Abstract
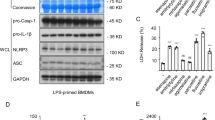
Cystic echinococcosis (CE), which primarily affects the liver, is associated with hepatocyte injury, although the underlying cellular and molecular mechanisms remain unclear. The aim of this study was to determine whether hepatocyte injury caused by Echinococcus granulosus sensu lato involves pyroptosis mediated through the ROS-NLRP3 inflammasome pathway. AML-12 hepatocytes were exposed to E. granulosus excretory-secretory products (ESPs), and cell viability, LDH release, cell death, and NLRP3 inflammasome-mediated pyroptosis were evaluated. The results indicated that ESP treatment reduced cell viability, increased LDH release and cell death, and upregulated markers of NLRP3 inflammasome-dependent pyroptosis. Pharmacological inhibition of NLRP3 with MCC950 markedly attenuated ESP-induced cytotoxicity and pyroptosis, confirming the critical role of NLRP3 activation in this process. Additionally, the ROS scavenger N-acetyl-L-cysteine (NAC) suppressed pyroptosis and reduced inflammasome activation, indicating that ESP-induced pyroptosis is ROS dependent. All of these results point to the possibility that ESPs cause hepatocyte damage in CE by inducing hepatocyte pyroptosis via the ROS-NLRP3 inflammasome pathway.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are included in the manuscript and its Supplementary Material.
References
Moro, P. & Schantz, P. M. Echinococcosis: A review. Int. J. Infect. Dis. 13, 125–133 (2009).
Agudelo Higuita, N. I., Brunetti, E. & McCloskey, C. Cystic echinococcosis. J. Clin. Microbiol. 54, 518–523 (2016).
Wen, H. et al. Echinococcosis: Advances in the 21st century. Clin. Microbiol. Rev. 32, e00075–e00018 (2019).
Hou, X. et al. Echinococcus granulosus: The establishment of the metacestode in the liver is associated with control of the CD4 + T-cell-mediated immune response in patients with cystic echinococcosis and a mouse model. Front. Cell. Infect. Microbiol. 12, 983119 (2022).
Monteiro, K. M., de Carvalho, M. O., Zaha, A. & Ferreira, H. B. Proteomic analysis of the Echinococcus granulosus metacestode during infection of its intermediate host. Proteomics 10, 1985–1999 (2010).
Wang, Y. et al. Proteomic analysis of the excretory/secretory products and antigenic proteins of Echinococcus granulosus adult worms from infected dogs. BMC Vet. Res. 11, 119 (2015).
Rao, Z. et al. Pyroptosis in inflammatory diseases and cancer. Theranostics 12, 4310–4329 (2022).
Yu, P. et al. Pyroptosis: Mechanisms and diseases. Sig. Transduct. Target. Ther. 6, 128 (2021).
P, B. & Vm, D. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 16, (2016).
Rc, C. & P, P. K, S. NLRP3 and pyroptosis blockers for treating inflammatory diseases. Trends Pharmacol. Sci. 43, (2022).
de Carvalho Ribeiro, M. & Szabo, G. Role of the inflammasome in liver disease. Annu. Rev. Pathol. 17, 345–365 (2022).
de Carvalho, R. V. H. & Zamboni, D. S. Inflammasome activation in response to intracellular protozoan parasites. Trends Parasitol. 36, 459–472 (2020).
Kong, D. L. et al. Soluble egg antigen of Schistosoma japonicum induces pyroptosis in hepatic stellate cells by modulating ROS production. Parasit. Vectors. 12, 475 (2019).
Palacios-Macapagal, D. et al. Cutting Edge: Eosinophils undergo caspase-1-mediated pyroptosis in response to necrotic liver cells. J. Immunol. 199, 847–853 (2017).
Casaravilla, C., Pittini, Á., Rückerl, D., Allen, J. E. & Díaz, Á. Activation of the NLRP3 inflammasome by particles from the Echinococcus granulosus laminated layer. Infect. Immun. 88, e00190–e00120 (2020).
Lin, R. et al. Echinococcus granulosus cyst fluid inhibits KDM6B-mediated demethylation of trimethylated histone H3 lysine 27 and interleukin-1β production in macrophages. Parasit. Vectors. 16, 422 (2023).
Jomova, K. et al. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: Chronic diseases and aging. Arch. Toxicol. 97, 2499–2574 (2023).
Park, C. et al. Induction of G2/M cell cycle arrest and apoptosis by genistein in human bladder cancer T24 cells through inhibition of the ROS-dependent PI3k/Akt signal transduction pathway. Antioxid. (Basel). 8, 327 (2019).
Bin Dajem, S. M. et al. Green tea (Camellia sinesis) ameliorates female Schistosoma mansoni-induced changes in the liver of Balb/C mice. Saudi J. Biol. Sci. 18, 361–368 (2011).
Rosa, C. P. et al. Reactive oxygen species trigger inflammasome activation after intracellular microbial interaction. Life Sci. 331, 122076 (2023).
Quan, J. H. et al. Toxoplasma gondii induces pyroptosis in human placental trophoblast and amniotic cells by inducing ROS production and activation of cathepsin B and NLRP1/NLRP3/NLRC4/AIM2 inflammasome. Am. J. Pathol. 193, 2047–2065 (2023).
Fu, C., Yang, C., Ni, C., Wang, L. & Hou, J. Echinococcus granulosus cyst fluid inhibits the type I interferon response by promoting ROS in macrophages. Acta Trop. 250, 107101 (2024).
