Abstract
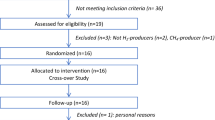
This study aimed to explore the effects of gut microbiota metabolic disorders caused by gut microbiota dysbiosis on heart failure due to congenital heart disease (CHD) through metabolomic analysis. Patients with congestive heart failure caused by left-to-right shunt CHD were selected as the subjects. Thirty infants with heart failure due to CHD admitted to the Department of Cardiovascular Surgery of our hospital from April 2022 to August 2022 were included in this study. Thirty healthy infants of the same age and sex who visited our hospital during the same period were selected as the control group. Faecal samples were collected from each participant and subjected to metabolomic analysis. Compared with those in the control group, the levels of 125 metabolites increased, whereas those of 147 metabolites decreased in the heart failure group. Compared with those in the control group, the levels of indoxyl, arachidonic acid, erucic acid, and DL-glycerol 1-phosphate were significantly increased in the heart failure group, whereas the level of 1-aminocyclopropanecarboxylic acid was significantly decreased. Pathway analysis of differentially abundant metabolites revealed that, compared with those in the control group, the metabolic pathways of linoleic acid metabolism, PPAR signalling, and arachidonic acid metabolism were significantly upregulated in the heart failure group. The NT-BNP level was significantly positively correlated with indoxyl, arachidonic acid and erucic acid (P < 0.05). There was a significant positive correlation between cardiac function scores and the levels of indoxyl and arachidonic acid (P < 0.05). In this exploratory study, infants with congestive heart failure due to CHD exhibited significant changes in gut microbiota metabolites and metabolic pathways. The gut metabolites of indoxyl and arachidonic acid significantly were increased, and the metabolic pathways of linoleic acid metabolism, the PPAR signalling pathway, and arachidonic acid metabolism were significantly upregulated in the heart failure infants. Increased gut metabolites of indoxyl and arachidonic acid were positively correlated with the severity of heart failure.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Abbreviations
- CHD:
-
Congenital heart disease
- LC:
-
Liquid chromatography
- MS:
-
Mass spectrometry
- QC:
-
Quality control
- PCA:
-
Principal component analysis
- PLS-DA:
-
Partial least square discriminant analysis
- VIP:
-
Variable importance on projection
- FC:
-
Fold change
References
Yancy, C. W. et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines and the heart failure Society of America. J. Am. Coll. Cardiol. 70(6), 776–803 (2017).
Metra, M. & Lucioli, P. Corrigendum to “Prevalence of heart failure and left ventricular dysfunction in China: The China Hypertension Survey, 2012–2015”. Eur. J. Heart Fail. 21, 1329–1337 (2019).
Mudd, J. O. & Kass, D. A. Tackling heart failure in the twenty-first century. Nature 451(7181), 919–928 (2008).
Chioncel, O. et al. Clinical phenotypes and outcome of patients hospitalized for acute heart failure: The ESC Heart Failure Long-Term Registry. Eur J Heart Fail. 19(10), 1242–1254 (2017).
Tang, W. H. W., Bäckhed, F., Landmesser, U. & Hazen, S. L. Intestinal microbiota in cardiovascular health and disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 73(16), 2089–2105 (2019).
Tang, W. H., Kitai, T. & Hazen, S. L. Gut microbiota in cardiovascular health and disease. Circ Res. 120(7), 1183–1196 (2017).
Nagatomo, Y. & Tang, W. H. Intersections between microbiome and heart failure: Revisiting the gut hypothesis. J. Card. Fail. 21(12), 973–980 (2015).
Luqman, A. et al. Role of the intestinal microbiome and its therapeutic intervention in cardiovascular disorder. Front. Immunol. 26(15), 1321395 (2024).
Branchereau, M., Burcelin, R. & Heymes, C. The gut microbiome and heart failure: A better gut for a better heart. Rev. Endocr. Metab. Disord. 20(4), 407–414 (2019).
Lupu, V. V. et al. The implication of the gut microbiome in heart failure. Cells 12(8), 1158 (2023).
Pasini, E. et al. Pathogenic gut flora in patients with chronic heart failure. JACC Heart Fail. 4(3), 220–227 (2016).
Luedde, M. et al. Heart failure is associated with depletion of core intestinal microbiota. ESC Heart Fail. 4(3), 282–290 (2017).
Jain, H. et al. Unveiling the relationship between gut microbiota and heart failure: Recent understandings and insights. Curr. Probl. Cardiol. 49(1), 102179 (2024).
