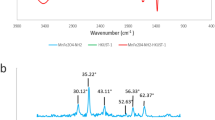
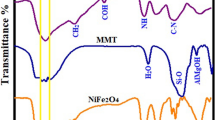
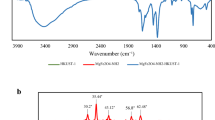
Abstract
Contamination of water resources by organic dyes is a major environmental issue associated with industrial activities. Methylene blue (MB), a widely used cationic aromatic dye, is particularly problematic due to its high chemical stability and toxicity, highlighting the need for efficient and cost-effective treatment strategies. In this study, a magnetic amino-functionalized hyper-crosslinked resin (Fe3O4@XDV-NH2) was synthesized and evaluated as an efficient adsorbent for MB removal from aqueous solutions. The material was prepared via post-synthetic amino functionalization of a hyper-crosslinked polymer followed by in situ deposition of Fe3O4 nanoparticles, combining high surface area, permanent porosity, and chemical stability with easy magnetic separation. Structural and magnetic characterizations confirmed the uniform incorporation of Fe3O4 without significant loss of porosity. Amino functionalization enhanced hydrophilicity and introduced polar active sites, promoting strong interactions with MB. Batch adsorption experiments revealed a maximum adsorption capacity of 79.60 mg/g at 298 K and pH 9 and 0.06 g L−1 adsorbent dosage. Adsorption kinetics followed the pseudo-second-order model (R2 = 0.976). Equilibrium data were best fitted by the Langmuir isotherm model (R2 = 0.996), indicating monolayer adsorption with a maximum theoretical capacity of 183.48 mg/g. Thermodynamic analysis showed that MB adsorption was spontaneous and exothermic. High removal efficiencies were maintained in complex matrices such as tap water (94.11%) and synthetic wastewater (78.41%). Moreover, the adsorbent retained approximately 89% of its initial capacity after six adsorption–desorption cycles, demonstrating good reusability. These results indicate that Fe3O4@XDV-NH2 is a promising and sustainable adsorbent for dye-contaminated wastewater treatment.
Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Jorge, A. M., Athira, K., Alves, M. B., Gardas, R. L. & Pereira, J. F. Textile dyes effluents: A current scenario and the use of aqueous biphasic systems for the recovery of dyes. J. Water Process. Eng. 55, 104125. https://doi.org/10.1016/j.jwpe.2023.104125 (2023).
Fröse, A., Schmidtke, K., Sukmann, T., Junger, I. J. & Ehrmann, A. Application of natural dyes on diverse textile materials. Optik 181, 215–219. https://doi.org/10.1016/j.ijleo.2018.12.099 (2018).
Zollinger, H. Color Chemistry: Synthesis, Properties, and Applications of Organic Dyes and Pigments, 92–100 (VCH, 1987).
Kayranli, B. Adsorption of textile dyes onto iron-base waterworks sludge from aqueous solution: Isotherm, kinetic and thermodynamic study. Chem. Eng. J. 173, 782–791. https://doi.org/10.1016/j.cej.2011.08.051 (2011).
Hunger, K. Industrial Dyes: Chemistry, Properties, Applications (Wiley-VCH, 2003). https://doi.org/10.1002/3527602011
Karri, R. R., Ravindran, G. & Dehghani, M. H. Wastewater—Sources, toxicity, and their consequences to human health. In Soft Computing Techniques in Solid Waste and Wastewater Management, 3–33 (eds. Karri, R. R. et al.). https://doi.org/10.1016/B978-0-12-824463-0.00001-X (2021).
Buitrón, G., Quezada, M. & Moreno, G. Aerobic degradation of the azo dye acid red 151 in a sequencing batch biofilter. Bioresour. Technol. 92, 143–149. https://doi.org/10.1016/j.biortech.2003.09.001 (2003).
Muthuraman, G. & Teng, T. T. Extraction of methyl red from industrial wastewater using xylene as an extractant. Prog. Nat. Sci. 19, 1215–1220. https://doi.org/10.1016/j.pnsc.2009.04.002 (2009).
