Abstract
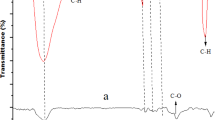
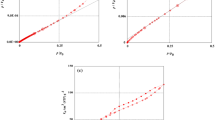
This study explored the potential of acid-treated orange peel (ATOP) and raw orange peel (ROP) as sustainable, low-cost biosorbents for the elimination of phenol from aqueous solutions. Acid modification was employed to enhance the textural and surface chemistry properties of orange peel–derived biomass. Comprehensive characterization using FTIR, SEM, BET, and XRD confirmed successful surface functionalization, improved porosity, and structural modification after adsorption. The findings showed that ATOP demonstrated superior performance relative to ROP, achieving a maximum removal efficiency of 85.25% at 45 °C and reaching equilibrium within 150 min. The maximum adsorption capacities obtained from the Langmuir model were 80.32 mg g−1 for ROP and 133.13 mg g−1 for ATOP. Equilibrium data indicated Langmuir behavior for ROP and Freundlich behavior for ATOP, reflecting increased surface heterogeneity induced by activation. Kinetic assessment showed that phenol removal on ROP followed the pseudo-first-order model, while ATOP was best explained by the pseudo-second-order model, signifying a shift toward stronger surface interactions after acid modification. Thermodynamic parameters established the process to be spontaneous and endothermic, with ΔH° values of 14.5 kJ mol−1 (ROP) and 45.32 kJ mol−1 (ATOP). Overall, the results demonstrate that acid-treated orange peel biomass is an efficient and environmentally benign adsorbent for phenol remediation in wastewater systems.
Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Ahmaruzzaman, M. et al. Phenolic compounds in water: From toxicity and source to sustainable solutions–an integrated review of removal methods, advanced technologies, cost analysis, and future prospects. J. Environ. Chem. Eng. 12(3), 112964. https://doi.org/10.1016/j.jece.2024.112964 (2024).
Mohd, A. Presence of phenol in wastewater effluent and its removal: An overview. Int. J. Environ. Anal. Chem. 102(6), 1362–1384. https://doi.org/10.1080/03067319.2020.1738412 (2022).
Patil, P., Jeppu, G., Vallabha, M. S. & Girish, C. R. Enhanced adsorption of phenolic compounds using biomass-derived high surface area activated carbon: Isotherms, kinetics and thermodynamics. Environ. Sci. Pollut. Res. https://doi.org/10.1007/s11356-024-32971-1 (2024).
Pavithra, K. G. et al. A review on recent advancements in extraction, removal and recovery of phenols from phenolic wastewater: Challenges and future outlook. Environ. Res. 237, 117005. https://doi.org/10.1016/j.envres.2023.117005 (2023).
Sun, J. et al. Oxidative degradation of phenols and substituted phenols in the water and atmosphere: A review. Adv. Compos. Hybrid Mater. 5, 627–640. https://doi.org/10.1007/s42114-022-00435-0 (2022).
Mainali, K. Phenolic compounds contaminants in water: A glance. CTCSE 4(4), 1–3. https://doi.org/10.33552/CTCSE.2020.04.000593 (2020).
Khan, M. N. et al. Synthesis, characterization, and application of Ag-biochar composite for sono-adsorption of phenol. Front. Environ. Sci. 10, 823656. https://doi.org/10.3389/fenvs.2022.823656 (2022).
Njimou, J. R. et al. Biocomposite spheres based on aluminum oxide dispersed with orange-peel powder for adsorption of phenol from batch membrane fraction of olive mill wastewater. Colloid Interface Sci. Commun. 42, 100402. https://doi.org/10.1016/j.colcom.2021.100402 (2021).
Mamman, S. et al. Influence of adsorption parameters on phenolic compounds removal from aqueous solutions: A mini review. Desalin. Water Treat. 320, 100631. https://doi.org/10.1016/j.dwt.2024.100631 (2024).
