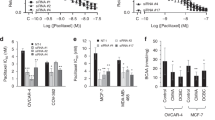
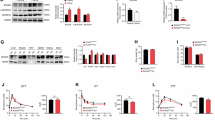
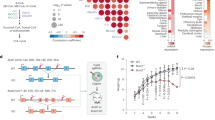
Abstract
Elevated plasma branched-chain amino acid (BCAA) levels are observed in some metabolic disorders, such as obesity and diabetes, due to impaired hepatic BCAA catabolism. In pancreatic cancer, elevated plasma BCAA levels are observed years before diagnosis, possibly due to increased protein degradation. However, the status of BCAA catabolism in pancreatic cancer remains unclear. Here, we hypothesized that defective hepatic BCAA utilization contributes to this elevation in pancreatic cancer, similar to observations in obesity and diabetes. Consistent with our hypothesis, increased phosphorylation of the hepatic BCAA catabolic enzyme, branched-chain α-keto acid dehydrogenase (BCKDH), was observed in a murine nerve invasion model with human pancreatic cancer cells, indicative of an inactive state. Notably, this was associated with reduced whole-body BCAA flux, increased skeletal muscle degradation, and elevated plasma BCAA levels. Patients with stage III pancreatic cancer with severe neural invasion exhibited high plasma BCAA and reduced muscle mass. In addition, patients with stage IV pancreatic cancer showed hepatic BCKDH phosphorylation, which correlated with poor prognosis. Overall, these results suggest the potential existence of impaired BCAA catabolism via hepatic BCKDH downregulation in pancreatic cancer.
Similar content being viewed by others
Data availability
Qualified researchers can contact Shuichi Mitsunaga to request individual-level patient data. The datasets from the animal experiments in the current study are available from Asami Hagiwara upon reasonable request.
References
Sivanand, S. & Vander Heiden, M. G. Emerging roles for branched-chain amino acid metabolism in cancer. Cancer Cell 37, 147–156 (2020).
Mansoori, S., Ho, M. Y., Ng, K. K. & Cheng, K. K. Branched-chain amino acid metabolism: Pathophysiological mechanism and therapeutic intervention in metabolic diseases. Obes. Rev. 26, e13856 (2024).
Herman, M. A., She, P., Peroni, O. D., Lynch, C. J. & Kahn, B. B. Adipose tissue branched chain amino acid (BCAA) metabolism modulates circulating BCAA levels. J. Biol. Chem. 285, 11348–11356 (2010).
Harris, R. A., Joshi, M. & Jeoung, N. H. Mechanisms responsible for regulation of branched-chain amino acid catabolism. Biochem. Biophys. Res. Commun. 313, 391–396 (2004).
Lu, G. et al. Protein phosphatase 2cm is a critical regulator of branched-chain amino acid catabolism in mice and cultured cells. J. Clin. Invest. 119, 1678–1687 (2009).
Sketcher, R. D., Fern, E. B. & James, W. P. The adaptation in muscle oxidation of leucine to dietary protein and energy intake. Br. J. Nutr. 31, 333–342 (1974).
Felig, P., Marliss, E. & Cahill, G. F. Jr. Plasma amino acid levels and insulin secretion in obesity. N. Engl. J. Med. 281, 811–816 (1969).
Shah, S. H. et al. Association of a peripheral blood metabolic profile with coronary artery disease and risk of subsequent cardiovascular events. Circ. Cardiovasc. Genet. 3, 207–214 (2010).
Tizianello, A. et al. Branched-chain amino acid metabolism in chronic renal failure. Kidney Int. Suppl. 16, S17–S22 (1983).
Holeček, M. Branched-chain amino acids in health and disease: Metabolism, alterations in blood plasma, and as supplements. Nutr. Metab. (Lond.) 15, 33 (2018).
Lian, K. et al. Impaired adiponectin signaling contributes to disturbed catabolism of branched-chain amino acids in diabetic mice. Diabetes 64, 49–59 (2015).
Neinast, M. D. et al. Quantitative analysis of the whole-body metabolic fate of branched-chain amino acids. Cell Metab. 29, 417–429 (2019).
Hattori, A. et al. Cancer progression by reprogrammed BCAA metabolism in myeloid leukaemia. Nature 545, 500–504 (2017).
Zhang, L. & Han, J. Branched-chain amino acid transaminase 1 (BCAT1) promotes the growth of breast cancer cells through improving mTOR-mediated mitochondrial biogenesis and function. Biochem. Biophys. Res. Commun. 486, 224–231 (2017).
Mayers, J. R. et al. Elevation of circulating branched-chain amino acids is an early event in human pancreatic adenocarcinoma development. Nat. Med. 20, 1193–1198 (2014).
Imoto, A. et al. Neural invasion induces cachexia via astrocytic activation of neural route in pancreatic cancer. Int. J. Cancer 131, 2795–2807 (2012).
Mitsunaga, S. et al. Low serum level of cholinesterase at recurrence of pancreatic cancer is a poor prognostic factor and relates to systemic disorder and nerve plexus invasion. Pancreas 36, 241–248 (2008).
Mitsunaga, S. et al. Detail histologic analysis of nerve plexus invasion in invasive ductal carcinoma of the pancreas and its prognostic impact. Am. J. Surg. Pathol. 31, 1636–1644 (2007).
Hutson, S. M., Sweatt, A. J. & Lanoue, K. F. Branched-chain [corrected] amino acid metabolism: Implications for establishing safe intakes. J. Nutr. 135, 1557S-1564S (2005).