Aslam, H., Bi, S. & Irshadullah, M. Analysis of antioxidant enzymes and oxidative stress markers in the liver of naturally infected Indian water buffalo (Bubalus bubalis) with cystic echinococcosis. J. Parasit. Dis. 47, 340–348 (2023).
Rodriguez Rodrigues, C. et al. Modulation of the mTOR pathway plays a central role in dendritic cell functions after Echinococcus granulosus antigen recognition. Sci. Rep. 11, 17238 (2021).
Kagan, I. G. & Agosin, M. Echinococcus antigens. Bull. World Health Organ. 39, 13–24 (1968).
Khorsandi, H. O. & Tabibi, V. Similarities of human hydatid cyst fluid components and the host serum. Acta Med. Iran. 21, 161–172 (1978).
Virginio, V. G. et al. Excretory/secretory products from in vitro-cultured Echinococcus granulosus protoscoleces. Mol. Biochem. Parasitol. 183, 15–22 (2012).
Carmena, D., Martínez, J., Benito, A. & Guisantes, J. A. Characterization of excretory-secretory products from protoscoleces of Echinococcus granulosus and evaluation of their potential for immunodiagnosis of human cystic echinococcosis. Parasitology 129, 371–378 (2004).
Gottstein, B. et al. Immunology of alveolar and cystic echinococcosis (AE and CE). Adv. Parasitol. 96, 1–54 (2017).
Yang, H. C. et al. The expression of cytokeratin and apoptosis-related molecules in echinococcosis related liver injury. Mol. Biochem. Parasitol. 248, 111455 (2022).
Place, D. E., Lee, S. & Kanneganti, T.-D. PANoptosis in microbial infection. Curr. Opin. Microbiol. 59, 42–49 (2021).
Yanpiset, P. et al. Gasdermin D-mediated pyroptosis in myocardial ischemia and reperfusion injury: Cumulative evidence for future cardioprotective strategies. Acta Pharm. Sin B. 13, 29–53 (2023).
Amarante-Mendes, G. P. et al. Pattern recognition receptors and the host cell death molecular machinery. Front. Immunol. 9, 2379 (2018).
Lei, Q., Yi, T. & Chen, C. NF-κB-Gasdermin D (GSDMD) axis couples oxidative stress and NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome-mediated cardiomyocyte pyroptosis following myocardial infarction. Med. Sci. Monit. 24, 6044–6052 (2018).
Wang, H. et al. Hepatic macrophages play critical roles in the establishment and growth of hydatid cysts in the liver during Echinococcus granulosus sensu stricto infection. PLoS Negl. Trop. Dis. 17, e0011746 (2023).
D’Autréaux, B. & Toledano, M. B. ROS as signalling molecules: Mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell. Biol. 8, 813–824 (2007).
Dan Dunn, J., Alvarez, L. A., Zhang, X. & Soldati, T. Reactive oxygen species and mitochondria: A nexus of cellular homeostasis. Redox Biol. 6, 472–485 (2015).
Jo, E. K., Kim, J. K., Shin, D. M. & Sasakawa, C. Molecular mechanisms regulating NLRP3 inflammasome activation. Cell. Mol. Immunol. 13, 148–159 (2016).
Zhou, R., Yazdi, A. S., Menu, P. & Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature. 469, 221–225 (2011).
Dominic, A., Le, N. T. & Takahashi, M. Loop between NLRP3 inflammasome and reactive oxygen species. Antioxid. Redox Signal. 36, 784–796 (2022).
Li, M., Luo, Q., Tao, Y., Sun, X. & Liu, C. Pharmacotherapies for drug-induced liver injury: A current literature review. Front. Pharmacol. 12, 806249 (2021).
Pei, Y. et al. Biological activities and potential oral applications of N-acetylcysteine: Progress and prospects. Oxid. Med. Cell Longev. 2018, 2835787 (2018).
Zhang, M. et al. AVE 0991 attenuates pyroptosis and liver damage after heatstroke by inhibiting the ROS-NLRP3 inflammatory signalling pathway. Biomed. Res. Int. 2019, 1806234 (2019).
Zhang, K. et al. Saikosaponin d alleviates liver fibrosis by negatively regulating the ROS/NLRP3 inflammasome through activating the ERβ pathway. Front. Pharmacol. 13, 894981 (2022).
Dzik, J. M. Molecules released by helminth parasites involved in host colonization. Acta Biochim. Pol. 53, 33–64 (2006).
Luo, G. et al. Effects of protoscoleces excretory-secretory products of Echinococcus granulosus on hepatocyte growth, function, and glucose metabolism. Acta Trop. 249, 107066 (2024).
Acknowledgements
We would like to thank the staff of Shihezi slaughterhouse in Xinjiang Province for donating their abandoned sheep liver.
Funding
This work was supported by the National Natural Science Foundation of China (Grant number 81560334 to H.L.), the Key research and development program of Sichuan Provincial Science and Technology Department (Grant number 2022YFS0231 to H.L.) and the Chengdu Medical College Graduate Student Innovation Fund (Grant number YCX2024-01-08 to J.C.). The funders had no role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
J.C. and J.Q.C. carried out most of the experiments and wrote the manuscript. H.W.L. carried out statistical analysis. Y.J. designed the project and experiments. J.C. prepared figures. J.C. and H.L. were responsible for acquiring funding for this project. All authors contributed to the manuscript and have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cao, J., Chen, J., Li, H. et al. ROS-NLRP3 participates in the pyroptosis response of excretory-secretory products from protoscoleces of Echinococcus granulosus in hepatocytes. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45127-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45127-7