Ayach, J. et al. Comparing conventional and advanced approaches for heavy metal removal in wastewater treatment: An in-depth review emphasizing filter-based strategies. Polymers 16, 1959. https://doi.org/10.3390/polym16141959 (2024).
Ofudje, E. A. et al. Eggshell derived calcium oxide nanoparticles for Toluidine blue removal. Desalin. Water Treat. 247, 294–308. https://doi.org/10.5004/dwt.2022.28079 (2022).
Haydari, I. et al. Synergistic strategies for phenol removal from olive mill wastewater (OMWW): a combined experimental and theoretical investigation using Chlorococcum sp.-derived CuO nanoparticles. Biomass Bioenergy 192, 107483. https://doi.org/10.1016/j.biombioe.2024.107483 (2025).
Guediri, A. et al. Thermodynamic study and the development of a support vector machine model for predicting adsorption behavior of orange peel-derived beads in wastewater treatment. J. Mol. Liq. 403, 124860. https://doi.org/10.1016/j.molliq.2024.124860 (2024).
Singh, D. Insights into adsorbents: Activated carbon for effective adsorption. Jabirian J. Biointerface Res. Pharm. Appl. Chem. 1, 11–21. https://doi.org/10.55559/jabirian.v1i02.233 (2024).
Ullah, S. et al. Activated carbon derived from biomass for wastewater treatment: Synthesis, application and future challenges. J. Anal. Appl. Pyrolysis 179, 106480. https://doi.org/10.1016/j.jaap.2024.106480 (2024).
Reffas, A. et al. Adsorption of ethyl violet dye in aqueous solution by forest wastes, wild carob. Desalin. Water Treat. 57(21), 9859–9870. https://doi.org/10.1080/19443994.2015.1031707 (2016).
Bouguettoucha, A. et al. Novel activated carbon prepared from an agricultural waste, Stipa tenacissima, based on ZnCl2 activation—characterization and application to the removal of methylene blue. Desalin. Water Treat. 57(50), 24056–24069. https://doi.org/10.1080/19443994.2015.1137231 (2016).
Iheanacho, O. C., Nwabanne, J. T., Obi, C. C. & Onu, C. E. Packed bed column adsorption of phenol onto corn cob activated carbon: Linear and nonlinear kinetics modeling. S. Afr. J. Chem. Eng. 36, 80–93. https://doi.org/10.1016/j.sajce.2021.02.003 (2021).
Singh, R., Dutta, R. K., Naik, D. V., Ray, A. & Kanaujia, P. K. High surface area Eucalyptus wood biochar for the removal of phenol from petroleum refinery wastewater. Environ. Chall. 5, 100353. https://doi.org/10.1016/j.envc.2021.100353 (2021).
Elayadi, F. et al. Experimental and modeling studies of the removal of phenolic compounds from olive mill wastewater by adsorption on sugarcane bagasse. Environ. Chall. 4, 100184. https://doi.org/10.1016/j.envc.2021.100184 (2021).
Bhatia, D. & Saroha, A. K. Biochar derived from pyrolysis of rice straw as an adsorbent for removal of phenol from water. J. Water Process Eng. 59, 105003. https://doi.org/10.1016/j.jwpe.2024.105003 (2024).
Michael-Igolima, U. et al. Modified orange peel waste as a sustainable material for adsorption of contaminants. Materials 16(3), 1092. https://doi.org/10.3390/ma16031092 (2023).
Andriambahiny, R. N. A. et al. A review on the recent advancement of acid modified bio-adsorbents for the removal of methyl orange dye from wastewater treatment. Discov. Chem. 2, 92. https://doi.org/10.1007/s44371-025-00180-5 (2025).
Tang, K. H. D. Valorization of organic waste as biosorbents for wastewater treatment. Water Emerg. Contam. Nanoplastics 3, 25. https://doi.org/10.20517/wecn.2024.53 (2024).
