Abstract
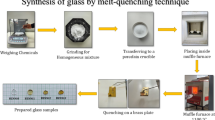
A novel series of Fe3+ doped mixed alkali zirconia-borate glasses with the composition 60B2O3–25Na2O–10Li2O–(5 − x)ZrO2–xFe2O3 (x = 0–1 mol%) were synthesized using the melt-quenching technique. The novelty of this work lies in combining the structural modifications with enhanced optical properties and orange-red luminescence in Fe3+ doped mixed alkali zirconia-borate glasses for warm photonic applications. X-ray diffraction confirmed the non-crystalline nature of glass, while SEM images and EDS spectra were utilised for morphological and elemental analysis. The density of the glasses decreased from 2.4197 to 2.4168 g cm−3 before gradually increasing to 2.4324 g cm−3. FTIR and Raman spectral studies showed the formation of non-bridging orthoborate units from pentaborate and di-pentaborate units. The optical absorption band at 450 nm is associated with the Fe3+ transition 6A1g (6S) → 4A1g (4G); 4Eg (4G). The decrease in direct bandgap from 3.90 to 3.10 eV and indirect bandgap from 3.44 to 2.77 eV, along with an increase in Urbach energy from 0.245 to 0.273 eV, indicates the disorder in structure. An increase in refractive index leads to an increase in third-order susceptibility and nonlinear refractive index. The photoluminescence spectra exhibited orange-red emission at (4T2g(4G) → 6A1g(6S)) with 550 nm, 560 nm and 570 nm excitation wavelengths. The CIE chromaticity and CCT values show that Fe3+ doped glasses are suitable for warm-emitting orange-red photonic applications.
Similar content being viewed by others
Data availability
The data used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Vasantharani, P. & Vichitra, S. N. Structural and elastic studies of strontium doped manganese borate glasses. IOSR J. Appl. Phys. 09, 0904014449. https://doi.org/10.9790/4861-0904014449 (2017).
Vinay, D., Devaraja, C. & Deka, U. A comprehensive review of structural, and optical properties of boronated glasses doped with 3-d transition metal oxides (TMOs). J. Alloys Compd. 1024, 180145. https://doi.org/10.1016/J.JALLCOM.2025.180145 (2025).
Aktas, B. et al. Effect of Er2O3 on structural, mechanical, and optical properties of Al2O3–Na2O–B2O3–SiO2 glass. J. Non-Cryst. Solids 584, 121516. https://doi.org/10.1016/j.jnoncrysol.2022.121516 (2022).
Aktas, B., Yalcin, S., Dogru, K., Uzunoglu, Z. & Yilmaz, D. Structural and radiation shielding properties of chromium oxide doped borosilicate glass. Radiat. Phys. Chem. 156, 144–149. https://doi.org/10.1016/j.radphyschem.2018.11.012 (2019).
Devaraja, C., Gowda, G. V. J., Keshavamurthy, K. & Eraiah, B. The optical and physical properties of holmium (Ho3+) ions doped bismuth-tellurite glasses. AIP Conf. Proc. 2162, 020172. https://doi.org/10.1063/1.5130382 (2019).
Brauer, D. S. & Möncke, D. Introduction to the structure of silicate, phosphate and borate glasses. In RSC Smart Materials (eds Boccaccini, A. R. et al.) 61–88 (Royal Society of Chemistry, 2016). https://doi.org/10.1039/9781782622017-00061.
Shiva Kumar, B. N. & Vinay, D. Effect of cerium oxide on physical, structural, and spectroscopic properties of tellurium-borate glasses for cool greenish light emitting devices. Sci. Rep. https://doi.org/10.1038/s41598-026-40883-y (2026).
Gomaa, H. M. et al. Influence of incorporation of Fe2O3 content on the structural and the dielectric relaxation properties of lithium boro-vanadate oxide glass: Toward ideal cathode glasses. Appl. Phys. A https://doi.org/10.1007/S00339-022-06350-X (2023).
Zhang, H. et al. Luminescence properties of chalcohalide devitrified glass containing BaCl2:Tm3+, Er3+ nanocrystals. J. Lumin. 273, 120688. https://doi.org/10.1016/J.JLUMIN.2024.120688 (2024).
Arya, S. K., Chhina, M. K., Choudhary, R., Dua, V. & Singh, K. Growth of different nanocrystalline phases in ZnO–Li2O–B2O3–TiO2–V2O5 glass and their effect on photoluminescence and photocatalytic activity. Ceram. Int. https://doi.org/10.1016/j.ceramint.2022.04.030 (2022).