Badr, Y., El-Wahed, M. A. & Mahmoud, M. A. Photocatalytic degradation of methyl red dye by silica nanoparticles. J. Hazard. Mater. 154, 245–253. https://doi.org/10.1016/j.jhazmat.2007.10.020 (2008).
Ercan, Ö., Deniz, S., Yetimoğlu, E. K. & Aydın, A. Degradation of reactive dyes using advanced oxidation method. Clean: Soil. Air Water. 43, 1031–1036. https://doi.org/10.1002/clen.201400195 (2015).
Duta, A. & Visa, M. Simultaneous removal of two industrial dyes by adsorption and photocatalysis on a fly-ash–TiO2 composite. J. Photochem. Photobiol. A. 306, 21–30. https://doi.org/10.1016/j.jphotochem.2015.03.007 (2015).
Boumaza, S., Kaouah, F., Omeiri, S., Trari, M. & Bendjama, Z. Removal of dyes by an integrated process coupling adsorption and photocatalysis in batch mode. Res. Chem. Intermed. 41, 2353–2375. https://doi.org/10.1007/s11164-013-1351-5 (2015).
Nataraj, S., Hosamani, K. & Aminabhavi, T. Nanofiltration and reverse osmosis thin film composite membrane module for the removal of dye and salts from the simulated mixtures. Desalination 249, 12–17. https://doi.org/10.1016/j.desal.2009.06.008 (2009).
Anjanyeulu, Y., Sreedhara Chary, N. & Suman Raj, D. S. Decolourization of industrial effluents—available methods and emerging technologies—A review. Rev. Environ. Sci. Biotechnol. 4, 245–273. https://doi.org/10.1007/s11157-005-1246-z (2005).
Vikrant, K. et al. Recent advancements in bioremediation of dye: Current status and challenges. Bioresour. Technol. 253, 355–367. https://doi.org/10.1016/j.biortech.2018.01.029 (2018).
Legerská, B., Chmelová, D. & Ondrejovič, M. Decolourization and detoxification of monoazo dyes by laccase from the white-rot fungus Trametes versicolor. J. Biotechnol. 285, 84–90. https://doi.org/10.1016/j.jbiotec.2018.08.011 (2018).
Rani, B. et al. Bioremediation of dyes by fungi isolated from contaminated dye effluent sites for bio-usability. Braz. J. Microbiol. 45, 1055–1063. https://doi.org/10.1590/s1517-83822014000300039 (2014).
Chebli, D., Fourcade, F., Brosillon, S., Nacef, S. & Amrane, A. Supported photocatalysis as a pre-treatment before biological degradation for the removal of some dyes from aqueous solutions: Acid red 183, Biebrich Scarlet, methyl red sodium salt, and orange II. J. Chem. Technol. Biotechnol. 85, 555–563. https://doi.org/10.1002/jctb.2342 (2010).
Manjunatha, A. S., Sukhdev, A. & Puttaswamy Oxidative decolorization of methyl red dye with chloramine-T kinetic and mechanistic chemistry. Indian J. Chem. Technol. 20, 416–422 (2013).
Ighalo, J. O. et al. Recent advances in hydrochar application for the adsorptive removal of wastewater pollutants. Process Saf. Environ. Prot. 184, 419–456. https://doi.org/10.1016/j.cherd.2022.06.028 (2022).
Gkika, D. A., Mitropoulos, A. C. & Kyzas, G. Z. Why reuse spent adsorbents? The latest challenges and limitations. Sci. Total Environ. 822, 153612. https://doi.org/10.1016/j.scitotenv.2022.153612 (2022).
Sajid, M. Chitosan-based adsorbents for analytical sample preparation and removal of pollutants from aqueous media: Progress, challenges and outlook. Trends Environ. Anal. Chem. 36, e00185. https://doi.org/10.1016/j.teac.2022.e00185 (2022).