Chioncel, O. & Ambrosy, A. P. Trimethylamine N-oxide and risk of heart failure progression: Marker or mediator of disease. Eur. J. Heart Fail. 21(7), 887–890 (2019).
Zhao, P., Zhao, S., Tian, J. & Liu, X. Significance of gut microbiota and short-chain fatty acids in heart failure. Nutrients 14(18), 3758 (2022).
Guan, X. & Sun, Z. The role of intestinal flora and its metabolites in heart failure. Infect Drug Resist. 5(16), 51–64 (2023).
Shi, M., Wei, J., Yuan, H., Li, Y. & Guo, Z. The role of the gut microbiota and bile acids in heart failure: A review. Medicine (Baltimore) 102(45), e35795 (2023).
Yukino-Iwashita, M. et al. Short-chain fatty acids in gut-heart axis: Their role in the pathology of heart failure. J Pers Med. 12(11), 1805 (2022).
Masenga, S. K., Povia, J. P., Lwiindi, P. C. & Kirabo, A. Recent advances in microbiota-associated metabolites in heart failure. Biomedicines 11(8), 2313 (2023).
Zhang, Q. L. et al. Relationship between disorders of the intestinal microbiota and heart failure in infants with congenital heart disease. Front. Cell. Infect. Microbiol. 13, 1152349 (2023).
Huang, Y. et al. Mapping the early life gut microbiome in neonates with critical congenital heart disease: Multiomics insights and implications for host metabolic and immunological health. Microbiome. 10(1), 245 (2022).
Zheng, X., Wang, R. & Yin, C. An untargeted metabolomics investigation in liver of flaviviruses-infected mice. Virology 582, 12–22 (2023).
Wu, D. et al. Female zebrafish are more affected than males under polystyrene microplastics exposure. J. Hazard Mater. 15(482), 136616 (2025).
Yu, H. et al. Improving red pitaya fruit quality by nano-selenium biofortification to enhance phenylpropanoid and betalain biosynthesis. Ecotoxicol. Environ. Saf. 15(267), 115653 (2023).
Ogata, H. et al. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 27(1), 29–34 (1999).
Läer, S. et al. Carvedilol therapy in pediatric patients with congestive heart failure: A study investigating clinical and pharmacokinetic parameters. Am. Heart J. 143(5), 916–922 (2002).
Hayashi, T. et al. Uncovering the role of gut microbiota in amino acid metabolic disturbances in heart failure through metagenomic analysis. Front. Cardiovasc. Med. 29(8), 789325 (2021).
Carrillo-Salinas, F. J. et al. Gut dysbiosis induced by cardiac pressure overload enhances adverse cardiac remodeling in a T cell-dependent manner. Gut Microbes. 12(1), 1–20 (2020).
Kelly, C. J. et al. Crosstalk between microbiota-derived short-chain fatty acids and intestinal epithelial HIF augments tissue barrier function. Cell Host Microbe. 17(5), 662–671 (2015).
Vijay, A. et al. Dietary interventions reduce traditional and novel cardiovascular risk markers by altering the gut microbiome and their metabolites. Front. Cardiovasc. Med. 14(8), 691564 (2021).
Tang, T. W. H. et al. Loss of gut microbiota alters immune system composition and cripples postinfarction cardiac repair. Circulation 139(5), 647–659 (2019).
Askin, L., Polat, E. & Tanriverdi, O. Propionic acidemia, cardiomyopathies, and arrhythmias. J. Clin. Pract. Res. 46(1), 11–18 (2024).
Chen, X., Li, H. Y., Hu, X. M., Zhang, Y. & Zhang, S. Y. Current understanding of gut microbiota alterations and related therapeutic intervention strategies in heart failure. Chin. Med. J. (Engl). 132(15), 1843–1855 (2019).
Yu, W., Jiang, Y., Xu, H. & Zhou, Y. The Interaction of Gut Microbiota and Heart Failure with Preserved Ejection Fraction: From Mechanism to Potential Therapies. Biomedicines. 11(2), 442 (2023).
Savji, N. et al. The association of obesity and cardiometabolic traits with incident HFpEF and HFrEF. JACC Heart Fail. 6(8), 701–709 (2018).
Xu, J. & Yang, Y. Gut microbiome and its meta-omics perspectives: profound implications for cardiovascular diseases. Gut Microbes 13(1), 1936379 (2021).
Rose, D. P. Aspects of tryptophan metabolism in health and disease: A review. J. Clin. Pathol. 25(1), 17–25 (1972).
