Abstract
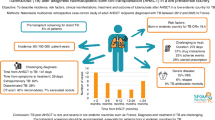
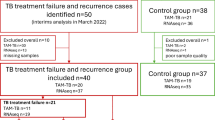
There is an urgent need to rapidly diagnose tuberculosis (TB) disease and effectively monitor anti-TB treatment responses. Host-directed therapy (HDT) is a promising platform to mitigate challenges in TB diagnosis and anti-TB treatment response monitoring. Identifying changes in systemic proteins and immune cell distributions during the disease is an integral aspect of developing targeted therapies. Here, samples were collected from healthy individuals (CTRL) [n = 32 plasma, n = 9 bronchoalveolar lavage (BAL)] and newly diagnosed TB patients (TB treatment group) [n = 82 plasma, n = 28 BAL] to analyze full blood count, secreted levels of full-length osteopontin (OPN), and inflammatory markers. Peripheral blood and BAL samples were collected at a single time-point from CTRL, while in TB participants, they were collected at TB diagnosis (TBDx), week 1 (TBW1), month 2 (TBM2), and month 6 (TBM6). We observed a significantly increased monocyte-to-lymphocyte ratio (MLR) and plasma OPN in TB group at TBDx compared to the CTRL group. Inflammatory markers including IL-6, VEGF-A, and sFasL showed significant increase at TBDx when compared to CTRL, but these significantly declined by TBM6. Plasma OPN significantly declined at TBW1 and TBM2 when compared to TBDx but significantly increased at TBM6. BAL OPN showed no significant differences between CTRL and TB patients at TBDx, whereas a significant increase was observed in TB group between TBDx and TBM6. Given the study limitations, these findings should be considered preliminary and exploratory. Our results add to literature and identify MLR and plasma OPN as potential targets for early TB diagnosis and treatment monitoring.
Similar content being viewed by others
Data availability
Data are contained within the article.
References
Global tuberculosis report 2025. Geneva: World Health Organization [Internet]. Geneva. (2025). Available from: https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2025
Dheda, K. et al. The epidemiology, pathogenesis, transmission, diagnosis, and management of multidrug-resistant, extensively drug-resistant, and incurable tuberculosis. Lancet Respir Med. 5 (4), 291–360 (2017).
Geneva & World Health Organization. ; Practical manual on tuberculosis laboratory strengthening, 2022 update [Internet]. (2022). Available from: https://iris.who.int/server/api/core/bitstreams/6ed85122-48a2-437e-a47c-e4a0709728d9/content
Parrish, N. M. & Carroll, K. C. Role of the Clinical Mycobacteriology Laboratory in Diagnosis and Management of Tuberculosis in Low-Prevalence Settings. J. Clin. Microbiol. 49 (3), 772–776 (2011).
Lawn, S. D. et al. Advances in tuberculosis diagnostics: The Xpert MTB/RIF assay and future prospects for a point-of-care test. Lancet Infect. Dis. 13(4), 349–61 (2013).
Naranbhai, V. et al. Ratio of Monocytes to Lymphocytes in Peripheral Blood Identifies Adults at Risk of Incident Tuberculosis Among HIV-Infected Adults Initiating Antiretroviral Therapy. J. Infect. Dis. 209 (4), 500–509 (2014).
La Manna, M. P. et al. Quantitative and qualitative profiles of circulating monocytes may help identifying tuberculosis infection and disease stages. Wilkinson KA, editor. PLOS ONE. 12(2): e0171358. (2017).
Estévez, O. et al. Multi-parameter flow cytometry immunophenotyping distinguishes different stages of tuberculosis infection. J. Infect. 81(1), 57–71 (2020).
Grassi, G. et al. PMN-MDSC frequency discriminates active versus latent tuberculosis and could play a role in counteracting the immune-mediated lung damage in active disease. Front. Immunol. 12, 594376 (2021).
Denhardt, D. T., Noda, M., O’Regan, A. W., Pavlin, D. & Berman, J. S. Osteopontin as a means to cope with environmental insults: Regulation of inflammation, tissue remodeling, and cell survival. J. Clin. Invest. 107(9), 7 (2001).