Younas, F. et al. Current and emerging adsorbent technologies for wastewater treatment: Trends, limitations, and environmental implications. Water 13 (2), 215. https://doi.org/10.3390/w13020215 (2021).
Bhatnagar, A., Hogland, W., Marques, M. & Sillanpää, M. An overview of the modification methods of activated carbon for its water treatment applications. Chem. Eng. J. 219, 499–511. https://doi.org/10.1016/j.cej.2012.12.038 (2012).
Peng, J., He, Y., Zhou, C., Su, S. & Lai, B. The carbon nanotubes-based materials and their applications for organic pollutant removal: A critical review. Chin. Chem. Lett. 32 (5), 1626–1636. https://doi.org/10.1016/j.cclet.2020.10.026 (2020).
Sodha, V. et al. Comprehensive review on zeolite-based nanocomposites for treatment of effluents from wastewater. Nanomaterials 12 (18), 3199. https://doi.org/10.3390/nano12183199 (2022).
Rout, D. R., Jena, H. M., Baigenzhenov, O. & Hosseini-Bandegharaei, A. Graphene-based materials for effective adsorption of organic and inorganic pollutants: A critical and comprehensive review. Sci. Total Environ. 863, 160871. https://doi.org/10.1016/j.scitotenv.2022.160871 (2022).
Abdollahi, N., Moussavi, G. & Giannakis, S. A review of heavy metals’ removal from aqueous matrices by Metal-Organic Frameworks (MOFs): State-of-the art and recent advances. J. Environ. Chem. Eng. 10 (3), 107394. https://doi.org/10.1016/j.jece.2022.107394 (2022).
Naseem, K., Arif, M., Anwar, A., Haider, S. & Akhtar, M. S. Investigating adsorptive potential of Raphanus caudatus leaves biomass for methyl orange dye: isotherm and kinetic study. Z.Phys. Chem. 237 (8), 1183–1205. https://doi.org/10.1515/zpch-2023-0255 (2023).
Juela, D. M. Promising adsorptive materials derived from agricultural and industrial wastes for antibiotic removal: A comprehensive review. Sep. Purif. Technol. 284, 120286. https://doi.org/10.1016/j.seppur.2021.120286 (2021).
Mosaffa, E., Malekshah, R. E., Chavez, F. A. & Banerjee, A. Biogenic chitosan cross-linked cherry gum and N-doped biochar beads: An eco-friendly platform for versatile dual-site dye removal. Int. J. Biol. Macromol. 339 (Pt 2), 150037. https://doi.org/10.1016/j.ijbiomac.2025.150037 (2025).
Mosaffa, E., Patel, R. I., Purohit, A. M., Basak, B. B. & Banerjee, A. Efficient decontamination of cationic dyes from synthetic textile wastewater using poly(acrylic acid) composite containing amino functionalized biochar: A mechanism kinetic and isotherm study. J. Polym. Environ. 31, 2486–2503. https://doi.org/10.1007/s10924-022-02744-3 (2023).
Mosaffa, E. et al. Physiochemically cross-linked cherry gum–LDH@Biochar chitosan/PVA biosorbent for antibiotic removal: Monte Carlo and mechanistic insights. Desalination 619, 119545. https://doi.org/10.1016/j.desal.2025.119545 (2025).
Mosaffa, E. et al. Bacterial cellulose microfiber reinforced hollow chitosan beads decorated with cross-linked melamine plates for the removal of the Congo red. Int. J. Biol. Macromol. 254 (Pt 2), 127794. https://doi.org/10.1016/j.ijbiomac.2023.127794 (2023).
Fontanals, N., Marcé, R. M., Borrull, F. & Cormack, P. A. G. Hyper-crosslinked materials: Preparation, characterisation and applications. Polym. Chem. 6 (41), 7231–7244. https://doi.org/10.1039/c5py00771b (2015).