Kalengyo, R. B. et al. Utilizing orange peel waste biomass in textile wastewater treatment and its recyclability for dual biogas and biochar production: A techno-economic sustainable approach. Biomass Conv. Bioref 14, 19875–19888. https://doi.org/10.1007/s13399-023-04111-1 (2024).
Kumar, L. et al. Adsorption of phenol from aqueous solution using activated char synthesized by one-step and two-step KOH activation and pyrolysis of orange peel. Water Air Soil Pollut. 236, 803. https://doi.org/10.1007/s11270-025-08431-y (2025).
Mesquita, M. D. S., Tanabe, E. H. & Bertuol, D. A. Adsorption of phenol using Eucalyptus saligna biochar activated with NiCl2. Water Air Soil Pollut. 235(5), 311. https://doi.org/10.1007/s11270-024-07049-w (2024).
Ezzati, R., Azizi, M. & Ezzati, S. A theoretical approach for evaluating the contributions of pseudo-first-order and pseudo-second-order kinetics models in the Langmuir rate equation. Vacuum 222, 113018. https://doi.org/10.1016/j.vacuum.2023.113018 (2024).
Guo, X. & Wang, J. A novel monolayer adsorption kinetic model based on adsorbates “infect” adsorbents inspired by epidemiological model. Water Res. 253, 121313. https://doi.org/10.1016/j.watres.2024.121313 (2024).
Din, S. U. et al. Sorghum husks as potential low cost adsorbent for Congo red adsorption. Sci. Rep. 15(1), 38265. https://doi.org/10.1038/s41598-025-22082-3 (2025).
Fito, J. et al. Adsorption of methylene blue from textile industrial wastewater using activated carbon developed from Rumex abyssinicus plant. Sci. Rep. 13, 5427. https://doi.org/10.1038/s41598-023-32431-8 (2023).
Nafisyah, E., Arrisujaya, D. & Susanti, E. The utilization of water hyacinth (Eichhornia crassipes) harvested from the phytoremediation process as activated carbon in Cr(VI) adsorption. IOP Conf. Ser. Earth Environ. Sci. 1211, 012019. https://doi.org/10.1088/1755-1315/1211/1/012019 (2023).
Ananpreechakorn, W. & Seetawan, T. Synthesis and characterization of activated carbon from water hyacinth. J. Phys. Conf. Ser. 2013, 012025. https://doi.org/10.1088/1742-6596/2013/1/012025 (2021).
Ghosh, N., Sen, S., Biswas, G., Saxena, A. & Haldar, P. K. Adsorption and desorption study of reusable magnetic iron oxide nanoparticles modified with Justicia adhatoda leaf extract for the removal of textile dye and antibiotic. Water Air Soil Pollut. 234(3), 202. https://doi.org/10.1007/s11270-023-06217-8 (2023).
Chebli, D. et al. Valorization of an agricultural waste, Stipa tenassicima fibers, by biosorption of an anionic azo dye, Congo red. Desalin. Water Treat. 54, 245–254. https://doi.org/10.1080/19443994.2014.880154 (2015).
Bouguettoucha, A. et al. The use of a forest waste biomass, cone of Pinus brutia for the removal of an anionic azo dye Congo red from aqueous medium. Desalin. Water Treat. 55, 1956–1965. https://doi.org/10.1080/19443994.2014.928235 (2015).
Kori, A. K., Ramavandi, B., Mahmoodi, S. M. M. & Javanmardi, F. Magnetization and ZIF-67 modification of Aspergillus flavus biomass for tetracycline removal from aqueous solutions: A stable and efficient composite. Environ. Res. 252(Pt 2), 118931. https://doi.org/10.1016/j.envres.2024.118931 (2024).
Chikri, R., Elhadiri, N., Benchanaa, M. & Maguana, Y. Efficiency of sawdust as low-cost adsorbent for dyes removal. J. Chem. 2020, 1–17. https://doi.org/10.1155/2020/6902409 (2020).
