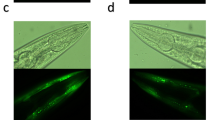
Abstract
Parkinson’s disease (PD) is a rapidly escalating neurodegenerative disorder marked by dopaminergic neurodegeneration, α-synuclein aggregation, and motor and non-motor impairments. Current therapies largely provide symptomatic relief and fail to prevent disease progression, underscoring the need for novel disease-modifying strategies. The marine biome has emerged as an unexplored reservoir of bioactive metabolites with neuroprotective potential, yet their therapeutic relevance in PD remains incompletely explored. Here, we report that Stutzerimonas stutzeri, a marine bacterium isolated from the Gulf of Mannar, exerts robust neuroprotective effects in Caenorhabditis elegans PD models. Dietary administration of S. stutzeri rescued dopaminergic neuronal loss, mitigated α-synuclein expression, and improved motor and sensory phenotypes. Mechanistic analyses revealed suppression of ferroptosis, evidenced by restoration of iron homeostasis, attenuation of lipid peroxidation, and recovery of ftn-1 expression. Our findings establish S. stutzeri as a previously unrecognized marine-derived therapeutic prospect for PD intervention and highlight ferroptosis modulation as a tractable therapeutic axis in neurodegeneration.
Similar content being viewed by others
Data availability
The datasets generated for the current study are available in the NCBI Gene Expression Omnibus (GEO) repository under accession code GSE312231. The Reviewer Token is uzqbimekrvydlkt for accessing the GEO dataset.The processed data downloaded from the Galaxy platform after analysis using the LIMMA package is provided in **Supplementary Information 4** .
Abbreviations
- PD:
-
Parkinson’s disease
- DA:
-
Dopaminergic
- SNpc:
-
Substantia nigra pars compacta
- NGM:
-
Nematode growth medium
- L1:
-
First larval stage
- MDA:
-
Malondialdehyde
- DEGs:
-
Differentially expressed genes
References
Collaborators, G. B. D. N. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 18, 459–480. https://doi.org/10.1016/S1474-4422(18)30499-X (2019).
Dorsey, E. R., Sherer, T., Okun, M. S. & Bloem, B. R. The Emerging Evidence of the Parkinson Pandemic. J. Parkinsons Dis. 8, S3–S8. https://doi.org/10.3233/JPD-181474 (2018).
Shin, H. W., Hong, S. W. & Youn, Y. C. Clinical Aspects of the Differential Diagnosis of Parkinson’s Disease and Parkinsonism. J. Clin. Neurol. 18, 259–270. https://doi.org/10.3988/jcn.2022.18.3.259 (2022).
Asano, H. et al. Safety comparisons among monoamine oxidase inhibitors against Parkinson’s disease using FDA adverse event reporting system. Sci. Rep. 13, 19272. https://doi.org/10.1038/s41598-023-44142-2 (2023).
Pandey, S. & Srivanitchapoom, P. Levodopa-induced Dyskinesia: Clinical Features, Pathophysiology, and Medical Management. Ann. Indian Acad. Neurol. 20, 190–198. https://doi.org/10.4103/aian.AIAN_239_17 (2017).
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072. https://doi.org/10.1016/j.cell.2012.03.042 (2012).
Li, J. et al. Ferroptosis: past, present and future. Cell. Death Dis. 11, 88. https://doi.org/10.1038/s41419-020-2298-2 (2020).
Dexter, D. T. et al. Increased nigral iron content in postmortem parkinsonian brain. Lancet 2, 1219–1220. https://doi.org/10.1016/s0140-6736(87)91361-4 (1987).
Hirsch, E. C., Brandel, J. P., Galle, P., Javoy-Agid, F. & Agid, Y. Iron and aluminum increase in the substantia nigra of patients with Parkinson’s disease: an X-ray microanalysis. J. Neurochem. 56, 446–451. https://doi.org/10.1111/j.1471-4159.1991.tb08170.x (1991).
Bellinger, F. P. et al. Glutathione Peroxidase 4 is associated with Neuromelanin in Substantia Nigra and Dystrophic Axons in Putamen of Parkinson’s brain. Mol. Neurodegener. 6, 8. https://doi.org/10.1186/1750-1326-6-8 (2011).