Bengisu, M. Borate glasses for scientific and industrial applications: A review. J. Mater. Sci. https://doi.org/10.1007/s10853-015-9537-4 (2016).
Liang, H. et al. High performing ferrous ion-doped phosphosilicate glass for energy-efficient windows. Chem. Eng. J. 511, 161985. https://doi.org/10.1016/J.CEJ.2025.161985 (2025).
D’Silva, A. J., Maheshvaran, K. & Rayappan, I. A. Influence of alkali and alkaline-earth metal oxides on Sm3+ ions-doped borate glasses: Synthesis, structural, and optical investigations for reddish-orange solid-state-lighting applications. J. Mater. Sci. Mater. Electron. 35, 1630. https://doi.org/10.1007/s10854-024-13368-9 (2024).
Obayes, H. K., Wagiran, H., Hussin, R. & Saeed, M. A. Structural and optical properties of strontium/copper co-doped lithium borate glass system. Mater. Des. 94, 121–131. https://doi.org/10.1016/J.MATDES.2016.01.018 (2016).
Guntu, R. K. et al. Investigations on thermoluminescence, photoluminescence and radiation shielding properties of Dy2O3 doped Li2O–Al2O3–SiO2–Y2O3 glasses for thermoluminescent dosimeter applications. Appl. Phys. A 131(9), 701. https://doi.org/10.1007/s00339-025-08757-8 (2025).
Shelby, J. E. Introduction to Glass Science and Technology (Royal Society of Chemistry, 2020). https://doi.org/10.1039/9781839169229.
Sayer, M. & Mansingh, A. Oxide glasses. Noncryst. Semicond. https://doi.org/10.1016/B978-008043958-7/50030-3 (2002).
Graça, M. P. F. et al. Electrical and dielectric analysis of phosphate based glasses doped with alkali oxides. Mater. Des. 86, 427–435. https://doi.org/10.1016/J.MATDES.2015.07.043 (2015).
Varshneya, A. K. & Mauro, J. C. Composition–structure–property relationship principles. In Fundamentals of Inorganic Glasses 165–172 (2019). https://doi.org/10.1016/B978-0-12-816225-5.00006-7.
Karmakar, B. Fundamentals of glass and glass nanocomposites. In Glass Nanocomposites: Synthesis, Properties and Applications 3–53 (2016). https://doi.org/10.1016/B978-0-323-39309-6.00001-8.
Roopa, E. B. Experimental and theoretical approach on the physical, structural and optical properties of ZrO2–Na2O–B2O3 glasses doped with Dy2O3. J. Non-Cryst. Solids 551, 120394. https://doi.org/10.1016/J.JNONCRYSOL.2020.120394 (2021).
Srinivasa Rao, L., Aruna Prabha, K., Naidu, C. D. & Hussain, S. Structural aspects of Bi2O3–B2O3–MnO2–ZrO2 glasses evaluated by Raman and photoluminescence spectroscopy for solid state lighting. Inorg. Chem. Commun. 173, 113787. https://doi.org/10.1016/J.INOCHE.2024.113787 (2025).
Gomaa, H. M. et al. Structure and electrical properties of some Fe-doped sodium borate devitrified glasses: Self-heating action. Appl. Phys. A Mater. Sci. Process. 129, 322. https://doi.org/10.1007/S00339-023-06605-1 (2023).
Bhogi, A. & Kistaiah, P. Spectroscopic properties of alkali alkaline earth borate glasses doped with Fe3+ ions. J. Aust. Ceram. Soc. 56, 127–138. https://doi.org/10.1007/s41779-019-00426-3 (2020).
Huang, Q. et al. Characterization of Fe2O3 doping on structure, optical and luminescence properties of magnesium aluminosilicate-based glasses. J. Non Cryst. Solids 563, 120786. https://doi.org/10.1016/j.jnoncrysol.2021.120786 (2021).
Pattar, V. et al. Investigation of structural, physical and optical properties of sodium boro-tellurite glasses doped with iron oxide. Ceram. Int. 50, 30434–30444. https://doi.org/10.1016/J.CERAMINT.2024.05.341 (2024).
Xu, Y. et al. Effect of Fe2O3 on the structure and properties of Mo-containing borosilicate glasses for nuclear waste immobilization. Int. J. Appl. Glass Sci. 16, e16680. https://doi.org/10.1111/ijag.16680 (2025).