Jawad, A. H., Abdulhameed, A. S. & Mastuli, M. S. Acid-factionalized biomass material for methylene blue dye removal: A comprehensive adsorption and mechanism study. J. Taibah Univ. Sci. 14(1), 305–313. https://doi.org/10.1080/16583655.2020.1736767 (2020).
Oyetade, J. A., Machunda, R. L. & Hilonga, A. Fenton-mediated solar-driven photocatalysis of industrial dye effluent with polyaniline impregnated with activated TiO2-Nps. J. Photochem. Photobiol. A Chem. 20, 100231. https://doi.org/10.1016/j.jpap.2024.100231 (2024).
Djebbar, M., Djafri, F., Bouchekara, M. & Djafri, A. Adsorption of phenol on natural clay. Appl. Water Sci. 2(2), 77–86. https://doi.org/10.1007/s13201-012-0031-8 (2012).
Sarici-Ozdemir, Ç. & Kiliç, F. Kinetics behavior of methylene blue onto agricultural waste. Part. Sci. Technol. 36(2), 194–201. https://doi.org/10.1080/02726351.2016.1240127 (2018).
Albert, A. A., Chenping, G., Runping, H. & Lingbo, Q. Functionalized magnetic biocomposite based on peanut husk for the efficient sequestration of basic dyes in single and binary systems: Adsorption mechanism and antibacterial study. J. Environ. Chem. Eng. 10(4), 108205. https://doi.org/10.1016/j.jece.2022.108205 (2022).
Ogundiran, A. A., Ofudje, E. A., Ogundiran, O. O. & Adewusi, A. M. Cationic dye adsorptions by eggshell waste: Kinetics, isotherms and thermodynamics studies. Desalin. Water Treat. 280, 157–167. https://doi.org/10.5004/dwt.2022.29080 (2022).
Somsesta, N., Sricharoenchaikul, V. & Aht-Ong, D. Adsorption removal of methylene blue onto activated carbon/cellulose biocomposite films: Equilibrium and kinetic studies. Mater. Chem. Phys. 240, 122221. https://doi.org/10.1016/j.matchemphys.2019.122221 (2020).
Damahe, D., Mayilswamy, N. & Kandasubramanian, B. Biochar/metal nanoparticles-based composites for dye remediation: A review. Hybrid Adv. 6, 100254. https://doi.org/10.1016/j.hybadv.2024.100254 (2024).
Djama, C. et al. Experimental and Theoretical study of methylene blue adsorption on a new raw material, Cynara scolymus—a statistical physics assessment. Sustainability 15, 10364. https://doi.org/10.3390/su15131036 (2023).
El-Bery, H. M. et al. High adsorption capacity of phenol and methylene blue using activated carbon derived from lignocellulosic agriculture wastes. Sci. Rep. 12, 5499. https://doi.org/10.1038/s41598-022-09475-4 (2022).
Lütke, S. F. et al. Preparation of activated carbon from black wattle bark waste and its application for phenol adsorption. J. Environ. Chem. Eng. 7, 103396. https://doi.org/10.1016/j.jece.2019.103396 (2019).
Ngueagni, P. T. et al. Cellulose-based adsorbent of animal waste for the adsorption of lead and phenol. BioResources 20(2), 3923–3952. https://doi.org/10.15376/biores.20.2.3923-3952 (2025).
Abdel-Gawwad, H. A. et al. Utilization of red clay brick waste in the green preparation of an efficient porous nanocomposite for phenol adsorption: Characterization, experiments and statistical physics treatment. Sustain. Chem. Pharm. 32, 101027. https://doi.org/10.1016/j.scp.2023.101027 (2023).
Yilmaz, P., Gunduzm, D. & Ozbek, B. Utilization of low-cost bio-waste adsorbent for methylene blue dye removal from aqueous solutions and optimization of process variables by response surface methodology approach. Desalin. Water Treat. 224, 367–388. https://doi.org/10.5004/dwt.2021.27206 (2021).