Nyström, T., Dunér, P. & Hultgårdh-Nilsson, A. A constitutive endogenous osteopontin production is important for macrophage function and differentiation. Exp. Cell Res. 313(6), 1149–1160 (2007).
Koh, A. et al. Role of osteopontin in neutrophil function. Immunology 122 (4), 466–475 (2007).
Nishimichi, N. et al. Osteopontin Undergoes Polymerization in Vivo and Gains Chemotactic Activity for Neutrophils Mediated by Integrin α9β1. J. Biol. Chem. 286 (13), 11170–11178 (2011).
Xiang, J. et al. Preoperative monocyte-to-lymphocyte ratio in peripheral blood predicts stages, metastasis, and histological grades in patients with ovarian cancer. Transl. Oncol. 10(1), 33–9 (2017).
Huang, Y. et al. Relationship between monocytes to lymphocytes ratio and axial spondyloarthritis. Int. Immunopharmacol. 57, 43–6 (2018).
Domingo-Gonzalez, R., Prince, O., Cooper, A. & Khader, S. A. Cytokines and Chemokines in Mycobacterium tuberculosis Infection. Microbiol. Spectr. 4(5), TBTB2-0018-2016. https://doi.org/10.1128/microbiolspec.TBTB2-0018-2016 (2016).
Giachelli, C. M., Lombardi, D., Johnson, R. J. & Almeida, M. Evidence for a Role of Osteopontin in Macrophage Infiltration in Response to Pathological Stimuli in vivo. Am. J. Pathol. 152(2), 353–358 (1998).
Weber, G. F. et al. Phosphorylation-dependent interaction of osteopontin with its receptors regulates macrophage migration and activation. J. Leukoc. Biol. 72(4), 752–761. https://doi.org/10.1189/jlb.72.4.752 (2002).
Crawford, H. C., Matrisian, L. M. & Liaw, L. Distinct Roles of Osteopontin in Host Defense Activity and Tumor Survival during Squamous Cell Carcinoma Progression in vivo. Cancer Res. 58(22), 5206–5215 (1998).
Scatena, M., Liaw, L. & Giachelli, C. M. Osteopontin: A multifunctional molecule regulating chronic inflammation and vascular disease. Arterioscler. Thromb. Vasc. Biol. 27(11), 2302–2309 (2007).
Inoue, M. & Shinohara, M. L. Intracellular osteopontin (iOPN) and immunity. Immunol. Res. 49(1–3), 160–72 (2011).
Masuda, K., Takahashi, N., Tsukamoto, Y., Honma, H. & Kohri, K. N-glycan structures of an osteopontin from human bone. Biochem. Biophys. Res. Commun. 268(3), 814–817 (2000).
Cho, E. H., Cho, K. H., Lee, H. A. & Kim, S. W. High serum osteopontin levels are associated with low bone mineral density in postmenopausal women. J. Korean Med. Sci. 28(10), 1496 (2013).
Koguchi, Y. et al. High plasma osteopontin level and its relationship with interleukin-12-mediated type 1 T helper cell response in tuberculosis. Am. J. Respir Crit. Care Med. 167 (10), 1355–1359 (2003).
Nau, G. J. et al. A chemoattractant cytokine associated with granulomas in tuberculosis and silicosis. Proc. Natl. Acad. Sci. 94 (12), 6414–6419 (1997).
Liaw, L. et al. Altered Wound Healing in Mice Lacking a Functional Osteopontin Gene (spp1). J Clin Invest https://doi.org/10.1172/JCI1122 (1998).
Ragno, S. et al. Changes in gene expression in macrophages infected with Mycobacterium tuberculosis: A combined transcriptomic and proteomic approach. Immunology 104(1), 99–108 (2001).
Kanayama, M. et al. Skewing of the population balance of lymphoid and myeloid cells by secreted and intracellular osteopontin. Nat Immunol 18(9), 973–84 (2017).
Denhardt, D. T. & Guo, X. Osteopontin: A protein with diverse functions. FASEB J. 7(15), 1475–82 (1993).
O’Regan, A. W., Hayden, J. M. & Berman, J. S. Osteopontin augments CD3-mediated interferon-gamma and CD40 ligand expression by T cells, which results in IL-12 production from peripheral blood mononuclear cells. J Leukoc Biol https://doi.org/10.1189/jlb.68.4.495 (2000).
