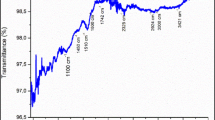
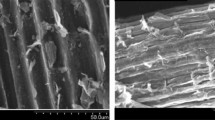
Abstract
This study explored the adsorption of methylene blue (MB) from aqueous solutions using natural clay as an adsorbent. The effects of contact time, initial dye concentration, adsorbent dosage, and pH were systematically evaluated. Analysis of correlation coefficients (R2) for the kinetic and isotherm models indicated that the adsorption kinetics follow a pseudo-first-order model, while the Langmuir isotherm provided the best fit to the experimental data at 303, 313, and 323 K. To further elucidate the adsorption mechanism, a generalized model based on statistical physics was applied. Steric analysis examined the number of dye molecules bound per active site, the estimated number of layers formed, receptor site density, and overall adsorption capacity. This model also yielded two adsorption energies associated with MB-clay and MB-MB interactions, ranging from 29.845 to 31.191 kJ/mol and from 21.981 to 24.692 kJ/mol, respectively. Thermodynamic parameters confirmed that MB adsorption is spontaneous under all studied conditions. Overall, these results highlight natural clay as a cost-effective and efficient adsorbent for removing dyes from aqueous solutions.
Similar content being viewed by others
Data availability
Data available from the corresponding author on reasonable request.
References
Rashid, R. A., Ishak, M. A. M. & Mohamed, K. Adsorptive removal of methylene blue by commercial coconut shell activated carbon. Sci. Lett. 12(1), 77–101 (2018).
Bouguettouchaa, A., Reffasb, A., Cheblia, D., Mekhalifc, T. & Amraned, A. Novel activated carbon prepared from an agricultural waste, Stipa tenacissima, based on ZnCl2 activation—characterization and application to the removal of methylene blue. Desalin. Water Treat. 57, 24056–24069 (2016).
Hajjaji, W., Andrejkovičová, A., Pullar, R. C. & Tobaldi, D. M. Effective removal of anionic and cationic by kaolinite and TiO2/kaolinite composite. Clay Miner. 51, 19–27 (2016).
Reffas, A., Bouguettoucha, A., Chebli, D. & Amrane, A. Adsorption of ethyl violet dye in aqueous solution by forest wastes, wild carob. Desalin. Water Treat. 57(21), 9859–9870 (2016).
Kausar, A. et al. Dyes adsorption using clay and modified clay: a review. J. Mol. Liq. 256, 395–407 (2018).
Bounaas, M. et al. High efficiency of methylene blue removal using a novel low-cost acid treated forest wastes, Cupressus semperirens cones: Experimental results and modeling. Particulate Sci. Technol 37(4), 504–513 (2019).
Wang, C.-C., Juang, L.-C. & Hsu, T.-C. Adsorption of basic dyes onto montmorillonite. J. Colloid Interface Sci. 273, 80–86 (2004).
Han, X., Wang, W. & Ma, X. Adsorption characteristics of methylene blue onto low cost biomass material lotus leaf. Chem. Eng. J. 171, 1–8 (2011).
Kheirabadi, M. et al. Welldesigned Ag/ZnO/3D graphene structure for dye removal: Adsorption, photocatalysis and physical separation capabilities. J. Colloid Interface Sci. 537, 66–78 (2019).
Liu, J., Liu, A., Wang, W., Li, R. & Zhang, W. X. Feasibility of nanoscale zero-valent iron (nZVI) for enhanced biological treatment of organic dyes. Chemosphere 237, 124470 (2019).
Meshko, V., Markovska, L. & Mincheva, M. Adsorption of basic dyes on granular activated carbon and natural zeolite. Water Res. 35, 3357–3366 (2001).
Sarma, G. K., SenGupta, S. & Bhattacharyya, K. G. Methylene blue adsorption on natural and modified clays. Sep. Sci. Technol. 46, 1602–1614 (2011).
Bessaha F, Bessaha G, Benhouria A, Benalioua B, Bezzekhami MA, Bendahma F. Application of eco-friendly material as an inexpensive adsorbent for methyl violet dye removal: experimental, response surface methodology and statistical physics. J. Dispers. Sci. Technol. (2024).
Bessaha, F. et al. Efficient removal of a pharmaceutical compound on organoclay: batch experiment, DFT calculation, statistical physics, and modeling. Chem. Eng. Commun. https://doi.org/10.1080/00986445.2024.2428964 (2025).
Bessahaa, F. et al. Highly efficient batch adsorption of anionic dye in wastewater using nanocomposite: Experimental and theoretical studies. Desalin. Water Treat. 317, 100292 (2024).
Majouri, N., El Mankibi, M. & Sghaier, J. Unlocking the potential of Tunisian clays: Sustainable characterization for construction applications. ACS Omega 10, 28874–28886 (2025).
Hamdi, S. et al. Tetracycline adsorption/desorption by raw and activated Tunisian clays. Enviro. Res. 242, 117536 (2024).
Rida, K. SarraBouraoui, Selma Hadnine, adsorption of methylene blue from aqueous solution by kaolin and zeolite. Appl. Clay Sci. 83–84, 99–105 (2013).
Hamza, W., Dammak, N., Hadjltaief, H. B., Eloussaief, M. & Benzina, M. Sono-assistedadsorption of cristal violet dye onto Tunisian smectite clay: Characterization, kinetics andadsorption isotherms. Ecotoxicol. Environ. Saf. 163, 365–371 (2018).