Khan, S., Kaur, G. & Singh, K. Effect of ZrO2 on dielectric, optical and structural properties of yttrium calcium borosilicate glasses. Ceram. Int. 43, 722–727. https://doi.org/10.1016/j.ceramint.2016.09.219 (2017).
Kumar, D., Rao, S. M. & Singh, S. P. Structural, optical and thermoluminescence study of Dy3+ ion doped sodium strontium borate glass. J. Non Cryst. Solids 464, 51–55. https://doi.org/10.1016/j.jnoncrysol.2017.03.029 (2017).
Aqdim, S., Naji, M., Chakir, A. & El Bouari, A. Design, synthesis and structural properties of borate glasses: Towards an alkali-free bioactive glass. J. Non Cryst. Solids 597, 121876. https://doi.org/10.1016/j.jnoncrysol.2022.121876 (2022).
Kilic, G. et al. The role of B2O3 on the structural, thermal, and radiation protection efficacy of vanadium phosphate glasses. Appl. Phys. A Mater. Sci. Process. 127, 265. https://doi.org/10.1007/s00339-021-04409-9 (2021).
Mirkazemi, M. et al. Effect of ZrO2 nucleant on crystallisation behaviour, microstructure and magnetic properties of BaO–Fe2O3–B2O3–SiO2 glass ceramics. Ceram. Int. https://doi.org/10.1016/j.ceramint.2005.10.011 (2007).
Hovington, P. et al. Can we detect Li K X-ray in lithium compounds using energy dispersive spectroscopy?. Scanning https://doi.org/10.1002/sca.21302 (2016).
Rohilla, R., Khasa, S. & Hooda, A. Structural and impedance spectroscopic investigations of eco-friendly alkali phosphoborate glass–ceramics containing zirconium ion. Environ. Sci. Pollut. Res. 30, 98609–98618. https://doi.org/10.1007/s11356-022-21867-7 (2023).
Thumma, J. & Guntu, R. K. Design and evaluation of TiO2-enhanced phosphate glasses for dual-functionality: Photonic applications and radiation safety. Physica B Condens. Matter 716, 417735. https://doi.org/10.1016/J.PHYSB.2025.417735 (2025).
Usman, I., Sanusi, M. S. M., Ahmad, N. E. & Shafiq bin Shaharin, M. Structural and optical properties of tungsten–sodium–zinc-borate glass doped α-Fe2O3 nanoparticle. Ceram. Int. 49, 26256–26266. https://doi.org/10.1016/J.CERAMINT.2023.05.079 (2023).
Vamshi Babu, K., Chanakya, N., Aravind, S. & Upender, G. Structural, thermal, optical and DC conductivity studies of 30TeO2–40B2O3–(30−x)BaO–xNa2O (0 ≤ x ≤ 30 mol%) glass system. J. Inorg. Organomet. Polym. Mater. https://doi.org/10.1007/s10904-023-02914-3 (2023).
Shiva Kumar, B. N. et al. Implications of silver nitrate doping on the physical, structural, and optical attributes of Na2O−ZnO−borate glasses. J. Mol. Struct. 1325, 140985. https://doi.org/10.1016/J.MOLSTRUC.2024.140985 (2025).
El-Moneim, A. A., Ahmed, R. M., Sallam, O. I. & El-Mallawany, R. Physical, structural and elastic properties of Fe2O3-doped oxyfluoroborate glasses. Appl. Phys. A Mater. Sci. Process. 131, 1–14. https://doi.org/10.1007/S00339-025-08247-X/FIGURES/13 (2025).
Sayyed, M. I., Abdo, M. A., Elhosiny Ali, H. & Sadeq, M. S. Fe2O3 within Na2O–Al2O3–B2O3 glasses to study the structural and optical features changes. Opt. Mater. https://doi.org/10.1016/j.optmat.2022.112419 (2022).
Al-Ghamdi, H. et al. Effect of iron ion doping on mechanical, dielectric properties, and radiation protection effectiveness of lead barium borate glasses. Appl. Phys. A https://doi.org/10.1007/s00339-023-07168-x (2024).
Vinay, D. et al. Silver oxide embedded alkali zinc boro tellurite glasses: Physical, structural and optical properties for possible optical switching applications. J. Mol. Struct. 1339, 142388. https://doi.org/10.1016/J.MOLSTRUC.2025.142388 (2025).