Mitchell, W. K. et al. Human skeletal muscle protein metabolism responses to amino acid nutrition. Adv. Nutr. 7, 828S-838S (2016).
von Haehling, S., Ebner, N. & Anker, S. D. The Journal of Cachexia, Sarcopenia and Muscle in 2019. J. Cachexia Sarcopenia Muscle 10, 715–720 (2019).
Fearon, K. et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 12, 489–495 (2011).
Cala, M. P. et al. Multiplatform plasma fingerprinting in cancer cachexia: a pilot observational and translational study. J. Cachexia Sarcopenia Muscle 9, 348–357 (2018).
Liu, S. et al. Elevated branched-chain alpha-keto acids exacerbate macrophage oxidative stress and chronic inflammatory damage in type 2 diabetes mellitus. Free Radic. Biol. Med. 175, 141–154 (2021).
Ortega, M. A. et al. Oxidative stress markers are associated with a poor prognosis in patients with pancreatic cancer. Antioxidants 11, 759 (2022).
Qiu, X., Hou, Q. H., Shi, Q. Y., Jiang, H. X. & Qin, S. Y. Identification of hub prognosis-associated oxidative stress genes in pancreatic cancer using integrated bioinformatics analysis. Front. Genet. 11, 595361 (2020).
Martinez-Useros, J., Li, W., Cabeza-Morales, M. & Garcia-Foncillas, J. Oxidative stress: A new target for pancreatic cancer prognosis and treatment. J. Clin. Med. 6, 29 (2017).
Danai, L. V. et al. Altered exocrine function can drive adipose wasting in early pancreatic cancer. Nature 558, 600–604 (2018).
Woo, S. M. et al. Efficacy of pancreatic exocrine replacement therapy for patients with unresectable pancreatic cancer in a randomized trial. Pancreatology 16, 1099–1105 (2016).
Li, J. et al. Tumor cell-intrinsic factors underlie heterogeneity of immune cell infiltration and response to immunotherapy. Immunity 49, 178-193.e7 (2018).
Suzuki, H. et al. Clinical and tumor characteristics of patients with high serum levels of growth differentiation factor 15 in advanced pancreatic cancer. Cancers (Basel) 13, 4842 (2021).
Mitsunaga, S. et al. Fibroblast inhibition by tocilizumab enabled gemcitabine/nab-paclitaxel rechallenge for pancreatic cancer. Cancer Sci. 114, 4006–4019 (2023).
Mitsunaga, S. et al. Nerve invasion distance is dependent on laminin gamma2 in tumors of pancreatic cancer. Int. J. Cancer. 127, 805–819 (2010).
Ayala, J. E. et al. Hyperinsulinemic-euglycemic clamps in conscious, unrestrained mice. J. Vis. Exp. 57, 3188 (2011).
Acknowledgments
We thank Kayo Takei, Miho Akimoto, and Yuriko Sato at the National Cancer Center Hospital East and Michiko Hamamoto at the Exploratory Oncology Research & Clinical Trial Center at the National Cancer Center for their secretarial support; Natsuko Maruyama and Mariko Yajima at the Exploratory Oncology Research & Clinical Trial Center at the National Cancer Center; Toshie Yoshino and Ru Wang in WDB eureka for their support with the experiment; and Miyu Narita at the Tsuruoka Metabolomics Laboratory at the National Cancer Center for her technical assistance. We would like to thank Editage (www.editage.jp) for English language editing. This work was supported in part by research funds from the Yamagata Prefectural Government and City of Tsuruoka.
Funding
This work was supported in part by research funds from the Yamagata Prefectural Government and City of Tsuruoka.
Author information
Authors and Affiliations
Contributions
H **. Michida** : Formal analysis, investigation, methodology, and writing—original draft. **A.H.** : Conceptualization, formal analysis, investigation, methodology, writing—original draft, writing—review & editing, and project administration. **Y.S.** : Investigation, methodology, writing, review & editing. **N.K.** and **M. Kanda** : Formal analysis and investigation. **M. Kawasaki** and **S.K.** : Formal analysis and methodology. **S.S.** : Formal analysis, investigation, writing—original draft and writing—review & editing. **M.I.** : Resources, methodology, writing, review & editing. **H. Makinoshima:** Investigation, methodology, writing—original draft and writing—review & editing. **S.M.** : Conceptualization, formal analysis, investigation, methodology, validation, writing—original draft, writing—review & editing, project administration, and supervision. All authors have read and approved the final version of this manuscript.
Corresponding author
Ethics declarations
Competing interests
H. Michida, A.H., Y.S., N.K., M. Kanda, M. Kawasaki, and S. K. are employed by Ajinomoto Co. Inc. S.M. reports grants from Ajinomoto, Ono Pharmaceutical, and Pfizer Inc. outside the submitted work; lecture fees from Ono Pharmaceutical, Otsuka Pharmaceutical, and Meiji Seika Pharmaceutical outside the submitted work; and a patent for a biomarker using free plasma amino acids to predict sarcopenia outside the submitted work. M.I. reports grants from Boehringer Ingelheim, Delta-Fly Pharma, N.V., Invitrogen, NIHON SERVIER, and Novartis outside the submitted work; consulting fees from Astellas Pharma, Novartis, and NIHON SERVIER outside the submitted work; and lecture fees from Guardant Health Japan, NIHON SERVIER, Taiho, Nippon Kayaku, and Yakult outside the submitted work. The authors declare no other competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Michida, H., Hagiwara, A., Saito, Y. et al. Branched-chain amino acid catabolism is potentially deficient in pancreatic cancer with high-neural invasion. Sci Rep (2026). https://doi.org/10.1038/s41598-026-47741-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-47741-x