1007/s10555-016-9643-z (2016).
Katsuta, E., Rashid, O. M. & Takabe, K. Clinical relevance of tumor microenvironment: immune cells, vessels, and mouse models. Hum. Cell. 33, 930–937. https://doi.org/10.1007/s13577-020-00380-4 (2020).
Arnold, M. et al. Global burden of cutaneous melanoma in 2020 and projections to 2040. JAMA Dermatol. 158 (5), 495–503. https://doi.org/10.1001/jamadermatol.2022.0160 (2022).
Wu, Z. et al. The role of angiogenesis in melanoma: Clinical treatments and future expectations. Front. Pharmacol. 13, 1028647. https://doi.org/10.3389/fphar.2022.1028647 (2022).
Turlej, E. et al. Cross-Talk between cancer and its cellular environment-A role in cancer progression. Cells 14 (6), 403. https://doi.org/10.3390/cells14060403 (2025).
García Melián, M. F., Moreno, M., Cerecetto, H. & Calzada, V. Aptamer-Based Immunotheranostic Strategies. Cancer Biother Radiopharm. 38 (4), 246–255 (2023).
Odeh, F. et al. Molecules. Aptamers chemistry: Chemical modifications and conjugation strategies. (2020).
Shi, H. et al. Nucleolin is a receptor that mediates antiangiogenic and antitumor activity of endostatin. Blood 110, 2899–2906 (2007).
Huang, Y. et al. The angiogenic function of nucleolin is mediated by vascular endothelial growth factor and nonmuscle myosin. Blood 107, 3564–3571 (2006).
Rosenberg, J. E. et al. A phase II trial of AS1411 (a novel nucleolin-targeted DNA aptamer) in metastatic renal cell carcinoma. Investig. New. Drugs. 32, 178–187 (2014).
Reyes-Reyes, E. M., Šalipur, F. R., Shams, M., Forsthoefel, M. K. & Bates, P. J. Mechanistic studies of anticancer aptamer AS1411 reveal a novel role for nucleolin in regulating Rac1 activation. Mol. Oncol. 9, 1392–1405 (2015).
Pérez-Torres, A. et al. The synthetic parasite-derived peptide GK1 increases survival in a preclinical mouse melanoma model. Cancer Biother Radiopharm. 28 (9), 682–690 (2013).
Pérez-Torres, A. et al. Hematological Effects, Serum, and Pulmonary Cytokine Profiles in a Melanoma Mouse Model Treated with GK1. Cancer Biother Radiopharm. 30 (6), 247–254. https://doi.org/10.1089/cbr.2015.1835 (2015).
Vera-Aguilera, J. et al. Novel Treatment of Melanoma: Combined Parasite-Derived Peptide GK-1 and Anti-Programmed Death Ligand 1 Therapy. Cancer Biother Radiopharm. 32 (2), 49–56. https://doi.org/10.1089/cbr.2016.2123 (2017).
Torres-García, D. et al. GK-1 peptide reduces tumor growth, decreases metastatic burden, and increases survival in a murine breast cancer model. Vaccine 35, 5653–5661. https://doi.org/10.1016/j.vaccine.2017.08.060 (2017).
Hernández-Aceves, J. A. et al. GK-1 effectively reduces angiogenesis and prevents T cell exhaustion in a breast cancer murine experimental model. Cancer Immunol. Immunother. 72 (11), 3825–3838. https://doi.org/10.1007/s00262-023-03538-9 (2023).
Alali, S. & Vitkin, I. A. Polarized light imaging in biomedicine: emerging Mueller matrix methodologies for bulk tissue assessment. J. Biomed. Opt. 20 (6), 061104 (2015).
He, C. et al. Polarisation optics for biomedical and clinical applications: a review. Light Sci. Appl. 10, 194 (2021).
Badieyan, S., Ameri, A., Razzaghi, M. R., Rafii-Tabar, H. & Sasanpour, P. Mueller matrix imaging of prostate bulk tissues; polarization parameters as a discriminating benchmark. Photodiag Photodyn Ther. 26, 90–96 (2019).