Devaraja, C., Jagadeesha Gowda, G. V., Eraiah, B. & Keshavamurthy, K. FTIR and Raman studies of Eu3+ ions doped alkali boro tellurite glasses. AIP Conf. Proc. 2115, 030230. https://doi.org/10.1063/1.5113069 (2019).
Bhogi, A., Pothuganti, P. K. & Kistaiah, P. Structural properties of Li2O–BaO–B2O3–Fe2O3 glasses. AIP Conf. Proc. 2220, 080058. https://doi.org/10.1063/5.0001158/890125 (2020).
Hassaan, M. Y., Bendary, A. A., Abdel-Wahed, M. H., Atta, M. R. & Abdel-Moety, A. S. Influence of ZrO2 additions on the structural and optical properties of Na–B–Te glass for optoelectronic applications. J. Egypt. Soc. Basic Sci. Phys. 2, 16–22. https://doi.org/10.21608/JESBSP.2025.315233.1024 (2025).
Kamitsos, E. I., Karakassides, M. A. & Chryssikos, G. D. Vibrational spectra of magnesium-sodium-borate glasses. 2. Raman and mid-infrared investigation of the network structure. J. Phys. Chem. 91, 1073–1079. https://doi.org/10.1021/J100289A014/SUPPL_FILE/J100289A014_SI_001.PDF (1987).
Abdel-Aziz, A. M., Shams, M. S., Ahmed, E. M., Rammah, Y. S. & Elsad, R. A. Fabrication, physical, FTIR, ultrasonic waves, and mechanical properties of quaternary B2O3–Bi2O3–NaF–ZrO2 glasses: Experimental study. Appl. Phys. A https://doi.org/10.1007/s00339-022-05732-5 (2022).
Siddalingeshwara, B. P. et al. Structural correlation to linear and non-linear optical properties of Bi3+ ion doped Li2O–SrO–B2O3 glasses: Optical switching and limiting applications. Ceram. Int. 50, 37880–37899. https://doi.org/10.1016/J.CERAMINT.2024.07.1512 (2024).
Ardelean, I., Toderas, M. & Pǎşcuţǎ, P. Structural study of the Fe2O3–B2O3–BaO glass system by FTIR spectroscopy. Mod. Phys. Lett. B https://doi.org/10.1142/S0217984903006098 (2003).
Varsamis, C. P. E., Makris, N., Valvi, C. & Kamitsos, E. I. Short-range structure, the role of bismuth and property–structure correlations in bismuth borate glasses. Phys. Chem. Chem. Phys. https://doi.org/10.1039/d1cp00301a (2021).
Möncke, D. et al. Transition and post-transition metal ions in borate glasses: Borate ligand speciation, cluster formation, and their effect on glass transition and mechanical properties. J. Chem. Phys. https://doi.org/10.1063/1.4962323 (2016).
Abdel Wahab, E. A., Shaaban, K. S. & Yousef, E. S. Enhancement of optical and mechanical properties of sodium silicate glasses using zirconia. Opt. Quantum Electron. https://doi.org/10.1007/s11082-020-02575-3 (2020).
Budida, J., Rao, C. S., Chand, N. R. & Guntu, R. K. Spectral and structural tailoring of Sm2O3-doped LiF-La2O3–PbO–B2O3 glasses for advanced luminescent devices. J. Electron. Mater. 54(11), 10611–10647. https://doi.org/10.1007/s11664-025-12286-8 (2025).
Vinay, D. et al. Investigation of physical, structural, optical, and luminescence properties of nickel oxide doped alkali zinco-borate glasses. Sci. Rep. 15(1), 1–20. https://doi.org/10.1038/s41598-025-91852-w (2025).
Kaky, K. M. et al. Structural, thermal, and optical analysis of zinc boro-aluminosilicate glasses containing different alkali and alkaline modifier ions. J. Non-Cryst. Solids https://doi.org/10.1016/j.jnoncrysol.2016.10.044 (2017).
Vignarooban, K., Skipper, C., Welton, A. & Boolchand, P. Linking the ring-morphology of (Li2O)x(B2O3)100−x and (Na2O)x(B2O3)100−x borate glasses with topological phases and melt dynamics. J. Non-Cryst. Solids 654, 123450. https://doi.org/10.1016/j.jnoncrysol.2025.123450 (2025).
Meera, B. N. & Ramakrishna, J. Raman spectral studies of borate glasses. J. Non-Cryst. Solids 159, 1. https://doi.org/10.1016/0022-3093(93)91277-A (1993).