Niwa, T. Role of indoxyl sulfate in the progression of chronic kidney disease and cardiovascular disease: Experimental and clinical effects of oral sorbent AST-120. Ther. Apher Dial. 15(2), 120–124 (2011).
Lee, J. H. & Lee, J. Indole as an intercellular signal in microbial communities. FEMS Microbiol. Rev. 34(4), 426–444 (2010).
Shimizu, H., Saito, S., Higashiyama, Y., Nishijima, F. & Niwa, T. CREB, NF-κB, and NADPH oxidase coordinately upregulate indoxyl sulfate-induced angiotensinogen expression in proximal tubular cells. Am. J. Physiol. Cell Physiol. 304(7), C685–C692 (2013).
Dou, L. et al. The uremic solute indoxyl sulfate induces oxidative stress in endothelial cells. J. Thromb Haemost. 5(6), 1302–1308 (2007).
Ma, K. et al. Therapeutic and prognostic significance of arachidonic acid in heart failure. Circ Res. 130(7), 1056–1071 (2022).
Aspromonte, N. et al. Modulation of cardiac cytochrome P450 in patients with heart failure. Expert Opin. Drug. Metab. Toxicol. 10(3), 327–339 (2014).
Li, C. et al. Qishen granules inhibit myocardial inflammation injury through regulating arachidonic acid metabolism. Sci. Rep. 11(6), 36949 (2016).
Sonnweber, T., Pizzini, A., Nairz, M., Weiss, G. & Tancevski, I. Arachidonic acid metabolites in cardiovascular and metabolic diseases. Int. J. Mol. Sci. 19(11), 3285 (2018).
Maekawa, S. et al. Linoleic acid improves assembly of the CII subunit and CIII2/CIV complex of the mitochondrial oxidative phosphorylation system in heart failure. Cell Commun. Signal. 17(1), 128 (2019).
Wang, S. et al. Shengmai Yin formula exerts cardioprotective effects on rats with chronic heart failure via regulating linoleic acid metabolism. Prostaglandins Other Lipid Mediat. 158, 106608 (2022).
Chicco, A. J. et al. Linoleate-rich high-fat diet decreases mortality in hypertensive heart failure rats compared with lard and low-fat diets. Hypertension 52(3), 549–555 (2008).
Cheema, S. K., Tappia, P. S. & Dhalla, N. S. Modification of gene expression in rat cardiomyocytes by linoleic and docosahexaenoic acids 1. Can. J. Physiol. Pharmacol. 97(4), 320–327 (2019).
Xu, H. et al. Aged microbiota exacerbates cardiac failure by PPARα/PGC1α pathway. Biochimica et Biophys. Acta (BBA) 1870(7), 167271 (2024).
Guo, Z. et al. Cardiomyopeptide- regulated PPARγ expression plays a critical role in maintaining mitochondrial integrity and preventing cardiac ischemia/reperfusion injury. Int. J. Med. Sci. 22(1), 44–52 (2025).
De Bernardo, G. et al. Analysis of the fecal metabolomic profile in breast vs. different formula milk feeding in late preterm infants. Metabolites 14(1), 72 (2024).
Wang, M., Valizadegan, N., Fields, C. J. & Donovan, S. M. Fecal microbiome and metabolomic profiles of mixed-fed infants are more similar to formula-fed than breastfed infants. Microorganisms 13(1), 166 (2025).
Sillner, N. et al. Longitudinal profiles of dietary and microbial metabolites in formula- and breastfed infants. Front. Mol. Biosci. 8, 660456 (2021).
Acknowledgements
Not applicable.
Funding
This work was sponsored by Joint Funds for the innovation of science and Technology, Fujian province (Grant number: 2021Y9186). This work was sponsored by Fujian Provincial Health Commission [grant number ETK2023013]. This work was funded by the Startup Fund for scientific research, Fujian Medical University [grant number 2021QH1190].
Author information
Authors and Affiliations
Contributions
ZQL, WZC, OQX and WY designed the experiments, performed the statistical analysis, and drafted the manuscript. LYN and ZYT collected the data. CQ supervised the study. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the ethics committee of Fujian Children’s Hospital (2022ETKLR10079) and strictly adhered to the tenets of the Declaration of Helsinki. The parents or guardians of the patients gave written informed consent for their respective minors to participate in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Clinical trial number
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, QL., Ou, QX., Wang, Y. et al. Metabolomic analysis reveals the role of gut microbiota metabolic disorders in heart failure due to congenital heart disease. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46524-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46524-8