Pawar, R. R., Gupta, P., Sawant, S. Y., Shahmoradi, B. & Lee, S. M. Porous synthetic hectoriteclay-alginate composite beads for effective adsorption of methylene blue dye from aqueoussolution. Int. J. Biol. Macromol. 114, 1315–1324 (2018).
Xie, S. et al. Application of clay minerals as adsorbents for removing heavy metals from the environment. Green Smart Min. Eng. 1, 249–261 (2024).
Jawad, A. H. & Abdulhameed, A. S. Mesoporous Iraqi red kaolin clay as an efficient adsorbent for methylene blue dye: adsorption kinetic, isotherm and mechanism study. Surf. Interfaces 18, 100422 (2020).
Boukhemkhem, A. & Rida, K. Improvement adsorption capacity of methylene blue onto modified Tamazertkaolin. Adsorpt. Sci. Technol. 35(9–10), 753–773 (2017).
Arab, P. B., Araújo, T. P. & Pejon, O. J. Identification of clay minerals in mixtures subjected to differential thermal and thermogravimetry analyses and methylene blueadsorption tests. Appl. Clay Sci. 114, 133–140 (2015).
El Mouzdahir, Y., Elmchaouri, A., Mahboub, R., Gil, A. & Korili, S. A. Equilibrium modeling for the adsorption of methylene blue from aqueous solutions on activated clay minerals. Desalination 250, 335–338 (2010).
Khan, M. I. Adsorption of methylene blue onto natural Saudi Red Clay: Isotherms, kinetics and thermodynamic studies. Mater. Res. Express. 7, 055507 (2020).
Rida, K., Bouraoui, S. & Hadnine, S. Adsorption of methylene blue from aqueous solution by kaolin and zeolite. Appl. Clay Sci. 83–84, 99–105 (2013).
Removal of Organic Matter using Douiret clay (Tataouine, Tunisia) — adsorption kinetics and thermodynamics. Mater. (Basel) (2023).
Loutfi, M., Mariouch, R., Mariouch, I., Belfaquir, M. & ElYoubi, M. S. Adsorption of methylene blue dye from aqueous solutions onto natural clay: equilibrium and kinetic studies. Mater. Today 72, 3638–3643 (2023).
Dehmani, Y. et al. A critical review of natural clay minerals: structural characterization, textural properties, and adsorption mechanisms for sustainable wastewater treatment. Mater. Today Adv. 29, 100682 (2026).
Djama, C. et al. Experimental and theoretical study of methylene blue adsorption on a new raw material, Cynara scolymus-a statistical physics assessment. Sustainability 15, 10364 (2023).
Dehmani, Y. et al. Comparison of phenol adsorption property and mechanism onto different moroccan clays. Water 15, 1881 (2023).
Elhassan, M., Kooh, M. R. R., Chau, Y.-F. & Abdullah, R. Hydrochar from shorea spp.: a dual-purpose approach for sustainable biofuel and efficient methylene blue adsorbent. Biomass Convers. Biorefinery https://doi.org/10.1007/s13399-024-05376-w (2025).
Kooh, M. R. R., Thotagamuge, R., Chau, Y.-F., Mahadi, A. H. & Lim, C. M. Machine learning approaches to predict adsorption capacity of Azolla pinnata in the removal of methylene blue. J. Taiwan Inst. Chem. Eng. 132, 104134 (2022).
Suhaimi, N. et al. The use of gigantochloa bamboo-derived biochar for the removal of methylene blue from aqueous solution. Adsorption Sci. Technol. 2022, 8245797 (2022).
Bouzgarrou, S. M. et al. Experiments and physical Investigation on the adsorption of methylene on activated carbon. Sci. Rep. 15, 45075 (2025).
Guediri, A. et al. Thermodynamic study and the development of a support vector machine model for predicting adsorption behavior of orange peel-derived beads in wastewater treatment. J. Mol. Liq. 403, 124860 (2024).
Chen, H. et al. Removal capacity and adsorption mechanism of heat-treated palygorskite clay for methylene blue. Chem. Eng. J. 174, 143–150 (2011).
Sapawe, N. et al. Cost-effective microwave rapid synthesis of zeolite NaA for removal of methylene blue. Chem. Eng. J. 229, 388–398 (2013).
Yavuz, O. & Saka, C. Surface modiEcation with cold plasma application on kaolin and its effects on the adsorption of methylene blue. Appl. Clay Sci. 85, 96–102 (2013).
Fil, A. B., Ozmetin, C. & Korkmaz, M. Cationic dye (methylene blue) removal from aqueous solution by montmorillonite. Bull. Korean Chem. Soc. 33, 3184–3190 (2012).
Bée, A., Obeid, L., Mbolantenaina, R., Welschbillig, M. & Talbot, D. Magnetic chitosan/clay beads: a magsorbent for the removal of cationic dye from water. J. Magn. Magn. Mater. 421, 59–64 (2017).
Auta, M. & Hameed, B. H. ModiEed mesoporous clay adsorbent for adsorption isotherm and kinetics of methylene blue. Chem. Eng. J. 198–199, 219–227 (2012).
Acknowledgements
The authors gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through Project number: (JU-202502103-DGSSR-ORA-2025).
Author information
Authors and Affiliations
Contributions
Jedli Hedi: Conceptualization, data curation, investigationSouhail Mohammed bouzgarrou, Rym methodology: Validation, visualization. Ehab Sabi: Acquisition Khalifa Slimi: Writing–review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jedli, H., Bouzgarrou, S.M., Hassani, R. et al. Adsorption of methylene blue onto clay: experiments and statistical physics interpretation. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46966-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-46966-0