Fseha, Y. H., Shaheen, J. & Sizirici, B. Phenol contaminated municipal wastewater treatment using date palm frond biochar: Optimization using response surface methodology. Emerg. Contam. 9(1), 100202. https://doi.org/10.1016/j.emcon.2022.100202 (2023).
Michael-Igolima, U., Abbey, S. J., Ifelebuegu, A. O. & Eyo, E. U. Modified orange peel waste as a sustainable material for adsorption of contaminants. Materials (Basel) 16(3), 1092. https://doi.org/10.3390/ma16031092 (2023).
Kumar, L. et al. Systematic studies on the effect of structural modification of orange peel for remediation of phenol contaminated water. Water Environ. Res. 95(5), e10872. https://doi.org/10.1002/wer.10872 (2023).
Tolkou, A. K., Tsoutsa, E. K., Kyzas, G. Z. & Katsoyiannis, I. A. Sustainable use of low-cost adsorbents prepared from waste fruit peels for the removal of selected reactive and basic dyes found in wastewaters. Environ. Sci. Pollut. Res. Int. 31(10), 14662–14689. https://doi.org/10.1007/s11356-024-31868-3 (2024).
Liu, L. et al. Development of efficient biochar produced from orange peel for effective La(III) and Y(III) adsorption. Adsorpt. Sci. Technol. 2023, 5519783. https://doi.org/10.1155/2023/551978 (2023).
Rana, S., Rana, R. S. & Suresh, S. Studies of biosorption kinetics of phenol by orange peel and tea waste. Dig. J. Nanomater. Biostruct. 12(2), 579–588 (2017).
Hasdemir, İM. et al. Adsorption of acetic acid onto activated carbons produced from hazelnut shell, orange peel, and melon seeds. Appl. Water Sci. 12, 271. https://doi.org/10.1007/s13201-022-01797-y (2022).
Kumar, L. et al. Adsorption of aminophenol from aqueous solution using KOH pretreated biochar derived from orange peel pyrolysis: Optimization, kinetics, and isotherm study. Environ. Process. 12, 36. https://doi.org/10.1007/s40710-025-00775-1 (2025).
Ali, H. J., Al-Heetim, D. T. A. & Abd Rashid, R. Biochar from orange (Citrus sinensis) peels by acid activation for methylene blue adsorption. Iran. J. Chem. Chem. Eng. 38(2), 91–105 (2019).
Zhou, Y. et al. Adsorption and mechanism study for phenol removal by 10% CO2 activated bio-char after acid or alkali pretreatment. J. Environ. Manag. 348, 119317. https://doi.org/10.1016/j.jenvman.2023.119317 (2023).
Monica, K., Roopa, D. & Stephan, B. Utilisation of low-cost adsorbent made from agro-waste based material for the removal of phenol from aqueous solutions. Int. J. Innov. Res. Sci. Eng. Technol. 14(7), 140. https://doi.org/10.15680/IJIRSET.2025.1407022 (2025).
Acknowledgements
Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2026R13), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Funding
This work was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2026R13), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Zahrah Alqahtani carried out the characterizations and statistical analysis, Oladejo Emmanuel Kola conceived the work, and performed the experiments, Aliyah Alsharif read the first draft and performed statistical analysis, Ali Shawabkeh and Afnan M. Alnajeebi provide resources, and performed data analysis, Roaa A. Tayeb performed statistical analysis and contributed resources, Amani Fahm Mohammed Al Solami and Hamad AlMohamadi performed data analysis, contributed in characterization, and contributed resources, Jamelah S. Al-Otaibi contributed financial assistance, performed statistical analysis, and resources.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
N/A.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Alqahtani, Z., Kola, O.E., Alsharif, A. et al. Acid activated orange peel waste adsorbent for the elimination of phenol with insights into isotherm, kinetics, and thermodynamics. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46890-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46890-3