Pearce, R. K., Owen, A., Daniel, S., Jenner, P. & Marsden, C. D. Alterations in the distribution of glutathione in the substantia nigra in Parkinson’s disease. J. Neural Transm (Vienna). 104, 661–677. https://doi.org/10.1007/BF01291884 (1997).
Vallerga, C. L. et al. Analysis of DNA methylation associates the cystine-glutamate antiporter SLC7A11 with risk of Parkinson’s disease. Nat. Commun. 11, 1238. https://doi.org/10.1038/s41467-020-15065-7 (2020).
Do Van, B. et al. Ferroptosis, a newly characterized form of cell death in Parkinson’s disease that is regulated by PKC. Neurobiol. Dis. 94, 169–178. https://doi.org/10.1016/j.nbd.2016.05.011 (2016).
Tian, Y. et al. FTH1 Inhibits Ferroptosis Through Ferritinophagy in the 6-OHDA Model of Parkinson’s Disease. Neurotherapeutics 17, 1796–1812. https://doi.org/10.1007/s13311-020-00929-z (2020).
Bai, L. et al. Thioredoxin-1 Rescues MPP(+)/MPTP-Induced Ferroptosis by Increasing Glutathione Peroxidase 4. Mol. Neurobiol. 58, 3187–3197. https://doi.org/10.1007/s12035-021-02320-1 (2021).
Zuo, Y. et al. Ferritinophagy-mediated ferroptosis involved in paraquat-induced neurotoxicity of dopaminergic neurons: implication for neurotoxicity in PD. Oxid. Med. Cell. Longev. 2021 (9961628). https://doi.org/10.1155/2021/9961628 (2021).
Pingale, T. & Gupta, G. L. Classic and evolving animal models in Parkinson’s disease. Pharmacol. Biochem. Behav. 199, 173060. https://doi.org/10.1016/j.pbb.2020.173060 (2020).
Cooper, J. F. & Van Raamsdonk, J. M. Modeling Parkinson’s Disease in C. elegans. J. Parkinsons Dis. 8, 17–32. https://doi.org/10.3233/JPD-171258 (2018).
Alves, C. et al. From Marine Origin to Therapeutics: The Antitumor Potential of Marine Algae-Derived Compounds. Front. Pharmacol. 9, 777. https://doi.org/10.3389/fphar.2018.00777 (2018).
El Gamal, A. A. Biological importance of marine algae. Saudi Pharm. J. 18, 1–25. https://doi.org/10.1016/j.jsps.2009.12.001 (2010).
Koppula, S. et al. Recent advances on the neuroprotective potential of antioxidants in experimental models of Parkinson’s disease. Int. J. Mol. Sci. 13, 10608–10629. https://doi.org/10.3390/ijms130810608 (2012).
Mena, M. A. et al. NP7 protects from cell death induced by oxidative stress in neuronal and glial midbrain cultures from parkin null mice. FEBS Lett. 583, 168–174. https://doi.org/10.1016/j.febslet.2008.11.051 (2009).
Lee, H. W., Choi, H., Nam, S. J., Fenical, W. & Kim, H. Potent Inhibition of Monoamine Oxidase B by a Piloquinone from Marine-Derived Streptomyces sp. CNQ-027. J. Microbiol. Biotechnol. 27, 785–790. https://doi.org/10.4014/jmb.1612.12025 (2017).
Silva, J. et al. Marine-derived components: can they be a potential therapeutic approach to Parkinson’s disease? Mar. Drugs 21 https://doi.org/10.3390/md21080451 (2023).
Lenchi, N., Ahmedi, W. & Kebbouche-Gana, S. Assessment of bioremediation potential of Stutzerimonas stutzeri NL3 and Enterobacter cloacae NL4 isolates from an Algerian oilfield. Environ. Technol. 46, 3168–3184. https://doi.org/10.1080/09593330.2025.2458796 (2025).
Li, L. et al. Stutzerimonas stutzeri culture enhances microbial community structure and tomato seedling growth in saline soil. J. Appl. Microbiol. 136 https://doi.org/10.1093/jambio/lxaf026 (2025).
Tripathi, V. C. et al. The discovery of antioxidants in marine microorganisms and their protective effects on the hepatic cells from chemical-induced oxidative stress. Free Radic Res. 54, 150–161. https://doi.org/10.1080/10715762.2020.1725499 (2020).
Assembler & Baser Sequence, D. N. A. v3. x (2012) Heracle BioSoft SRL Romania. (2012).