Ashok, K., Yuganand, N., Guntu, R. K. & Daniel Francis, E. Raman deconvolution, radiation shielding, TL effective and dielectric investigations of Dy3+ doped alumina lead borate glasses. Radiat. Phys. Chem. 224, 112057. https://doi.org/10.1016/J.RADPHYSCHEM.2024.112057 (2024).
Hivrekar, M. M., Sable, D. B., Solunke, M. B. & Jadhav, K. M. Network structure analysis of modifier CdO doped sodium borate glass using FTIR and Raman spectroscopy. J. Non-Cryst. Solids 474, 58–65. https://doi.org/10.1016/J.JNONCRYSOL.2017.08.028 (2017).
Abdel-Ghany, A. M., Abu-Khadra, A. S. & Sadeq, M. S. Influence of Fe cations on the structural and optical properties of alkali-alkaline borate glasses. J. Non-Cryst. Solids 548, 120320. https://doi.org/10.1016/J.JNONCRYSOL.2020.120320 (2020).
Sadeq, M. S. et al. Composition dependence of transparency, optical, ligand field and radiation shielding properties in CdO–Fe2O3–Na2O–B2O3 glasses. Ceram. Int. https://doi.org/10.1016/j.ceramint.2023.06.071 (2023).
Jagadeesha Gowda, G. V. et al. Structural, physical, and optical properties of lead boro-tellurite glasses doped with europium trioxide. Rasayan J. Chem. 16, 692. https://doi.org/10.31788/RJC.2023.1628300 (2023).
Gowda, G. V. J. et al. Exploration of physical, structural, thermal, and optical properties of alkali zinc boro tellurite glasses doped with europium trioxide. Appl. Phys. A Mater. Sci. Process. 130, 1–15. https://doi.org/10.1007/S00339-024-07920-X/FIGURES/18 (2024).
Abul-Magd, A. A., Abu-Khadra, A. S. & Abdel-Ghany, A. M. Physical, thermal, optical, shielding and elastic properties of Bi2O3–B2O3–TeO2 glass system doped with Fe2O3. Radiat. Phys. Chem. 217, 111444. https://doi.org/10.1016/j.ceramint.2021.03.326 (2024).
Sorkhabi, S. Y., Rezvani, M. & Oskoui, P. R. Effects of Fe2O3 and CuO on structural, thermal and optical properties of P2O5–ZnO–Na2O glass system. Results Opt. 18, 100779. https://doi.org/10.1016/j.rio.2025.100779 (2025).
Aloraini, D. A., Almuqrin, A., Albarzan, B., Shaaban, K. S. & Wahab, E. A. A. Impact of Fe2O3 on structural properties, nonlinear optical behavior, and radiation shielding efficiency of lead phosphate glasses. JOM 77(12), 9894–9909. https://doi.org/10.1007/s11837-025-07806-0 (2025).
Alfryyan, N. et al. Structural, physical, optical properties as well γ-ray attenuation capability of Bismuth Borate containing Fe2O3 and CuO: Potential use in optical and radiation shielding applications. J. Mater. Eng. Perform. 34(23), 27749–27758. https://doi.org/10.1007/s11665-025-11962-4 (2025).
Bawazeer, O. & Sadeq, M. S. Compositional dependency of transparency, optical and radiation shielding parameters inside Gd2O3–Fe2O3–Na2O–SiO2–B2O3 glass. Ceram. Int. 50, 159–173. https://doi.org/10.1016/j.ceramint.2023.10.079 (2024).
Siddalingeshwara, B. P., Sivasankara Reddy, N. & Jagannathan, A. The effect of NiO on the structural and optical properties of lithium strontium borate glasses. J. Opt. 2024, 1–13. https://doi.org/10.1007/S12596-024-02008-4 (2024).
Abdel Wahab, E. A. & Shaaban, K. S. Structural and optical features of aluminum lead borate glass doped with Fe2O3. Appl. Phys. A Mater. Sci. Process. 127, 1–12. https://doi.org/10.1007/S00339-021-05062-Y/FIGURES/11 (2021).
Dimitrov, V. & Komatsu, T. Classification of simple oxides: A polarizability approach. J. Solid State Chem. 163, 100–112. https://doi.org/10.1006/JSSC.2001.9378 (2002).