Patarca, R. et al. Structural and functional studies of the early T lymphocyte activation 1 (Eta-1) gene. Definition of a novel T cell-dependent response associated with genetic resistance to bacterial infection. J. Exp. Med. 170 (1), 145–161 (1989).
Uede, T. Osteopontin, intrinsic tissue regulator of intractable inflammatory diseases: Osteopontin, cytokine or matrix protein? Pathol. Int. 61 (5), 265–280 (2011).
Shete, A. et al. Elevated Levels of Galectin-9 but Not Osteopontin in HIV and Tuberculosis Infections Indicate Their Roles in Detecting MTB Infection in HIV Infected Individuals. Front. Microbiol. 11, 1685 (2020).
Shiratori, B. et al. Elevated OPN, IP-10, and Neutrophilia in Loop-Mediated Isothermal Amplification Confirmed Tuberculosis Patients. Mediators Inflamm. 2014, 1–8 (2014).
Zhu, Y. et al. Decreased Osteopontin Expression as a Reliable Prognostic Indicator of Improvement in Pulmonary Tuberculosis: Impact of the Level of Interferon-γ-Inducible Protein 10. Cell. Physiol. Biochem. 37 (5), 1983–1996 (2015).
Hasibuan, F. M. et al. Evaluation of matricellular proteins in systemic and local immune response to Mycobacterium tuberculosis infection: Matricellular proteins in tuberculosis. Microbiol. Immunol. 59 (10), 623–632 (2015).
Shiratori, B. et al. Immunological Roles of Elevated Plasma Levels of Matricellular Proteins in Japanese Patients with Pulmonary Tuberculosis. Int. J. Mol. Sci. 18 (1), 19 (2016).
Wang, D. et al. The association between osteopontin and tuberculosis: A systematic review and meta-analysis. Rottenberg ME, editor. PLOS ONE. 15(12):e0242702. (2020).
Heilmann, K. et al. Osteopontin as two-sided mediator of intestinal inflammation. J. Cell. Mol. Med. 13 (6), 1162–1174 (2009).
Kourepini, E. et al. Osteopontin expression by CD103 – dendritic cells drives intestinal inflammation. Proc. Natl. Acad. Sci. 111 (9), E856–E865 (2014).
Toyonaga, T. et al. Osteopontin Deficiency Accelerates Spontaneous Colitis in Mice with Disrupted Gut Microbiota and Macrophage Phagocytic Activity. Bamias G, editor. PLOS ONE. 10(8):e0135552. (2015).
Gupte, A. N. et al. Baseline IL-6 is a biomarker for unfavourable tuberculosis treatment outcomes: A multisite discovery and validation study. Eur. Respir. J. 59(4), 2100905 (2022).
Djoba Siawaya, J. F., Beyers, N., van Helden, P. & Walzl, G. Differential cytokine secretion and early treatment response in patients with pulmonary tuberculosis. Clin. Exp. Immunol. 156 (1), 69–77 (2009).
Gao, K. et al. Diagnostic value of the blood monocyte–lymphocyte ratio in knee osteoarthritis. J. Int. Med. Res. 47(9), 4413–21 (2019).
Wang, J. et al. Ratio of monocytes to lymphocytes in peripheral blood in patients diagnosed with active tuberculosis. Braz. J. Infect. Dis. 19(2), 125–31 (2015).
Naranbhai, V. et al. Distinct transcriptional and anti-mycobacterial profiles of peripheral blood monocytes dependent on the ratio of monocytes: Lymphocytes. EBioMedicine 2(11), 1619–26 (2015).
Zhou, Q., Shao, X., Xu, L., Zou, H. & Chen, W. Association between monocyte-to-lymphocyte ratio and inflammation in chronic kidney disease: A cross-sectional study. Kidney Blood Press. Res. 49(1), 1066–1074 (2024).
Li, X. et al. Association between monocyte-to-lymphocyte ratio and cardiovascular diseases: Insights from NHANES data. Diabetol. Metab. Syndr. 17(1), 98 (2025).