Wright, E. S., Yilmaz, L. S. & Noguera, D. R. DECIPHER, a search-based approach to chimera identification for 16S rRNA sequences. Appl. Environ. Microbiol. 78, 717–725 (2012).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94. https://doi.org/10.1093/genetics/77.1.71 (1974).
Manalo, R. V. M. & Medina, P. M. B. Caffeine reduces deficits in mechanosensation and locomotion induced by L-DOPA and protects dopaminergic neurons in a transgenic Caenorhabditis elegans model of Parkinson’s disease. Pharm. Biol. 58, 721–731. https://doi.org/10.1080/13880209.2020.1791192 (2020).
Zhang, Z. et al. Jujubae Fructus extract prolongs lifespan and improves stress tolerance in Caenorhabditis elegans dependent on DAF-16/SOD-3. Sci. Rep. 14, 13713. https://doi.org/10.1038/s41598-024-64045-0 (2024).
Promtang, S. et al. 2-Butoxytetrahydrofuran, Isolated from Holothuria scabra, Attenuates Aggregative and Oxidative Properties of alpha-Synuclein and Alleviates Its Toxicity in a Transgenic Caenorhabditis elegans Model of Parkinson’s Disease. ACS Chem. Neurosci. 15, 2182–2197. https://doi.org/10.1021/acschemneuro.4c00008 (2024).
Zhu, F. D. et al. Carpesii fructus extract exhibits neuroprotective effects in cellular and Caenorhabditis elegans models of Parkinson’s disease. CNS Neurosci. Ther. 30, e14515. https://doi.org/10.1111/cns.14515 (2024).
Verma, S., Jagtap, U., Goyala, A. & Mukhopadhyay, A. A novel gene-diet pair modulates C. elegans aging. PLoS Genet. 14, e1007608. https://doi.org/10.1371/journal.pgen.1007608 (2018).
Murayama, T. & Maruyama, I. N. Plate Assay to Determine Caenorhabditis elegans Response to Water Soluble and Volatile Chemicals. Bio Protoc. 8, e2740. https://doi.org/10.21769/BioProtoc.2740 (2018).
Sammi, S. R., Syeda, T., Conrow, K. D., Leung, M. C. K. & Cannon, J. R. Complementary biological and computational approaches identify distinct mechanisms of chlorpyrifos versus chlorpyrifos-oxon-induced dopaminergic neurotoxicity. Toxicol. Sci. 191, 163–178. https://doi.org/10.1093/toxsci/kfac114 (2023).
Yong, Y. Y. et al. Penthorum chinense Pursh inhibits ferroptosis in cellular and Caenorhabditis elegans models of Alzheimer’s disease. Phytomedicine 127, 155463. https://doi.org/10.1016/j.phymed.2024.155463 (2024).
Ko, B. & Van Raamsdonk, J. M. RNA sequencing of pooled samples effectively identifies differentially expressed genes. Biology (Basel) 12 https://doi.org/10.3390/biology12060812 (2023).
Takele Assefa, A., Vandesompele, J. & Thas, O. On the utility of RNA sample pooling to optimize cost and statistical power in RNA sequencing experiments. BMC Genom. 21, 312. https://doi.org/10.1186/s12864-020-6721-y (2020).
Cheng, C. et al. Multiplexed bulk and single-cell RNA-seq hybrid enables cost-efficient disease modeling with chimeric organoids. Nat. Commun. 15, 3946. https://doi.org/10.1038/s41467-024-48282-5 (2024).
Everett, L. J. et al. Gene expression networks in the Drosophila Genetic Reference Panel. Genome Res. 30, 485–496. https://doi.org/10.1101/gr.257592.119 (2020).
Galaxy, C. The Galaxy platform for accessible, reproducible, and collaborative data analyses: 2024 update. Nucleic Acids Res. 52, W83–W94. https://doi.org/10.1093/nar/gkae410 (2024).
Dennis, G. Jr. et al. Database for Annotation, Visualization, and Integrated Discovery. Genome Biol. 4, P3 (2003).
Kolberg, L., Raudvere, U., Kuzmin, I., Vilo, J. & Peterson, H. gprofiler2 -- an R package for gene list functional enrichment analysis and namespace conversion toolset g:Profiler. F1000Res 9 https://doi.org/10.12688/f1000research.24956.2 (2020).