Abid, A., Razzaque, S., Hussain, I. & Tan, B. Eco-friendly phosphorus and nitrogen-rich inorganic–organic hybrid hypercross-linked porous polymers via a low-cost strategy. Macromolecules 54 (12), 5848–5855. https://doi.org/10.1021/acs.macromol.1c00385 (2021).
Wang, R. et al. Swellable array strategy based on designed flexible double hypercross-linked polymers for synergistic adsorption of toluene and formaldehyde. Environ. Sci. Technol. 57 (16), 6682–6694. https://doi.org/10.1021/acs.est.3c00565 (2023).
Shi, P. et al. Thickness controllable Hyper-crosslinked porous polymer nanofilm with high CO2 capture capacity. J. Colloid Interface Sci. 563, 272–280. https://doi.org/10.1016/j.jcis.2019.12.038 (2019).
Hubert, O. et al. Sulfonated Hyper-crosslinked polymer enhanced structural composite supercapacitors. Compos. Sci. Technol. 242, 110152. https://doi.org/10.1016/j.compscitech.2023.110152 (2023).
Dong, Y. et al. Palladium supported on pyrrole functionalized Hyper-crosslinked polymer: Synthesis and its catalytic evaluations towards Suzuki–Miyaura coupling reactions in aqueous media. J. Mol. Liq. 368, 120679. https://doi.org/10.1016/j.molliq.2022.120679 (2022).
Razzaque, S., Cheng, Y., Hussain, I. & Tan, B. Synthesis of surface functionalized hollow microporous organic capsules for doxorubicin delivery to cancer cells. Polym. Chem. 11 (12), 2110–2118. https://doi.org/10.1039/c9py01772k (2020).
Wang, X., Li, G., Guo, D., Zhang, Y. & Huang, J. A novel polar-modified post-cross-linked resin and its enhanced adsorption to salicylic acid: Equilibrium, kinetics and breakthrough studies. J. Colloid Interface Sci. 470, 1–9. https://doi.org/10.1016/j.jcis.2016.02.046 (2016).
Moradi, M. R., Torkashvand, A., Penchah, H. R. & Ghaemi, A. Amine functionalized benzene-based Hyper-crosslinked polymer as an adsorbent for CO2/N2 adsorption. Sci. Rep. 13 (1). https://doi.org/10.1038/s41598-023-36434-4 (2023).
Castaldo, R. et al. Amino-functionalized hyper-crosslinked resins for enhanced adsorption of carbon dioxide and polar dyes. Chem. Eng. J. 418, 129463. https://doi.org/10.1016/j.cej.2021.129463 (2021).
Huang, J. & Turner, S. R. Hypercrosslinked polymers: A review. Polym. Rev. 58 (1), 1–41. https://doi.org/10.1080/15583724.2017.1344703 (2017).
Burevska-Atkovska, K. et al. Amino-modified microporous hyper-crosslinked resins for heavy metal ions adsorption. Colloids Surf. Physicochem. Eng. Aspects. 700, 134720. https://doi.org/10.1016/j.colsurfa.2024.134720 (2024).
Castaldo, R., Berezovska, I., Silverstein, M. S. & Gentile, G. PolyHIPEs containing hyper-cross-linked resins: hierarchical porosity with broad and versatile sorption properties. ACS Appl. Polym. Mater. 5 (9), 6920–6931. https://doi.org/10.1021/acsapm.3c00974 (2023).
Zhu, Y. et al. Three-dimensional sulfonic-functionalized porphyrin-based porous organic polymer for high-performance methylene blue and ciprofloxacin capture. Sep. Purif. Technol. 333, 125857. https://doi.org/10.1016/j.seppur.2023.125857 (2023).
Zhang, X. et al. Preparation of amino/hydroxy dual-functionalized Hyper-crosslinked polymers for effective removal of organic dyes from water. J. Hazard. Mater. 488, 137405. https://doi.org/10.1016/j.jhazmat.2025.137405 (2025).
