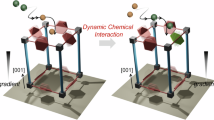
Abstract
A copper-based metal-organic framework (MOF) HKUST-1 exhibits high methane adsorption capacity, primarily due to strong Coulomb interactions near open metal sites (OMSs) and van der Waals interactions within ligand-enclosed cavities. While the adsorption sites have been widely studied, the diffusion pathways through which methane reaches these sites remain unclear, particularly regarding the influence of OMSs. In this study, molecular dynamics (MD) simulations were conducted to investigate methane diffusion in HKUST-1 with and without OMSs. First-principles calculations and methane uptake experiments were also performed to support and validate the simulation results. We found that methane was trapped in ligand-enclosed sites due to steric hindrance, while regions near OMSs serve as diffusion hubs. Moreover, in the absence of OMSs, methane backflow was observed at the surface of HKUST-1 due to the presence of stable adsorption sites on surface planes. When OMSs are present, these stable sites shift toward the OMS regions via Coulomb interactions, reducing surface backflow and enhancing methane uptake. The experimentally measured increase in methane adsorption for HKUST-1 with OMSs supports the simulation results. This study can guide the future design of MOFs with enhanced gas adsorption capacity.
Similar content being viewed by others
Data availability
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Allen, D. T. Methane emissions from natural gas production and use: Reconciling bottom-up and top-down measurements. Curr. Opin. Chem. Eng. 5, 78–83 (2014).
Mahmoud, E. Evolution of the design of CH4 adsorbents. Surfaces 3, 433–466 (2020).
Gutiérrez-Serpa, A., Pacheco-Fernández, I., Pasán, J. & Pino, V. Metal–organic frameworks as key materials for solid-phase microextraction devices—A review. Separations. 6, 1–29 (2019).
Uzun, A. & Keskin, S. Site characteristics in metal organic frameworks for gas adsorption. Prog. Surf. Sci. 89, 56–79 (2014).
Chong, K. C. et al. A review of HKUST-1 metal-organic frameworks in gas adsorption. IOP Conf. Ser. Earth Environ. Sci 1135, (2023).
Lin, Y., Kong, C., Zhang, Q. & Chen, L. Metal-organic frameworks for carbon dioxide capture and methane storage. Adv. Energy Mater. 7, (2017).
Getzschmann, J. et al. Methane storage mechanism in the metal-organic framework Cu 3(btc)2: An in situ neutron diffraction study. Microporous Mesoporous Mater. 136, 50–58 (2010).
Chen, L., Grajciar, L., Nachtigall, P. & Düren, T. Accurate prediction of methane adsorption in a metal-organic framework with unsaturated metal sites by direct implementation of an ab initio derived potential energy surface in GCMC simulation. J. Phys. Chem. C. 115, 23074–23080 (2011).
Zhang, Z., Zhao, Y., Gong, Q., Lib, Z. & Li, J. MOFs for CO2 capture and separation from flue gas mixtures: The effect of multifunctional sites on their adsorption capacity and selectivity. Chem. Commun. 49, 653–661 (2013).
Wu, H. et al. Metal-organic frameworks with exceptionally high methane uptake: Where and how is methane stored? Chem. - Eur. J. 16, 5205–5214 (2010).
Rappé, A. K., Casewit, C. J., Colwell, K. S., Goddard, W. A. & Skiff, W. M. UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations. J. Am. Chem. Soc. 114, 10024–10035 (1992).
Koh, H. S., Rana, M. K., Wong-Foy, A. G. & Siegel, D. J. Predicting methane storage in open-metal-site metal-organic frameworks. J. Phys. Chem. C. 119, 13451–13458 (2015).
Wang, J. & Hou, T. Application of molecular dynamics simulations in molecular property prediction II: Diffusion coefficient. J. Comput. Chem. 32, 3505–3519 (2011).
Kondrat, S. Physics and modelling of intracellular diffusion (2018).
Song, Y., Jiang, B. & Li, F. L. Molecular dynamic simulations of selective self-diffusion of CH4/CO2/H2O/N2 in coal. IOP Conf. Ser. Mater. Sci. Eng. 213, (2017).
Babaei, H., McGaughey, A. J. H. & Wilmer, C. E. Transient mass and thermal transport during methane adsorption into the metal-organic framework HKUST-1. ACS Appl. Mater. Interfaces. 10, 2400–2406 (2018).
Wang, Z. et al. Molecular insights into hybrid CH4 physisorption-hydrate growth in hydrophobic metal–organic framework ZIF-8: Implications for CH4 storage. Chem. Eng. J. 430, 132901 (2022).
Awadallah-F, A., Hillman, F., Al-Muhtaseb, S. A. & Jeong, H. K. Adsorption equilibrium and kinetics of nitrogen, methane and carbon dioxide gases onto ZIF-8, Cu10%/ZIF-8, and Cu30%/ZIF-8. Ind. Eng. Chem. Res. 58, 6653–6661 (2019).
Ahmed, H. et al. Acoustomicrofluidic assembly of oriented and simultaneously activated metal–organic frameworks. Nat. Commun. 10, 1–10 (2019).
Mao, Y. et al. Specific oriented metal-organic framework membranes and their facet-tuned separation performance. ACS Appl. Mater. Interfaces. 6, 15676–15685 (2014).
Alfonso-Herrera, L. A. et al. Elucidating structural stability, bandgap, and photocatalytic hydrogen evolution of (H2O/DMF)@HKUST-1 host-guest systems. Chempluschem. 89, (2024).
Prestipino, C. et al. Local structure of framework Cu(II) in HKUST-1 metallorganic framework: Spectroscopic characterization upon activation and interaction with adsorbates. Chem. Mater. 18, 1337–1346 (2006).
Yakovenko, A. A., Reibenspies, J. H., Bhuvanesh, N. & Zhou, H. C. Generation and applications of structure envelopes for porous metal-organic frameworks. J. Appl. Crystallogr. 46, 346–353 (2013).
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996).
Zhao, L. et al. A force field for dynamic Cu-BTC metal-organic framework. J. Mol. Model. 17, 227–234 (2011).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Stukowski, A. Visualization and analysis of atomistic simulation data with OVITO-the Open Visualization Tool. Model Simul. Mater. Sci. Eng. 18, (2010).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B. 47, 558–561 (1993).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B Condens. Matter Mater. Phys. 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B Condens. Matter Mater. Phys. 50, 17953 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Stefan Grimme. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Koh, H. S., Rana, M. K., Hwang, J. & Siegel, D. J. Thermodynamic screening of metal-substituted MOFs for carbon capture. Phys. Chem. Chem. Phys. 15, 4573–4581 (2013).
Lee, S., Lee, G. & Oh, M. Lattice-guided construction and harvest of a naturally nonpreferred metal-organic framework. ACS Nano. 15, 17907–17916 (2021).
Bae, J. et al. Multiple coordination exchanges for room-temperature activation of open-metal sites in metal-organic frameworks. ACS Appl. Mater. Interfaces. 9, 24743–24752 (2017).
Funding
This research was financially supported by the Fundamental Research Program (PNKA310) of the Korea Institute of Materials Science and the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (No. RS-2024-00453815).
Author information
Authors and Affiliations
Contributions
H.-S.J. wrote the main manuscript and conducted MD simulations. E.C. and H.J.L. conducted experiments and wrote experimental parts in manuscript. G.K. and B.-H.K. conducted first-principles calculations. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jang, HS., Cho, E., Kim, G. et al. Methane diffusion path in HKUST-1 metal-organic framework revealed by atomistic simulations. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45125-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45125-9