Rahman, A. et al. Whole genome sequence of denitrifying bacterium Stutzerimonas stutzeri strain NGHE31, collected from an eutrophic wetland in Sunamganj, Bangladesh, following the 2017 flash floods. Microbiol. Resour. Announc. 13, e0000124. https://doi.org/10.1128/mra.00001-24 (2024).
Gomila, M., Mulet, M., Garcia-Valdes, E. & Lalucat, J. Genome-based taxonomy of the genus stutzerimonas and proposal of S. frequens sp. nov. and S. degradans sp. nov. and emended descriptions of S. perfectomarina and S. chloritidismutans. Microorganisms 10 https://doi.org/10.3390/microorganisms10071363 (2022).
Gaeta, A. L., Caldwell, K. A. & Caldwell, G. A. Found in translation: the utility of C. elegans alpha-synuclein models of Parkinson’s disease. Brain Sci. 9 https://doi.org/10.3390/brainsci9040073 (2019).
Felton, C. M. & Johnson, C. M. Dopamine signaling in C. elegans is mediated in part by HLH-17-dependent regulation of extracellular dopamine levels. G3 (Bethesda). 4, 1081–1089. https://doi.org/10.1534/g3.114.010819 (2014).
Ward, S. Chemotaxis by the nematode Caenorhabditis elegans: identification of attractants and analysis of the response by use of mutants. Proc. Natl. Acad. Sci. U S A. 70, 817–821. https://doi.org/10.1073/pnas.70.3.817 (1973).
Bargmann, C. I., Hartwieg, E. & Horvitz, H. R. Odorant-selective genes and neurons mediate olfaction in C. elegans. Cell 74, 515–527. https://doi.org/10.1016/0092-8674(93)80053-h (1993).
Kaur, S., Sammi, S. R., Jadiya, P. & Nazir, A. RNAi of cat-2, a putative tyrosine hydroxylase, increases alpha synuclein aggregation and associated effects in transgenic C. elegans. CNS Neurol. Disord Drug Targets. 11, 387–394. https://doi.org/10.2174/187152712800792811 (2012).
Muhammad, F. et al. Neuroprotective effects of cannabidiol on dopaminergic neurodegeneration and alpha-synuclein accumulation in C. elegans models of Parkinson’s disease. Neurotoxicology 93, 128–139. https://doi.org/10.1016/j.neuro.2022.09.001 (2022).
Sherman, D. & Harel, D. Deciphering the underlying mechanisms of the pharyngeal pumping motions in Caenorhabditis elegans. Proc. Natl. Acad. Sci. U S A. 121, e2302660121. https://doi.org/10.1073/pnas.2302660121 (2024).
Maulik, M., Mitra, S., Bult-Ito, A., Taylor, B. E. & Vayndorf, E. M. Behavioral Phenotyping and Pathological Indicators of Parkinson’s Disease in C. elegans Models. Front. Genet. 8, 77. https://doi.org/10.3389/fgene.2017.00077 (2017).
Ayton, S. & Lei, P. Nigral iron elevation is an invariable feature of Parkinson’s disease and is a sufficient cause of neurodegeneration. Biomed. Res. Int. 2014 (581256). https://doi.org/10.1155/2014/581256 (2014).
He, N. et al. Increased iron-deposition in lateral-ventral substantia nigra pars compacta: A promising neuroimaging marker for Parkinson’s disease. Neuroimage Clin. 28, 102391. https://doi.org/10.1016/j.nicl.2020.102391 (2020).
Sun, D. et al. Lipid metabolism in ferroptosis: mechanistic insights and therapeutic potential. Front. Immunol. 16, 1545339. https://doi.org/10.3389/fimmu.2025.1545339 (2025).
Srinivasan, E. et al. Alpha-Synuclein Aggregation in Parkinson’s Disease. Front. Med. (Lausanne). 8, 736978. https://doi.org/10.3389/fmed.2021.736978 (2021).
Teil, M. et al. Targeting alpha-synuclein for PD therapeutics: a pursuit on all fronts. Biomolecules 10 https://doi.org/10.3390/biom10030391 (2020).
Yao, Z. et al. Ferroptosis in Parkinson’s disease -- The iron-related degenerative disease. Ageing Res. Rev. 101, 102477. https://doi.org/10.1016/j.arr.2024.102477 (2024).