You, X., Han, L. & Liu, Q. Adsorption of Rhodamine B from aqueous solutions using polarity-tunable hyper-cross-linked resins. New J. Chem. 47 (32), 15250–15260. https://doi.org/10.1039/d3nj02711b (2023).
Bushra, R. et al. Recent advances in magnetic nanoparticles: Key applications, environmental insights, and future strategies. Sustain. Mater. Technol. 40, e00985. https://doi.org/10.1016/j.susmat.2024.e00985 (2024).
Maglione, A. et al. Hyper-cross-linked cellulose nanofibrils with spontaneous and reversible adsorption of aromatic pollutants from water as a valid alternative to fossil-based adsorbents. ACS Appl. Mater. Interfaces. 17 (26), 38619–38634. https://doi.org/10.1021/acsami.5c05009 (2025).
Cirillo, C. et al. Magnetic resonance imaging monitoring of 4-chlorophenol removal by an efficient Horseradish Peroxidase-modified hyper-cross-linked resin catalyst. J. Environ. Chem. Eng. 13 (3), 116650. https://doi.org/10.1016/j.jece.2025.116650 (2025).
Guerritore, M. et al. Hierarchical micro-to-macroporous silica nanoparticles obtained by their grafting with hyper-crosslinked resin. Microporous Mesoporous Mater. 335, 111864. https://doi.org/10.1016/j.micromeso.2022.111864 (2022).
Dirgayanti, D. S., Koesnarpadi, S. & Hindryawati, N. Synthesis and characterization of Fe3O4-activated carbon and its application to adsorb methylene blue. IOP Conf. Ser. Earth Environ. Sci. 623(1), 012070. https://doi.org/10.1088/1755-1315/623/1/012070 (2021).
Iuliano, M., Ponticorvo, E., Cirillo, C. & Sarno, M. A new nanocomposite from Vesuvian slope pinecones for azo-dyes removal. Ind. Eng. Chem. Res. 61 (5), 1965–1976. https://doi.org/10.1021/acs.iecr.1c03574 (2022).
Chen, F., Xie, S., Huang, X. & Qiu, X. Ionothermal synthesis of Fe3O4 magnetic nanoparticles as efficient heterogeneous Fenton-like catalysts for degradation of organic pollutants with H2O2. J. Hazard. Mater. 322 (Pt A), 152–162. https://doi.org/10.1016/j.jhazmat.2016.02.073 (2016).
Iuliano, M. et al. Wax esters from waste fish oil catalysed by immobilized Candida rugosa lipase. Process Biochem. 130, 386–400. https://doi.org/10.1016/j.procbio.2023.04.028 (2023).
Shi, M., Tang, W., Guo, S. & Chen, F. Construction of hierarchically porous α-Fe2O3 nanosheets from Fe-PVP hybrid gel for sustainable Fenton-like degradation of tetracycline antibiotics with low H2O2 consumption. Environ. Res. 286 (Pt 2), 122854. https://doi.org/10.1016/j.envres.2025.122854 (2025).
Fecková, M. et al. Capture of DNAs by magnetic Hyper-crosslinked poly(styrene-co-divinylbenzene) microspheres. J. Mater. Sci. 56 (9), 5817–5829. https://doi.org/10.1007/s10853-020-05649-5 (2021).
Cirillo, C. et al. Magnetic resonance imaging monitoring of 4-chlorophenol removal by an efficient horseradish peroxidase-modified hyper-cross-linked resin catalyst. J. Environ. Chem. Eng. 116650. https://doi.org/10.1016/j.jece.2025.116650 (2025).
Zhang, W. et al. Adsorption equilibrium and heat of phenol onto aminated polymeric resins from aqueous solution. Colloids Surf. A. 346 (1–3), 34–38. https://doi.org/10.1016/j.colsurfa.2009.05.022 (2009).
Yang, W., Li, X., Pan, B., Lv, L. & Zhang, W. Effective removal of effluent organic matter (EfOM) from bio-treated coking wastewater by a recyclable aminated hyper-cross-linked polymer. Water Res. 47 (13), 4730–4738. https://doi.org/10.1016/j.watres.2013.05.032 (2013).