Kösehasanoğulları, M., Bilecik, N. A., Şen, S. B. & Koçyiğit, B. F. Elevated Monocyte-to-Lymphocyte and Platelet-to-Lymphocyte Ratios Are Associated with Disease Activity and Pain in Fibromyalgia: A Cross-Sectional Study. J. Clin. Med. 15 (1), 155 (2025).
Bot, F. J., van Eijk, L., Broeders, L., Aarden, L. A. & Löwenberg, B. Interleukin-6 synergizes with M-CSF in the formation of macrophage colonies from purified human marrow progenitor cells. Blood 73(2), 435–7 (1989).
Maeda, K. et al. Interleukin-6 aborts lymphopoiesis and elevates production of myeloid cells in systemic lupus erythematosus-prone B6.Sle1.Yaa animals. Blood 113 (19), 4534–4540 (2009).
Kethireddy, S. et al. Mycobacterium tuberculosis septic shock. Chest 144(2), 474–82 (2013).
Shamaei, M. et al. Tuberculosis-Associated Secondary Pneumothorax: A Retrospective Study of 53 Patients. Respir Care. 56 (3), 298–302 (2011).
Jordan, T. S., Spencer, E. M. & Davies, P. Tuberculosis, bronchiectasis and chronic airflow obstruction: Tuberculosis, Bronchiectasis and CAO. Respirology 15 (4), 623–628 (2010).
Kuo, S. C. et al. Association between tuberculosis infections and non-pulmonary malignancies: A nationwide population-based study. Br. J. Cancer 109(1), 229–34 (2013).
Chen, Y. Y. et al. Increased risk of incident osteoporosis and osteoporotic fracture in tuberculosis patients: A population-based study in a tuberculosis-endemic area. Osteoporos. Int. 28(5), 1711–21 (2017).
Azzurri, A. et al. IFN-γ-inducible protein 10 and pentraxin 3 plasma levels are tools for monitoring inflammation and disease activity in Mycobacterium tuberculosis infection. Microbes Infect. 7(1), 1–8 (2005).
Su, W. L. et al. Association of reduced tumor necrosis factor alpha, gamma interferon, and interleukin-1β (IL-1β) but increased IL-10 expression with improved chest radiography in patients with pulmonary tuberculosis. Clin. Vaccine Immunol. 17(2), 223–231 (2010).
Ronacher, K. et al. Distinct serum biosignatures are associated with different tuberculosis treatment outcomes. Tuberculosis 118, 101859 (2019).
Hong, J. Y. et al. Efficacy of IP-10 as a biomarker for monitoring tuberculosis treatment. J. Infect. 68(3), 252–8 (2014).
Kumar, N. P. et al. Plasma chemokines are baseline predictors of unfavorable treatment outcomes in pulmonary tuberculosis. Clin. Infect. Dis. ;ciaa1104. (2020).
de Oliveira, L. R. C. et al. da, Analysis of Toll-Like Receptors, iNOS and Cytokine Profiles in Patients with Pulmonary Tuberculosis during Anti-Tuberculosis Treatment. Torrelles JB, editor. PLoS ONE. 9(2): e88572. (2014).
Jacobs, A. J., Mongkolsapaya, J., Screaton, G. R., McShane, H. & Wilkinson, R. J. Antibodies and tuberculosis. Tuberculosis 101, 102–13 (2016).
Acknowledgements
The authors thank the participants and members of the Immunology Research Group for providing the samples.
Funding
AGL and BM is funded by the Centre for Tuberculosis Research (CTR) of the South African Medical Research Council (SAMRC). AGL is supported by the National Research Foundation (NRF) [CSUR Grant number 60502163639] and by the Trails of Excellence in Southern Africa (TESA) III of the European Development and Clinical Trails Partnership (EDCTP) [Grant number CSA2020NoE-3104].
Author information
Authors and Affiliations
Contributions
Conceptualization, A.G.L.; methodology, B.M., A.H., C.I.S., and I.R.; formal analysis, K.S.; writing—original draft preparation, B.M. and A.H.; writing—review and editing, K.S., I.R., C.I.S; supervision, A.G.L.; funding acquisition, A.G.L. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Motaung, B., Holtzhausen, AR., Stanley, K. et al. The value of the monocyte-to-lymphocyte ratio and osteopontin (SPP1) in tuberculosis treatment response monitoring. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46176-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46176-8