Gowda, G. V. J., Reddy, G. V. A., Eraiah, B. & Devaraja, C. R. Investigations of structural modifications, physical and optical properties of lead boro-tellurite glasses doped with europium trioxide for possible optical switching applications. J. Met. Mater. Miner. https://doi.org/10.55713/jmmm.v33i2.1610 (2023).
Umar, S. A. et al. Optical basicity, polarizability and spectroscopic investigations of CuO doped TeO2–B2O3 glass system. Mater. Chem. Phys. 297, 127309. https://doi.org/10.1016/J.MATCHEMPHYS.2023.127309 (2023).
Fattah Khedr, A. A. & Abul-Magd, A. A. Tuning the optical band gap and shielding efficiency of heavy metal borate glasses through controlled Fe2O3 doping. Opt. Mater. (Amst.) 160, 116717. https://doi.org/10.1016/J.OPTMAT.2025.116717 (2025).
Tichá, H. & Tichý, L. Semiempirical relation between non-linear susceptibility (refractive index), linear refractive index and optical gap and its application to amorphous chalcogenides. J. Optoelectron. Adv. Mater. 4, 381–386 (2002).
Doddoji, R., Meejitpaisan, P. & Kaewkhao, J. Determination of optical constants via the single oscillator Drude–Voigt dispersion model in fluoroborate glass for optical lenses: Nonlinear optical properties. Physica B Condens. Matter https://doi.org/10.1016/j.physb.2023.415398 (2023).
El-Sayed, M. A. et al. A detailed analysis of linear/nonlinear optical properties of boro-tellurite glasses reinforced with ZrO2 for optoelectronics applications. Appl. Phys. A Mater. Sci. Process. 131, 1–11. https://doi.org/10.1007/S00339-025-08386-1/FIGURES/9 (2025).
Wang, Y. et al. UV- and X-ray-activated broadband NIR garnet-type Ca3Ga2Sn3O12:Fe3+ phosphors with efficient persistent luminescence. Dyes Pigments https://doi.org/10.1016/j.dyepig.2024.112091 (2024).
Alyabyeva, L., Burkov, V. & Mill, B. Optical spectroscopy of La3Ga5SiO14 disordered crystals doped with Fe3+ ions. Opt. Mater. (Amst.) https://doi.org/10.1016/j.optmat.2015.02.023 (2015).
Wei, Y. et al. New strategy for designing orangish-red-emitting phosphor via oxygen-vacancy-induced electronic localization. Light Sci. Appl. https://doi.org/10.1038/s41377-019-0126-1 (2019).
McCamy, C. S. Correlated color temperature as an explicit function of chromaticity coordinates. Color Res. Appl. https://doi.org/10.1002/col.5080170211 (1992).
Pawar, P. P., Gedam, R. S. & Agarwal, K. Insight into the physical, structural, thermal and spectroscopic characteristics of intense green luminescent Tb3+ incorporated lithium alumino-borate glasses for green LED application. J. Mol. Struct. 1340, 142523. https://doi.org/10.1016/j.molstruc.2025.142523 (2025).
Chen, Q., Chen, L., Feng, Y., Gao, T. & Huang, T. KBiFe2O5 triggered-phase transition of Bi2Fe4O9 and Fe3O4 in tellurite glass with huge nonlinear and magnetic properties. Ceram. Int. 51, 2737–2755. https://doi.org/10.1016/J.CERAMINT.2024.05.383 (2025).
Acknowledgements
The authors would like to thank and acknowledge Manipal Institute of Technology Bengaluru, and Manipal Academy of Higher Education, Manipal, for their great encouragement and support towards research. The authors express thanks to the Centre for Nano and Soft Matter Sciences (CeNS), Bengaluru, for providing Raman Spectroscopy characterizations and Jawaharlal Nehru Centre for Advanced Scientific Research (JNCASR), Bengaluru, for their support with Powder XRD measurements.
Funding
Open access funding provided by Manipal Academy of Higher Education, Manipal
Author information
Authors and Affiliations
Contributions
Vinay D: Conceptualisation, data curation, investigation, formal analysis, writing—original draft. C Devaraja: Conceptualization, data curation, methodology, writing—review and editing, validation, supervision. Utpal Deka: Writing—review and editing, validation, supervision. R.S. Gedam: Validation, methodology, data curation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vinay, D., Devaraja, C., Deka, U. et al. Structural, optical and luminescence properties of Fe3+-doped mixed alkali zirconia-borate glasses for warm orange-red photonic applications. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45270-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45270-1