Xu, C. et al. Preparation of hypercrosslinked amine modification resin and its adsorption properties for nitroaromatics. Environ. Sci. Pollut. Res. 26 (11), 10767–10775. https://doi.org/10.1007/s11356-019-04370-4 (2019).
Santos, D. P. D., Andrade, G. F. S., Temperini, M. L. A. & Brolo, A. G. Electrochemical control of the time-dependent intensity fluctuations in surface-enhanced raman scattering (SERS). J. Phys. Chem. C. 113 (41), 17737–17744. https://doi.org/10.1021/jp907389v (2009).
Ramírez-Herrera, C. A., Cruz-Cruz, I., Jiménez-Cedeño, I. H., Martínez-Romero, O. & Elías-Zúñiga, A. Influence of the epoxy resin process parameters on the mechanical properties of produced bidirectional [± 45°] Carbon/Epoxy woven composites. Polymers 13 (8), 1273. https://doi.org/10.3390/polym13081273 (2021).
Sarno, M., Iuliano, M., De Pasquale, S. & Ponticorvo, E. A new nano-catalyst for sawdust hydrolysis. Appl. Catal. A. 602, 117686. https://doi.org/10.1016/j.apcata.2020.117686 (2020).
Wang, J., Yue, D. & Wang, H. In situ Fe3O4 nanoparticles coating of polymers for separating hazardous PVC from microplastic mixtures. Chem. Eng. J. 407, 127170. https://doi.org/10.1016/j.cej.2020.127170 (2020).
Peng, Q. et al. Synthesis of novel magnetic pitch-based hypercrosslinked polymers as adsorbents for effective recovery of Ag+ with high selectivity. J. Environ. Manag. 339, 117763. https://doi.org/10.1016/j.jenvman.2023.117763 (2023).
Modestino, M., Galluzzi, A., Sarno, M. & Polichetti, M. The effect of a DC magnetic field on the AC magnetic properties of oleic acid-coated Fe3O4 nanoparticles. Materials 16 (12), 4246. https://doi.org/10.3390/ma16124246 (2023).
Mørup, S., Frandsen, C. & Hansen, M. F. Magnetic properties of nanoparticles. Int. J. Mod. Phys. B, 1 (2010).
Modestino, M., Galluzzi, A., Sarno, M. & Polichetti, M. Study of the coercivity field in a sample of NiFe2O4–OA nanoparticles at different temperatures: Comparing the different processes to evaluate TB by DC measurements. In IEEE Nanotechnology Materials and Devices Conference, 259–263. https://doi.org/10.1109/nmdc57951.2023.10343818 (2023).
Modestino, M. et al. The effect of copper adsorption on iron oxide magnetic nanoparticles embedded in a sodium alginate bead. Nanomaterials 15 (15), 1196. https://doi.org/10.3390/nano15151196 (2025).
Bruvera, I. J., Zélis, P. M., Calatayud, M. P., Goya, G. F. & Sánchez, F. H. Determination of the blocking temperature of magnetic nanoparticles: The good, the bad, and the ugly. J. Appl. Phys. 118 (18). https://doi.org/10.1063/1.4935484 (2015).
Micha, J., Dieny, B., Régnard, J., Jacquot, J. & Sort, J. Estimation of the Co nanoparticles size by magnetic measurements in Co/SiO2 discontinuous multilayers. J. Magn. Magn. Mater. 272–276. https://doi.org/10.1016/j.jmmm.2003.12.268 (2004).
Iuliano, M., Ponticorvo, E., Cirillo, C., Adami, R. & Sarno, M. Catalytic hydrogenation of organic dyes by Ag and Au magnetic nanoparticles supported on nanocellulose from waste pistachio shells. Mol. Catal. 544, 113179. https://doi.org/10.1016/j.mcat.2023.113179 (2023).