Zhou, M. et al. Targeting ferroptosis in Parkinson’s disease: mechanisms and emerging therapeutic strategies. Int. J. Mol. Sci. 25 https://doi.org/10.3390/ijms252313042 (2024).
Devos, D. et al. Therapeutic modalities of deferiprone in Parkinson’s disease: SKY and EMBARK studies. J. Parkinsons Dis. 15, 72–86. https://doi.org/10.1177/1877718X241300295 (2025).
Zeng, W., Cai, J., Zhang, L. & Peng, Q. Iron deposition in Parkinson’s disease: a mini-review. Cell. Mol. Neurobiol. 44 https://doi.org/10.1007/s10571-024-01459-4 (2024).
Chen, Y. et al. The interplay of iron, oxidative stress, and alpha-synuclein in Parkinson’s disease progression. Mol. Med. 31, 154. https://doi.org/10.1186/s10020-025-01208-3 (2025).
66 Chen, Z. T. et al. Evaluation of ferritin and TfR level in plasma neural-derived exosomes as potential markers of Parkinson’s disease. Front. Aging Neurosci. 15, 1216905. https://doi.org/10.3389/fnagi.2023.1216905 (2023).
Friedman, A. & Galazka-Friedman, J. The history of the research of iron in parkinsonian substantia nigra. J. Neural Transm (Vienna). 119, 1507–1510. https://doi.org/10.1007/s00702-012-0894-8 (2012).
Perez-Padilla, V., Molina-Henares, M. A., Udaondo, Z., Ramos-Gonzalez, M. I. & Espinosa-Urgel, M. Genetic basis of biofilm formation and salt adaptation in the plant-beneficial strain Stutzerimonas stutzeri MJL19. Appl. Microbiol. Biotechnol. 109, 130. https://doi.org/10.1007/s00253-025-13523-0 (2025).
Zhang, B. et al. Unlocking N(2)O respiratory pathways in Stutzerimonas stutzeri PRE-2: Implications for reducing N(2)O emissions from estuaries. Mar. Environ. Res. 206, 107044. https://doi.org/10.1016/j.marenvres.2025.107044 (2025).
Devkar, H. U. et al. Antimicrobial Potential of Marine Sponge-Associated Bacillus velezensis and Stutzerimonas stutzeri from the Indian Coast: A Genome Mining and Metabolite Profiling Approach. Curr. Microbiol. 82, 280. https://doi.org/10.1007/s00284-025-04262-6 (2025).
Han, M. et al. Fucoidan-derived carbon dots as nanopenetrants of blood-brain barrier for Parkinson’s disease treatment. J. Colloid Interface Sci. 680, 516–527. https://doi.org/10.1016/j.jcis.2024.10.173 (2025).
Paudel, P., Park, S. E., Seong, S. H., Jung, H. A. & Choi, J. S. Bromophenols from Symphyocladia latiuscula Target Human Monoamine Oxidase and Dopaminergic Receptors for the Management of Neurodegenerative Diseases. J. Agric. Food Chem. 68, 2426–2436. https://doi.org/10.1021/acs.jafc.0c00007 (2020).
Acknowledgements
SS is supported by the University Grants Commission, India. AD is supported by the Council of Scientific & Industrial Research, India. SG is supported by the Ministry of Earth Sciences, India. We gratefully acknowledge the Director of CSIR-CDRI for supporting this work by providing the essential research facilities. The CSIR-CDRI communication number is 11139.
Funding
The work is supported by in-house funding (IHP0046) from the Director, CSIR-CDRI, to SV.
Author information
Authors and Affiliations
Contributions
**SS-** Investigation, Data curation, Validation, Formal analysis, Visualization, Conceptualization, Methodology, Writing—original draft.**AD-** Investigation, Data curation, Formal analysis, Visualization, Methodology, Writing—original draft, **SG-** Investigation; Data curation; Formal analysis; Visualization, Writing—original draft.**ML-** Investigation; Data curation**MP-** Resources; Investigation; Methodology; Writing—original draft, Writing—review & editing.**SV-** Conceptualization; Investigation; Data curation; Formal analysis; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Writing - original draft; and Writing—review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Generative AI statement
While preparing this work, the author(s) used Grammarly to improve language and readability. The author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Singh, S., Damodaran, A., Goswami, S. et al. Marine bacterium Stutzerimonas stutzeri mitigates Parkinson’s disease pathology in C. elegans via ferroptosis modulation. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46881-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46881-4