Pham, T. T. H. et.al. Analysis of polarization features of human breast cancer tissue by mueller matrix visualization. J. Biomed. Opt., 29, (2024).
Nishkarsh, K. et. al. Mueller matrix-based characterization of cervical tissue sections: a quantitative comparison of polar and differential decomposition methods. J. Biomedical Opt., 29, (2024).
Tumanova, K. et al. Mueller matrix polarization parameters correlate with local recurrence in patients with stage III colorectal cancer. Sci. Rep. 13, 13424 (2023).
Sharma, M. et al. Characterizing colon cancer stages through optical polarimetry-assisted digital staining. Lasers Med. Sci. 39, 59 (2024).
Vahidnia, A. et al. Quantitative polarimetry Mueller matrix decomposition approach for diagnosing melanoma and non-melanoma human skin cancer. OSA Continuum. 4, 2862–2874 (2021).
Du, V. N. & Le Cost-effective Mueller matrix microscopy for label-free differentiation of melanoma from normal skin and common skin cancer types. Biomed. Opt. Express. 16, 5235–5247 (2025). AustinBolden.
Nakhleh, R. E. et al. Interpretive diagnostic error reduction in surgical pathology and cytology: guideline from the College of American Pathologists Pathology and Laboratory Quality Center and the Association of Directors of Anatomic and Surgical Pathology. Arch. Pathol. Lab. Med. 140, 29–40 (2016).
Chipman, R. A. Handbook of Optics (McGraw-Hill, 1995). Ch. 22.
Montes-González, I., Rodríguez-Herrera, O. G. & Avendaño-Alejo, M. Effects of typical liquid-crystal retarder errors on optimized Stokes polarimeters. Appl. Opt. 61 (35), 10458–10464 (2022).
Montes-Gonzalez, I. et al. Generalized Eigenvalue Calibration Method for Mueller Imaging Polarimeters Based on Micro-Polarized Sensors With Optimal Set of Calibration Samples. Photonic Sens. 15, 250320 (2025).
Montes-González, I., Campos, J., Estévez, I. & Lizana, A. Modeling and validation of a Mueller matrix imaging polarimeter based on liquid crystals and a polarimetric camera, Proc. SPIE 13568, Modeling Aspects in Optical Metrology X, 135680 N (8 August 2025).
Vivanco-Rojas, O. et al. Corneal neovascularization is inhibited with nucleolin-binding aptamer, AS1411. Exp. Eye Res. 193, 107977. https://doi.org/10.1016/j.exer.2020.107977 (2020).
Acknowledgements
The authors acknowledge the optical microscopy service of the Universitat Autònoma de Barcelona, and the histotechnological assistance of Dulce Marlet Morales Sánchez and Damaris Ramírez Hernández.
Funding
This work was financially supported by the Ministerio de Ciencia e Innovación, Fondos FEDER, Spain (Grant Nos. PID2024-156240OBOB-C22, and PDC2022-133332-C21), Generalitat de Catalunya, Spain (Grant No. 2021SGR00138). and DGAPA UNAM through the project PAPIIT: IT201821 (FM/DI/086/2020).
Author information
Authors and Affiliations
Contributions
Conceptualization: I.M., A.P. and A.L. Optics and instrumentation: I.M. and J.C. Performed the experiments: J.B., I.M. and I.E. Analyzed the data and prepared all figures: J.B. I.M. and M. C. Provided Mueller imaging motivation and insight: A.L., M.C. and I.E. Supplied the tissue samples: A.P. and E.P. Identified the ROIs for analysis. A.P. I. M. and J. B. Provided clinical motivation and insight: A.P. and E. P. Writing and editing the manuscript: I.M., J.B., A. P. and A.L. Funding acquisition: A.L., J.C. and A.P. All authors participated approved the final document before submission and are responsible for its content.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Montes-Gonzalez, I., Bisbal-Amat, J., Perez-Torres, A. et al. Macroscopic polarimetric discrimination and quantification of antiangiogenic effect of AGRO aptamer and GK1 peptide in a preclinical melanoma model. Sci Rep (2026). https://doi.org/10.1038/s41598-026-44959-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-44959-7