Iuliano, M., Cirillo, C., Astorga, E. N. & Sarno, M. A new nanocomposite as adsorbent and catalyst for enhanced removal of methylene blue. Surf. Interfaces. 51, 104582. https://doi.org/10.1016/j.surfin.2024.104582 (2024).
Azim, E. A., Samy, M., Hanafy, M. & Mahanna, H. Novel mint-stalks derived biochar for the adsorption of methylene blue dye: Effect of operating parameters, adsorption mechanism, kinetics, isotherms, and thermodynamics. J. Environ. Manag. 357, 120738. https://doi.org/10.1016/j.jenvman.2024.120738 (2024).
Tran, T. H. et al. Comparative study on methylene blue adsorption behavior of coffee husk-derived activated carbon materials prepared using hydrothermal and soaking methods. J. Environ. Chem. Eng. 9 (4), 105362. https://doi.org/10.1016/j.jece.2021.105362 (2021).
Tran, T. H. et al. Adsorption isotherms and kinetic modeling of methylene blue dye onto a carbonaceous hydrochar adsorbent derived from coffee husk waste. Sci. Total Environ. 725, 138325. https://doi.org/10.1016/j.scitotenv.2020.138325 (2020).
Afroze, S., Sen, T. K., Ang, M. & Nishioka, H. Adsorption of methylene blue dye from aqueous solution by novel biomass Eucalyptus sheathiana bark: equilibrium, kinetics, thermodynamics and mechanism. Desalin. Water Treat. 57 (13), 5858–5878. https://doi.org/10.1080/19443994.2015.1004115 (2015).
Shu, J. et al. Adsorption of methylene blue on modified electrolytic manganese residue: Kinetics, isotherm, thermodynamics and mechanism analysis. J. Taiwan Inst. Chem. Eng. 82, 351–359. https://doi.org/10.1016/j.jtice.2017.11.020 (2017).
Nipa, S. T. et al. Adsorption of methylene blue on papaya bark fiber: Equilibrium, isotherm and kinetic perspectives. Results Eng. 17, 100857. https://doi.org/10.1016/j.rineng.2022.100857 (2022).
El-Bendary, N., El-Etriby, H. K. & Mahanna, H. High-performance removal of iron from aqueous solution using modified activated carbon prepared from corn cobs and luffa sponge. Desalin. Water Treat. 213, 348–357. https://doi.org/10.5004/dwt.2021.26721 (2021).
Duran, C., Özdeş, D., Gündoğdu, A. & Şentürk, H. B. Kinetics and isotherm analysis of basic dyes adsorption onto almond shell (Prunus dulcis) as a low cost adsorbent. J. Chem. Eng. Data/J. Chem. Eng. Data. 56 (5), 2136–2147. https://doi.org/10.1021/je101204j (2011).
Fu, J. et al. Adsorption of methylene blue by a high-efficiency adsorbent (polydopamine microspheres): Kinetics, isotherm, thermodynamics and mechanism analysis. Chem. Eng. J. 259, 53–61. https://doi.org/10.1016/j.cej.2014.07.101 (2014).
Farch, S. et al. Application of walnut shell biowaste as an inexpensive adsorbent for methylene blue dye: Isotherms, kinetics, thermodynamics, and modeling. Separations 10 (1), 60. https://doi.org/10.3390/separations10010060 (2023).
Yuan, W. et al. Synergistic dual-functionalities of starch-grafted-styrene hydrophilic porous resin for efficiently removing bisphenols from wastewater. Chem. Eng. J. 429, 132350. https://doi.org/10.1016/j.cej.2021.132350 (2021).
Alver, E. & Metin, A. Ü. Anionic dye removal from aqueous solutions using modified zeolite: Adsorption kinetics and isotherm studies. Chem. Eng. J. 200–202, 59–67. https://doi.org/10.1016/j.cej.2012.06.038 (2012).
Melo, B. C. et al. Cellulose nanowhiskers improve the methylene blue adsorption capacity of chitosan-g-poly(acrylic acid) hydrogel. Carbohydr. Polym. 181, 358–367. https://doi.org/10.1016/j.carbpol.2017.10.079 (2017).
Long, Z. et al. High-performance adsorption of methylene blue using novel bio-adsorbent based on sargassum fusiforme. Heliyon 10 (18), e37949. https://doi.org/10.1016/j.heliyon.2024.e37949 (2024).
Huang, D. et al. Indirect determination of sulfide ions in water samples at trace level by anodic stripping voltammetry using mercury film electrode. Anal. Methods. 2 (2), 154–158. https://doi.org/10.1039/b9ay00183b (2009).
Nasar, A. & Mashkoor, F. Application of polyaniline-based adsorbents for dye removal from water and wastewater—a review. Environ. Sci. Pollut. Res. 26 (6), 5333–5356. https://doi.org/10.1007/s11356-018-3990-y (2019).
Xing, Y. et al. Thermally and chemically stable Fe/Mg-layered double oxides-biochar for enhanced polystyrene nanoplastic adsorption and sustainable recycling. Chem. Eng. J. 508, 160918. https://doi.org/10.1016/j.cej.2025.160918 (2025).
Wang, P. et al. Kinetics and thermodynamics of adsorption of methylene blue by a magnetic graphene-carbon nanotube composite. Appl. Surf. Sci. 290, 116e124 (2014).
Allou, N. B. et al. Methylene blue magnetic adsorption separation process from aqueous solution using corn cob. Sci. Afr. 21, e01828. https://doi.org/10.1016/j.sciaf.2023.e01828 (2023b).
Gong, J. et al. Removal of cationic dyes from aqueous solution using magnetic multi-wall carbon nanotube nanocomposite as adsorbent. J. Hazard. Mater. 164 (2–3), 1517–1522. https://doi.org/10.1016/j.jhazmat.2008.09.072 (2008).
Ge, Y. et al. Enhanced adsorption and catalytic degradation of organic dyes by nanometer iron oxide anchored to single-wall carbon nanotubes. Appl. Surf. Sci. 488, 813–826. https://doi.org/10.1016/j.apsusc.2019.05.221 (2019).
Ahamad, T. et al. Effective and fast adsorptive removal of toxic cationic dye (MB) from aqueous medium using amino-functionalized magnetic multiwall carbon nanotubes. J. Mol. Liq. 282, 154–161. https://doi.org/10.1016/j.molliq.2019.02.128 (2019).
Foroutan, R., Mohammadi, R., Razeghi, J. & Ramavandi, B. Performance of algal activated carbon/Fe3O4 magnetic composite for cationic dyes removal from aqueous solutions. Algal Res. 40, 101509. https://doi.org/10.1016/j.algal.2019.101509 (2019).
Acknowledgements
Rachele Castaldo acknowledges financial support under the Italian Science Fund (FIS 2 Call) funded by the Italian Ministry of University and Research (MUR) for the project HIerarchically Porous materials with tailored ADsorption properties to face environmental pollution (HIPAD, Proposal code FIS-2023-01686, CUP B53C24009520001, Grant Assignment Decree No. n. 23314 adopted on 11/12/2024 by the Italian MUR).
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
C.C.: Conceptualization; data curation; formal analysis; investigation; methodology; validation; visualization; writing-original draft; writing-review and editing; supervision. M.I., Conceptualization; data curation; formal analysis; investigation; methodology; validation; visualization; writing-original draft; writing-review and editing. M.M.; A.G.: Data curation; conceptualization; investigation; methodology. investigation, writing-original draft. F.O.; R.C: Investigation; writing-original draft; visualization. G.G.; M.P.; M.S.: Supervision; validation; writing-original draft; writing-review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cirillo, C., Iuliano, M., Modestino, M. et al. Magnetic amino-functionalized hyper-crosslinked resin as a reusable adsorbent for methylene blue. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45545-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-45545-7